Physicochemical Properties
| Molecular Formula | C17H14O6 |
| Molecular Weight | 314.2895 |
| Exact Mass | 314.079 |
| CAS # | 158732-59-3 |
| PubChem CID | 10903113 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 636.8±55.0 °C at 760 mmHg |
| Flash Point | 352.9±28.0 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.829 |
| Source | Salvia miltiorrhiza, Vicia faba, and Melissa officinalis |
| LogP | 3.95 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 23 |
| Complexity | 460 |
| Defined Atom Stereocenter Count | 0 |
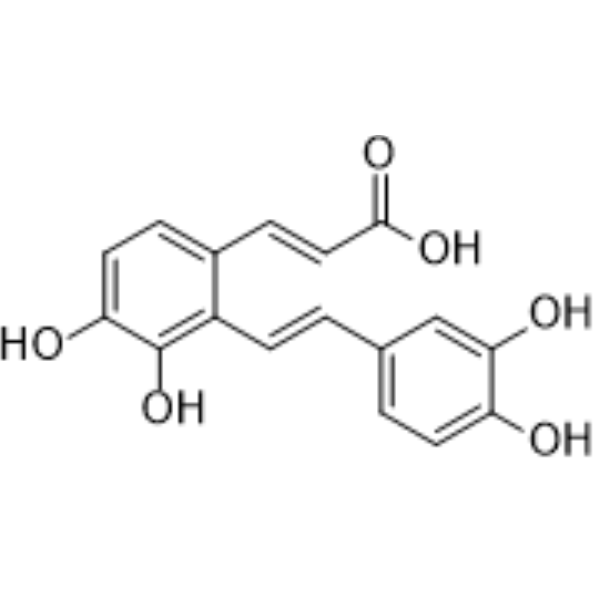
| SMILES | C1=CC(=C(C=C1/C=C/C2=C(C=CC(=C2O)O)/C=C/C(=O)O)O)O |
| InChi Key | PULWRMOKQNWQBD-LZSLGQGWSA-N |
| InChi Code | InChI=1S/C17H14O6/c18-13-6-2-10(9-15(13)20)1-5-12-11(4-8-16(21)22)3-7-14(19)17(12)23/h1-9,18-20,23H,(H,21,22)/b5-1+,8-4+ |
| Chemical Name | (E)-3-[2-[(E)-2-(3,4-dihydroxyphenyl)ethenyl]-3,4-dihydroxyphenyl]prop-2-enoic acid |
| Synonyms | salvianolic acid F; 158732-59-3; (E)-3-[2-[(E)-2-(3,4-dihydroxyphenyl)ethenyl]-3,4-dihydroxyphenyl]prop-2-enoic acid; Salvianolate F; (E)-3-(2-((E)-2-(3,4-dihydroxyphenyl)ethenyl)-3,4-dihydroxyphenyl)prop-2-enoic acid; 2-Propenoic acid,3-[2-[(1E)-2-(3,4-dihydroxyphenyl)ethenyl]-3,4-dihydroxyphenyl]-, (2E)-; salvianolicacidF; (2E)-3-[2-[(1E)-2-(3,4-Dihydroxyphenyl)ethenyl]-3,4-dihydroxyphenyl]-2-propenoic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product |
| ln Vitro | Salvianolic acids, the most effective and abundant compounds extracted from Salvia miltiorrhiza (Danshen), are well known for its good anti-oxidative activity. Danshen has been extensively used as a traditional medicine to treat cardiovascular-related diseases in China and other Asian countries for hundreds of years. Recently, more and more studies have demonstrated that salvianolic acids also have a good effect on the alleviation of fibrosis disease and the treatment of cancer. In vivo and in vitro experiments have demonstrated that salvianolic acids can modulate signal transduction within fibroblasts and cancer cells. It is discovered that the cancer treatment of salvianolic acids is not only because salvianolic acids promote the apoptosis of cancer cells, but also due to the inhibition of cancer-associated epithelial-mesenchymal transition processes. In this article, we review a variety of studies focusing on the comprehensive roles of salvianolic acids in the treatment of fibrosis disease and cancer. These perspectives on the therapeutic potential of salvianolic acids highlight the importance of these compounds, which could be the novel and attractive drugs for fibrosis disease and cancer. [2] |
| References |
[1]. Quality assessment of radix salviae miltiorrhizae. Chem Pharm Bull (Tokyo). 2005 May;53(5):481-6. [2]. Salvianolic Acids: Potential Source of Natural Drugs for the Treatment of Fibrosis Disease and Cancer. Front Pharmacol. 2019; 10: 97. |
| Additional Infomation |
salvianolic acid F has been reported in Salvia miltiorrhiza, Vicia faba, and Melissa officinalis with data available.
This paper describes an improved quality assessment method for Radix Salviae Miltiorrhizae (Root of Salvia miltiorrhiza BGE.) which was established using chromatographic fingerprinting and quantification of multiple marker compounds in the crude drug. High-performance thin-layer chromatography (HPTLC) fingerprinting of water-soluble phenolics and nonpolar tanshinones was performed separately and the authentication of Radix Salviae Miltiorrhizae was achieved by comparing the fingerprints of the samples with those of the reference crude drug and by comparing the Rf values of the bands in TLC fingerprints with those of reference compounds. HPLC fingerprints were obtained by simultaneous separation of phenolics and diterpenoids in Radix Salviae Miltiorrhizae. The HPLC fingerprints of seven batches of samples from different regions of China showed similar chromatographic patterns, and seven peaks were selected as characteristic peaks. The relative retention time of these characteristic peaks in the HPLC fingerprints was established as an important parameter for the identification of this herbal medicine. The pharmacologically active marker compounds salvianolic acid B, rosmarinic acid, and tanshinone IIA in herbal medicine were quantitatively determined using reverse-phase HPLC techniques. The HPLC quantitation methods of the three marker compounds were validated and the measurement uncertainty, which is important for setting the proposed content limit of the marker compounds in herbal medicine, were further evaluated. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~318.18 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1818 mL | 15.9089 mL | 31.8177 mL | |
| 5 mM | 0.6364 mL | 3.1818 mL | 6.3635 mL | |
| 10 mM | 0.3182 mL | 1.5909 mL | 3.1818 mL |