Physicochemical Properties
| Molecular Formula | C11H11N3O3 |
| Molecular Weight | 233.23 |
| Exact Mass | 233.08 |
| CAS # | 867276-98-0 |
| PubChem CID | 3323368 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 422.1±55.0 °C at 760 mmHg |
| Flash Point | 209.1±31.5 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.623 |
| LogP | -0.64 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 17 |
| Complexity | 294 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | KFRQROSRKSVROW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H11N3O3/c15-11(14-3-5-16-6-4-14)8-1-2-9-10(7-8)13-17-12-9/h1-2,7H,3-6H2 |
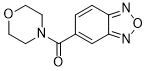
| Chemical Name | 2,1,3-benzoxadiazol-5-yl(morpholin-4-yl)methanone |
| Synonyms | Ampakine CX-717 Ampakine CX717 Ampakine CX 717 CX 717 CX717 CX-717 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Positive allosteric modulator of AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptors [1] |
| ln Vivo |
Intracortical infusion of CX 717 (CX717; 20 mg/kg) increases the efflux of norepinephrine, dopamine, and serotonin, but not glutamate. CX 717 also produces a rapid (up to 1 hour) increase in brain-derived neurotrophic factor (BDNF) and a more sustained (up to 6 hours) increase in p11 [2]. CX 717 (CX717; 20 mg/kg) has fast (30 minutes) but transient (up to 24 hours) antidepressant-like effects in the forced swim test. In well-trained, normal alert rhesus monkeys (Macaca mulatta), intravenous administration of CX717 (0.8 mg/kg) significantly enhanced performance on a cognitive delayed-match-to-sample (DMS) task. Mean overall performance improved from 74.9% correct under vehicle to 90.5% correct under CX717. This enhancement was dose-dependent (tested range 0.3-1.5 mg/kg IV), with maximal effects observed at 1.5 mg/kg. CX717 also markedly reduced response latencies during the task. [1] In monkeys subjected to 30-36 hours of sleep deprivation, which severely impaired DMS performance (mean correct 62.7%), administration of CX717 (0.8 mg/kg IV) prior to testing completely restored performance to a level significantly above the normal vehicle condition (mean correct 84.5%). This restoration was accompanied by a return of task-related response latencies to shorter durations. [1] Positron emission tomography (PET) imaging revealed that during the DMS task under normal alert conditions, CX717 administration increased cerebral metabolic rates for glucose (CMRglc) in the prefrontal cortex, dorsal striatum, and medial temporal lobe (including hippocampus) compared to the vehicle condition. Sleep deprivation altered CMRglc patterns in these regions (decreases in prefrontal cortex and dorsal striatum, increases in medial temporal lobe and precuneus). CX717 administration to sleep-deprived animals reversed these altered metabolic patterns, returning CMRglc in most affected brain regions (except the thalamus) towards the pattern seen under normal alert conditions. [1] Electroencephalogram (EEG) recordings in sleep-deprived monkeys showed increased power in delta (0.5-5 Hz) and beta (18-25 Hz) frequency bands, suggestive of microsleep episodes. Administration of CX717 reduced the power in these frequency bands, shifting the EEG profile towards that observed in the normal alert condition. [1] |
| Animal Protocol |
Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rats, body weight 280-350 g[2] Doses: 20 mg/kg (administered as 1 mL/kg) Route of Administration: intraperitoneal (ip) injection Experimental Results: Induced antidepressant-like effects (20 mg/kg, ip) does not alter extracellular concentrations of norepinephrine (NA), dopamine (DA), serotonin (5-HT), and glutamate in the medial prefrontal cortex (mPFC). Eleven adult male rhesus monkeys (8.0-11.0 kg) were used. They were individually housed under controlled conditions with a 12-hour light/dark cycle. [1] For behavioral testing, animals performed a visual delayed-match-to-sample (DMS) task daily while seated in a primate chair. They completed 150-300 trials per session. Correct responses were rewarded with diluted fruit juice. Most of their daily fluid requirement was contingent upon task performance to maintain motivation. [1] For sleep deprivation, animals were kept awake for 30-36 hours continuously in a separate, lit room. Sleep prevention methods included videos, music, occasional treats, gentle cage shaking, and interaction with technicians. [1] CX717 was dissolved in 10% w/v hydroxypropyl-beta-cyclodextrin and 0.45% saline vehicle. It was administered intravenously via a chronically implanted vascular access port at doses ranging from 0.3 to 1.5 mg/kg (with 0.8 mg/kg being the primary dose reported for key experiments), 10 minutes prior to the start of behavioral testing or PET scan procedures. Vehicle injections were administered prior to non-drug control sessions. [1] For PET scans measuring cerebral glucose metabolism (CMRglc), animals performed the DMS task for ten trials before receiving an IV injection of [18F]-FDG. They continued performing the task for 40 minutes post-injection during tracer uptake. Subsequently, they were anesthetized and scanned. Blood samples were collected at specified times for tracer concentration measurement. Image analysis was performed using Statistical Parametric Mapping (SPM99) software. [1] |
| References |
[1]. Facilitation of task performance and removal of the effects of sleep deprivation by an ampakine (CX717) in nonhuman primates. PLoS Biol. 2005 Sep;3(9):e299. [2]. Antidepressant-Like Effects of CX717, a Positive Allosteric Modulator of AMPA Receptors. Mol Neurobiol. 2020 Aug;57(8):3498-3507. |
| Additional Infomation |
CX-717 is an ampakine compound previously investigated for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) and Alzheimer's disease. Drug Indication Investigated for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD) and Alzheimer's disease. Mechanism of Action CX-717 is an ampakine compound. It is a positive allosteric modulator of AMPA receptors. Its action is theorized to be due to the facilitation of transmission at cortical synapses that use glutamate as a neurotransmitter. This in turn may promote plasticity at the synapse, which could translate into better cognitive performance. CX-717 works by allosterically binding to particular receptors in the brain, called AMPA-type glutamate receptors. This boosts the activity of glutamate, a neurotransmitter, and makes it easier to encode memory and to learn. In addition, CX717 could potentially strongly impact the up-regulation of BDNF (brain-derived neurotrophic factor) or NGF (nerve growth factor), two growth factors known to stimulate the formation of new circuitry in the brain associated with forming memory and cognition. CX717 is an ampakine, a class of compounds acting as positive allosteric modulators of AMPA-type glutamate receptors. It is proposed to enhance cognition by modulating glutamatergic synaptic transmission. [1] This study demonstrates that CX717 not only enhances cognitive performance (short-term/working memory) under normal alert conditions in nonhuman primates but also completely reverses the severe cognitive impairment induced by 30-36 hours of sleep deprivation, even restoring performance to above-normal levels. [1] The cognitive enhancement and sleep deprivation reversal effects are correlated with modulation of brain activity (CMRglc) in key regions associated with memory and executive function, such as the prefrontal cortex, dorsal striatum, and medial temporal lobe/hippocampus. [1] The study suggests ampakines like CX717 have potential for sustaining cognitive performance under adverse conditions like sleep loss, with a mechanism distinct from traditional psychostimulants. [1] The research was supported by the Defense Advanced Research Projects Agency (DARPA) and the National Institutes of Health (NIH). Cortex Pharmaceuticals provided the drug CX717. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~214.39 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.72 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.72 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (10.72 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2876 mL | 21.4381 mL | 42.8761 mL | |
| 5 mM | 0.8575 mL | 4.2876 mL | 8.5752 mL | |
| 10 mM | 0.4288 mL | 2.1438 mL | 4.2876 mL |