SYM2206 is a potent, specific and non-competitive antagonist of AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptor) receptors with IC50 value of 2.8 μM. AMPA receptor SYM 2206 exerts anticonvulsant effects, elevates the threshold for maximal electroshock-induced seizures in mice. TID20 and TID50(threshold increasing doses by 20% and 50%) values for SYM 2206 are 4.25 and 10.56 mg/kg in the MEST test in mice, respectively. In addition, it has been reported that inhibitors of glutamate receptors such as GYKI52466 and SYM2206 significantly decreased survival of pancreatic cancer cells, suggesting the presence of glutamate signaling in pancreatic cancer.
Physicochemical Properties
| Molecular Formula | C20H22N4O3 | |
| Molecular Weight | 366.41 | |
| Exact Mass | 366.169 | |
| CAS # | 173952-44-8 | |
| Related CAS # |
|
|
| PubChem CID | 5039877 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.37g/cm3 | |
| Index of Refraction | 1.673 | |
| LogP | 3.591 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 27 | |
| Complexity | 576 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | OFUDZKKOKPGXOH-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C20H22N4O3/c1-3-8-22-20(25)24-12(2)15-9-17-18(27-11-26-17)10-16(15)19(23-24)13-4-6-14(21)7-5-13/h4-7,9-10,12H,3,8,11,21H2,1-2H3,(H,22,25) | |
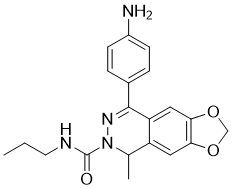
| Chemical Name | 8-(4-aminophenyl)-5-methyl-N-propyl-5H-[1,3]dioxolo[4,5-g]phthalazine-6-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
|
||
| ln Vivo |
|
||
| Animal Protocol |
|
||
| References | Front Cell Neurosci.2018 Oct 11;12:361;Neoplasia.2010 Aug;12(8):659-67. | ||
| Additional Infomation | 8-(4-aminophenyl)-5-methyl-N-propyl-5H-[1,3]dioxolo[4,5-g]phthalazine-6-carboxamide is a member of phthalazines. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (7.51 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.75 mg/mL (7.51 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7292 mL | 13.6459 mL | 27.2918 mL | |
| 5 mM | 0.5458 mL | 2.7292 mL | 5.4584 mL | |
| 10 mM | 0.2729 mL | 1.3646 mL | 2.7292 mL |