SW044248 is a novel, potent and selective Topoisomerase I inhibitor that was identified through a screen for chemicals selectively toxic for non-small cell lung cancer (NSCLC) cell lines. It was discovered that in sensitive cells, but not in insensitive cells, SW044248 quickly inhibits macromolecular synthesis. A panel of 74 NSCLC cell lines was treated with SW044248, which was nontoxic to immortalized human bronchial cell lines and killed about 15% of them. SW044248 inhibited topoisomerase 1 (Top1) but not topoisomerase 2, and in sensitive HCC4017 cells, the acute transcriptional response to the drug correlated significantly with topoisomerase inhibitors. Camptothecin did not exhibit the same selective toxicity as SW044248, and SW044248 inhibited Top1 in a different way than camptothecin. Although deleting Top1 was eventually toxic, it did provide some protection against SW044248 for cells when done so via siRNA. siRNA to CDKN1A sensitized cells resistant to SW044248, and those cells upregulated CDKN1A in response to the compound. Thus, at least part of the differential sensitivity of NSCLC cells to SW044248 is the ability to upregulate CDKN1A.
Physicochemical Properties
| Molecular Formula | C22H23N5O2S | |
| Molecular Weight | 421.52 | |
| Exact Mass | 421.157 | |
| Elemental Analysis | C, 62.69; H, 5.50; N, 16.61; O, 7.59; S, 7.61 | |
| CAS # | 522650-83-5 | |
| Related CAS # |
|
|
| PubChem CID | 3152990 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.675 | |
| LogP | 4.48 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 30 | |
| Complexity | 582 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | S(C1N=NC2=C(N=1)N(C([H])([H])C([H])([H])[H])C1=C([H])C([H])=C([H])C([H])=C12)C([H])(C(N([H])C1=C([H])C([H])=C([H])C([H])=C1OC([H])([H])[H])=O)C([H])([H])C([H])([H])[H] |
|
| InChi Key | PEVRGVRHMMZNGI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C22H23N5O2S/c1-4-18(21(28)23-15-11-7-9-13-17(15)29-3)30-22-24-20-19(25-26-22)14-10-6-8-12-16(14)27(20)5-2/h6-13,18H,4-5H2,1-3H3,(H,23,28) | |
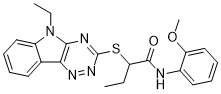
| Chemical Name | 2-[(5-ethyl-[1,2,4]triazino[5,6-b]indol-3-yl)sulfanyl]-N-(2-methoxyphenyl)butanamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Topoisomerase I | ||
| ln Vitro |
|
||
| ln Vivo |
|
||
| Cell Assay | In wells of 96-well plates, 100 μL of 50,000 cells/mL cell suspensions of distinct cell lines are added. The following day, 100 μL of cell medium was added to each well, substituting 2X concentration of either SW044248, camptothecin, or DMSO in triplicate. The wells' ATP concentration is determined using CelTiter-Glo after 96 and 120 hours. With the aid of a plate reader, luminescence is measured[1]. | ||
| Animal Protocol |
|
||
| References |
[1]. A Novel Inhibitor of Topoisomerase I Is Selectively Toxic for a Subset of Non-Small Cell Lung Cancer Cell Lines. Mol Cancer Ther. 2016 Jan;15(1):23-36. [2]. Systematic identification of molecular subtype-selective vulnerabilities in non-small-cell lung cancer. Cell. 2013 Oct 24;155(3):552-66. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (2.97 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3724 mL | 11.8618 mL | 23.7237 mL | |
| 5 mM | 0.4745 mL | 2.3724 mL | 4.7447 mL | |
| 10 mM | 0.2372 mL | 1.1862 mL | 2.3724 mL |