SUN11602 (SUN-11602), a novel synthetic aniline compound with basic fibroblast growth factor-like activity, can mimic the neuroprotective mechanisms of basic fibroblast growth factor. Neuroprotective properties and cell proliferation are two of the benefits of basic fibroblast growth factor (FGF-2/bFGF). Without stimulating cell division, SUN11602 demonstrated neuroprotective properties akin to those of FGF-2. Stimulation with FGF-2 or SUN11602 increased the expression of the calbindin D-28k (CalB) gene in primary cultures of hippocampal neurons and inhibited the death of neurons caused by glutamate. PD166866, a tyrosine kinase-specific inhibitor of FGF receptor 1 [FGFR1], was administered prior to treatment to eliminate these effects. It was revealed that SUN11602-mediated neuroprotection was associated with both elevated CalB expression and FGFR1 activation. FGFR1-bound (125)I-labeled FGF-2 was not affected by SUN11602, in contrast to FGF-2, according to receptor-binding experiments. In in vivo experiments, ibotenate (an NMDA receptor agonist) and aggregated Aβ1-40 were injected into wild-type (WT) and CalB-deficient (CalB(-/-)) mice to cause significant hippocampal tissue damage. Hippocampal damage was avoided in WT mice by treatment with SUN11602 (orally) or FGF-2 (intraparenchymally) at the midpoint of Aβ1-40 and ibotenate injections; in CalB(-/-) mice, however, this effect was eliminated. Therefore, by activating FGFR1 and upregulating CalB expression, SUN11602 had a protective effect on hippocampal neurons. Furthermore, the different biological functions of FGF-2 were necessary for SUN11602 to have its neuroprotective effects.
Physicochemical Properties
| Molecular Formula | C26H37N5O2 | |
| Molecular Weight | 451.60 | |
| Exact Mass | 451.29 | |
| Elemental Analysis | C, 69.15; H, 8.26; N, 15.51; O, 7.09 | |
| CAS # | 704869-38-5 | |
| Related CAS # |
|
|
| PubChem CID | 22245705 | |
| Appearance | white solid powder | |
| LogP | 2.834 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 33 | |
| Complexity | 641 | |
| Defined Atom Stereocenter Count | 0 | |
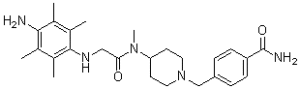
| SMILES | O=C(N)C1=CC=C(CN2CCC(N(C(CNC3=C(C)C(C)=C(N)C(C)=C3C)=O)C)CC2)C=C1 |
|
| InChi Key | KCODNOOPOPTZMO-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C26H37N5O2/c1-16-18(3)25(19(4)17(2)24(16)27)29-14-23(32)30(5)22-10-12-31(13-11-22)15-20-6-8-21(9-7-20)26(28)33/h6-9,22,29H,10-15,27H2,1-5H3,(H2,28,33) | |
| Chemical Name | 4-[[4-[[2-(4-amino-2,3,5,6-tetramethylanilino)acetyl]-methylamino]piperidin-1-yl]methyl]benzamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
bFGF SUN11602 protects rat cerebrocortical neurons in primary cultures against glutamate-induced neuronal death. Cerebrocortical neurons express the CALB1 gene at higher levels when exposed to SUN11602[1]. By activating FGFR1 and upregulating CalB expression, SUN11602 protects hippocampal neurons[2]. Primarily in cultured rat hippocampal neurons, SUN11602 stimulates neurite outgrowth[3]. |
| ln Vitro |
SUN11602 protects rat cerebrocortical neurons in primary cultures against glutamate-induced neuronal death. Cerebrocortical neurons express the CALB1 gene at higher levels when exposed to SUN11602[1]. By activating FGFR1 and upregulating CalB expression, SUN11602 protects hippocampal neurons[2]. Primarily in cultured rat hippocampal neurons, SUN11602 stimulates neurite outgrowth[3]. In primary cultures of rat cerebrocortical neurons, pretreatment with SUN11602 (0.1, 0.3, 1 μM) for 24 hours significantly reduced neuronal death induced by 150 μM glutamate in a concentration-dependent manner, as measured by MTT assay. Its efficacy was comparable to bFGF (5, 10 ng/mL). [1] The neuroprotective effect of SUN11602 (1 μM) was completely abolished by co-treatment with the FGFR-1 tyrosine kinase inhibitor PD166866 (0.3 μM) or the MEK inhibitor PD98059 (0.3, 1, 3 μM), indicating dependence on the FGFR-1-MEK/ERK pathway. [1] Treatment with SUN11602 (3 μM) or bFGF (10 ng/mL) for 48 hours increased the protein levels of calbindin-D28k (Calb) in primary cerebrocortical neurons from wild-type (WT) mice, as shown by immunocytochemistry. No Calb was detected in neurons from Calb⁻/⁻ mice. [1] In primary cerebrocortical neurons from WT mice, pretreatment with SUN11602 or bFGF for 24 hours suppressed the glutamate-induced (100 μM, 5 min) rise in intracellular Ca²⁺ levels, as measured by fluo 3-AM imaging. This suppressive effect was absent in neurons from Calb⁻/⁻ mice. [1] Western blot analysis showed that treatment of rat cerebrocortical neurons with SUN11602 (10, 100 μM) or bFGF (10 ng/mL) for 20 minutes induced phosphorylation of ERK1/2 without altering total ERK1/2 protein levels. [1] The neuroprotective effects of SUN11602 and bFGF were abolished by pretreatment with the transcription inhibitor actinomycin D (1 μg/mL) or the translation inhibitor cycloheximide (1 μg/mL), indicating a requirement for de novo protein synthesis. [1] In primary cultures from Calb⁻/⁻ mice, the neuroprotective effects of SUN11602 and bFGF against 50 μM glutamate were absent or greatly diminished. However, higher concentrations of SUN11602 (1, 3 μM) showed reduced but still detectable neuroprotection in Calb⁻/⁻ neurons, suggesting possible Calb-independent mechanisms. [1] SUN11602 did not competitively inhibit the binding of ¹²⁵I-labeled bFGF to the extracellular domain of FGFR in binding assays using hippocampus neurons and BHK-21 cells. [1] |
| ln Vivo | SUN11602 suppresses the glutamate-induced increase in intracellular Ca2+ in WT mice while increasing the amounts of freshly synthesized Calb in cerebrocortical neurons. Calb's capacity to capture Ca2+ enables neurons to withstand extremely harmful glutamate conditions[1]. Injections of ibotenate and oral administration of SUN11602 at the midpoint of Aβ1-40 both reduce deficits in spatial learning in the water maze task and short-term memory impairment in the Y-maze test. Furthermore, the administration of SUN11602 prevents the growth of PTBBS, or peripheral-type benzodiazepine-binding sites, a sign of gliosis[3]. |
| Enzyme Assay | SUN11602 is a novel aniline compound with basic fibroblast growth factor-like activity. |
| Cell Assay |
For twenty-four hours before glutamate toxicity sets in, cerebral cortical neurons are pretreated with vehicle (Hanks' Balanced Salt Solution), SUN11602, bFGF, or the other growth factors. Then, 200 μL of culture medium is added to each well (of the microplates) along with 10 μL of the MTT solution (5 mg/mL). Following a 24-hour drying period, 200 μL of DMSO is added to each well to completely dissolve the reaction products in preparation for the MTT assay[1]. Neuroprotection/Cell Viability (MTT) Assay: Primary cerebrocortical neurons from rat embryos (E18) or mouse embryos (E15-16, WT or Calb⁻/⁻) were cultured in neurobasal medium with supplements on poly-D-lysine or polyethyleneimine-coated plates. Non-neuronal cell proliferation was inhibited with AraC (1 μM). After 7-10 days in vitro, neurons were pretreated with vehicle, SUN11602, bFGF, or other growth factors for 24 hours. Cultures were then exposed to glutamate (75, 100, or 150 μM) for another 24 hours. Cell viability was assessed by adding MTT solution (5 mg/mL) for 15 minutes. The reaction was stopped, cells were dried, and the formed formazan was dissolved in DMSO. Absorbance was measured at 570 nm with a reference at 650 nm. [1] Intracellular Calcium Imaging: Primary cerebrocortical neurons from WT or Calb⁻/⁻ mice were cultured on glass dishes and pretreated with vehicle, SUN11602, or bFGF for 24 hours. Neurons were loaded with the Ca²⁺ indicator fluo 3-AM (4 μM) in HEPES-buffered saline containing DMSO and a dispersing agent for 60 min at 37°C. After washing, cytoplasmic free Ca²⁺ levels were measured before and after a 5-minute exposure to glutamate (100 μM for WT, 50 μM for Calb⁻/⁻) using laser confocal microscopy. [1] Western Blot for ERK1/2 Phosphorylation: Primary rat cerebrocortical neurons grown in plates were treated with SUN11602 (10, 100 μM) or bFGF (10 ng/mL) for 20 minutes and then lysed. Proteins (187.5 μg per lane) were separated by SDS-PAGE (12% gel) and transferred to a nitrocellulose membrane. Membranes were blocked and probed with primary antibodies against phosphorylated ERK1/2 (p-ERK1/2) and total ERK1/2, followed by HRP-conjugated secondary antibodies. Signals were developed using a peroxidase substrate. [1] Immunocytochemistry: Primary mouse cerebrocortical neurons cultured on glass dishes or mouse brain tissue sections were fixed. After blocking, samples were incubated with primary antibodies against Calb or FGFR1 overnight at 4°C. For cultured neurons, signals were visualized using a biotinylated secondary antibody, an avidin-biotin-peroxidase complex, and DAB as a chromogen. For fluorescence staining, an FITC-conjugated secondary antibody was used. Brain tissue sections from perfused mice were processed similarly for Calb immunostaining. [1] |
| Animal Protocol |
Rats: SUN11602 (0.1,1,and10mg/kg) is given orally to the rat hippocampal-lesion model once, 24 hours following the injection of Aβ1–40. Saline is given to the vehicle-treated groups in place of SUN11602[3]. Primary Neuron Culture Preparation: Cerebrocortical neurons were prepared from embryonic brains of Wistar rats (E18) or from embryonic brains of Calb⁻/⁻ and wild-type (WT) mice (E15–16). Cerebral cortices were dissociated with papain, and cells were suspended in neurobasal medium with supplements. Cells were seeded onto coated plates or dishes and maintained in a humidified incubator (10% CO₂, 37°C). AraC (1 μM) was added at day 4 in vitro to inhibit non-neuronal cell growth. Experiments were performed on 7- to 10-day-old cultures. [1] |
| References |
[1]. SUN11602, a novel aniline compound, mimics the neuroprotective mechanisms of basic fibroblast growth factor. ACS Chem Neurosci. 2013 Feb 20;4(2):266-76. [2]. SUN11602-induced hyperexpression of calbindin D-28k is pivotal for the survival of hippocampal neurons under neurotoxic conditions. Brain Res. 2015 Jan 12;1594:71-81. [3]. SUN11602 has basic fibroblast growth factor-like activity and attenuates neuronal damage and cognitive deficits in a rat model of Alzheimer's disease induced by amyloid β and excitatory amino acids. Brain Res. 2014 Oct 17;1585:159-66. |
| Additional Infomation |
SUN11602 (4-({4-[(4-amino-2,3,5,6-tetramethylanilino)acetyl](methyl)amino}-1-piperidinyl)methyl)benzamide) is a novel synthetic small molecule designed to mimic the neuroprotective effects of basic fibroblast growth factor (bFGF). [1] Its proposed neuroprotective mechanism involves activation of the FGFR-1 tyrosine kinase domain (independently of the bFGF extracellular binding site), leading to phosphorylation of ERK1/2 via the MEK/ERK pathway, subsequent upregulation of the calcium-binding protein calbindin-D28k (Calb) via new protein synthesis, and stabilization of intracellular Ca²⁺ homeostasis, which protects neurons from glutamate-induced excitotoxicity. [1] The study suggests that SUN11602 may represent a promising therapeutic candidate for neurological diseases involving excitotoxic injury, potentially overcoming limitations of bFGF such as poor blood-brain barrier permeability and short half-life. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.54 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.54 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2143 mL | 11.0717 mL | 22.1435 mL | |
| 5 mM | 0.4429 mL | 2.2143 mL | 4.4287 mL | |
| 10 mM | 0.2214 mL | 1.1072 mL | 2.2143 mL |