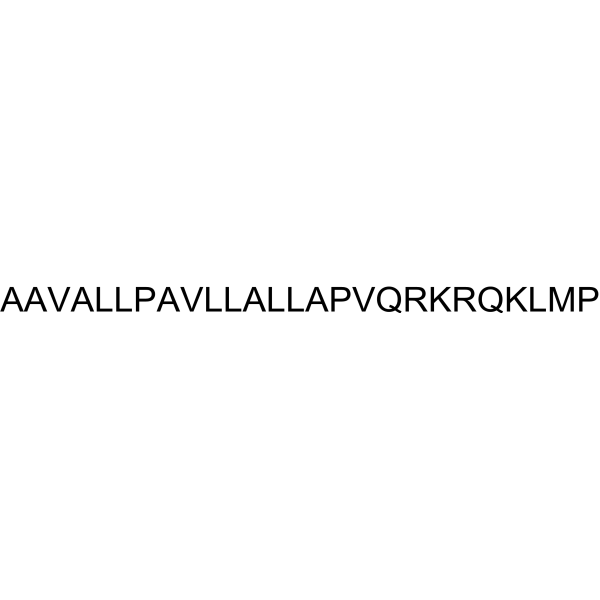Physicochemical Properties
| Molecular Formula | C129H230N36O29S |
| Molecular Weight | 2781.49532842636 |
| Exact Mass | 2779.74 |
| CAS # | 213546-53-3 |
| Related CAS # | 201608-17-5 |
| PubChem CID | 16209942 |
| Sequence | Ala-Ala-Val-Ala-Leu-Leu-Pro-Ala-Val-Leu-Leu-Ala-Leu-Leu-Ala-Pro-Val-Gln-Arg-Lys-Arg-Gln-Lys-Leu-Met-Pro; H-Ala-Ala-Val-Ala-Leu-Leu-Pro-Ala-Val-Leu-Leu-Ala-Leu-Leu-Ala-Pro-Val-Gln-Arg-Lys-Arg-Gln-Lys-Leu-Met-Pro-OH; L-alanyl-L-alanyl-L-valyl-L-alanyl-L-leucyl-L-leucyl-L-prolyl-L-alanyl-L-valyl-L-leucyl-L-leucyl-L-alanyl-L-leucyl-L-leucyl-L-alanyl-L-prolyl-L-valyl-L-glutaminyl-L-arginyl-L-lysyl-L-arginyl-L-glutaminyl-L-lysyl-L-leucyl-L-methionyl-L-proline |
| SequenceShortening | AAVALLPAVLLALLAPVQRKRQKLMP; H-AAVALLPAVLLALLAPVQRKRQKLMP-OH |
| Appearance | White to off-white solid powder |
| LogP | 9.812 |
| Hydrogen Bond Donor Count | 34 |
| Hydrogen Bond Acceptor Count | 35 |
| Rotatable Bond Count | 92 |
| Heavy Atom Count | 195 |
| Complexity | 6190 |
| Defined Atom Stereocenter Count | 26 |
| SMILES | C[C@@H](C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(=N)N)C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N3CCC[C@H]3C(=O)O)N |
| InChi Key | FAWLNURBQMTKEB-URDPEVQOSA-N |
| InChi Code | InChI=1S/C129H230N36O29S/c1-64(2)57-87(156-117(183)92(62-69(11)12)158-123(189)100(72(17)18)161-106(172)78(25)143-119(185)94-41-35-54-164(94)126(192)93(63-70(13)14)159-118(184)90(60-67(7)8)154-104(170)76(23)144-121(187)99(71(15)16)160-105(171)77(24)141-102(168)74(21)132)113(179)142-75(22)103(169)153-89(59-66(5)6)116(182)157-88(58-65(3)4)114(180)145-79(26)124(190)163-53-34-42-95(163)120(186)162-101(73(19)20)122(188)151-85(45-47-98(134)167)112(178)149-82(39-32-51-139-128(135)136)108(174)146-80(37-28-30-49-130)107(173)148-83(40-33-52-140-129(137)138)109(175)150-84(44-46-97(133)166)111(177)147-81(38-29-31-50-131)110(176)155-91(61-68(9)10)115(181)152-86(48-56-195-27)125(191)165-55-36-43-96(165)127(193)194/h64-96,99-101H,28-63,130-132H2,1-27H3,(H2,133,166)(H2,134,167)(H,141,168)(H,142,179)(H,143,185)(H,144,187)(H,145,180)(H,146,174)(H,147,177)(H,148,173)(H,149,178)(H,150,175)(H,151,188)(H,152,181)(H,153,169)(H,154,170)(H,155,176)(H,156,183)(H,157,182)(H,158,189)(H,159,184)(H,160,171)(H,161,172)(H,162,186)(H,193,194)(H4,135,136,139)(H4,137,138,140)/t74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,94-,95-,96-,99-,100-,101-/m0/s1 |
| Chemical Name | (2S)-1-[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-1-[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-1-[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-aminopropanoyl]amino]propanoyl]amino]-3-methylbutanoyl]amino]propanoyl]amino]-4-methylpentanoyl]amino]-4-methylpentanoyl]pyrrolidine-2-carbonyl]amino]propanoyl]amino]-3-methylbutanoyl]amino]-4-methylpentanoyl]amino]-4-methylpentanoyl]amino]propanoyl]amino]-4-methylpentanoyl]amino]-4-methylpentanoyl]amino]propanoyl]pyrrolidine-2-carbonyl]amino]-3-methylbutanoyl]amino]-5-oxopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-oxopentanoyl]amino]hexanoyl]amino]-4-methylpentanoyl]amino]-4-methylsulfanylbutanoyl]pyrrolidine-2-carboxylic acid |
| Synonyms | SN50; 213546-53-3; NFkappaB Inhibitor; NF-kappaB Inhibitor, SN50; H-ALA-ALA-VAL-ALA-LEU-LEU-PRO-ALA-VAL-LEU-LEU-ALA-LEU-LEU-ALA-PRO-VAL-GLN-ARG-LYS-ARG-GLN-LYS-LEU-MET-PRO-OH; NF-kappaB SN50; SN50 trifluoroacetate salt; AAVALLPAVLLALLAPVQRKRQKLMP; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | NF-κB |
| ln Vitro |
When compared to groups treated with a vehicle, pretreatment with SN50 causes a significant decrease in the number of PI-positive cells at 12, 24, and 48 hours after a TBI[1]. Topical SN50 lowers the frequency of epithelial defects and ulceration in healing corneas via suppressing nuclear factor-κB activation in local cells. In treated corneas compared to controls, there is a decrease in myofibroblast production, macrophage infiltration, matrix metalloproteinase activity, basement membrane breakdown, and cytokine expression[2]. The effects of LY294002 in inducing cell death after 24 hours may be markedly enhanced by treating the human gastric cancer cells SGC7901 with SN50[3]. LPS-induced lung injury is linked to the translocation of NF-kB and the generation of inflammatory cytokines, both of which SN50 can inhibit[4]. In this study,it was found that treating the human gastric cancer cells SGC7901 with SN50 could significantly enhance the effects of LY294002 on inducing cell death after 24 h, compared to the control group (p < 0.05). Detection of mitochondrial potential and transmission electron microscopic examination indicated that the rate of cell death increased progressively. The expression of p53, PUMA and Beclin1 was up-regulated. Conclusions: The NF-κB inhibitor SN50 could enhance the role of LY294002 on inducing cell death of human gastric cancer cells SGC7901, which might be a promising new approach to gastric cancer therapy.[3] |
| ln Vivo | From the first to the fourth day, SN50 treatment expedites the recovery of motor functional result. At 7 and 8 days after TBI, animals who received SN50 pretreatment show a significant reduction in their visuospatial learning latencies compared to the control group. When SN50 is pretreated, the levels of TNF-a and NF-κB p65 protein are significantly reduced from 12 to 48 hours after TBI and 6 to 48 hours after TBI, respectively[1]. |
| Enzyme Assay |
Detection of mitochondrial potential (ΔΨ)[3] Mitochondrial ΔΨ was determined using the KeyGEN Mitochondrial Membrane Sensor Kit. The Mitosensor dye aggregates in the mitochondria of healthy cells and emits red fluorescence against a green monomeric cytoplasmic background staining. However, in cells with a collapsed mitochondrial ΔΨ, the dye cannot accumulate in the mitochondria and remains in monomeric form throughout the cells with green fluorescence. Briefly, SGC7901 cells were incubated with LY294002, SN50 and LY294002 + SN50 in 6-well plates for the indicated times and then pelleted, washed with PBS, and resuspended in 0.5 ml of diluted Mitosensor reagent (1 µmol/ml in incubation buffer). After the cells were incubated with the Mitosensor reagent for 20 min, 0.2 ml of incubation buffer was added and cells were centrifuged then resuspended in 40 µl of incubation buffer. Finally, the cells were washed and resuspended in 1 ml PBS for flow cytometry analysis. Real-time quantitative RT-PCR analysis of p53, Beclin1 and PUMA[3] Total RNA was extracted using the RNAiso kit. For extracting total mRNA, SGC7901 cells were treated with LY294002 (50 µmol/l), SN50 (18 µmol/l) and LY294002+SN50 for 6 h before being harvested. First-strand cDNA was generated via reverse transcription of 2 µg of total RNA using random primers and the Primescript RT Reagent Kit in a total reaction volume of 20 µl according to the manufacturer's instructions. The sequences of the forward and reverse oligonucleotide primers, specific to the chosen candidate and housekeeping genes, were designed with Primer5 software (available from frodo.wi.mit.edu/cgi-bin/primer5/primer5_www.cgi). For p53 the primers were: forward, 5’-ACTAAGCG-AGCACTGCCCAAC-3’; reverse, 5’-CCTCATTCA- GCTCTCGGAACATC-3’. For PUMA the primers were: forward, 5’-CGACCTCAACGCACAGTACGA-3’; reverse, 5’-GGCACCTAATTGGGCTCCATC-3’. For Beclin1 the primers were: forward, 5’- CCAGATGC-GTTATGCCCAGAC-3’; reverse, 5’-CATTCCATTC-CACGGGAACAC-3’. For β-actin the primers were: forward, 5’-ATTGCCGACAGGATGCAGA-3’; reverse, 5’-GAGTACTTGCGCTCAGGAGGA-3’. Real-time quantitative RT-PCR was performed using the iCycler 5 thermal cycler. An 80-fold dilution of each cDNA was amplified in a 20 µl volume, using the SYBR Premix EX Taq kit, with a 500 nM final concentration of each primer. The amplification specificity was checked using melting curve analysis. Threshold cycle Ct, which correlates inversely with the target mRNA level, was calculated using the second derivative maximum algorithm provided by the Light-Cycler software. For each cDNA, all target gene mRNA levels were normalized to β-actin mRNA levels. Results are expressed as the ratio of normalized target gene mRNA levels in treated cells relative to those in untreated cells. Transmission electron microscopic examination[3] Pursuant to treatment with LY294002, SN50 or LY294002 + SN50, cells were fixed in ice-cold 2.5% glutaraldehyde in 0.1 M PBS and preserved at 4°C for further processing. When processing resumed, cells were post-fixed in 1% osmium tetroxide in the same buffer, dehydrated in graded alcohols, embedded in Epon 812, sectioned with an ultra-microtome, stained with uranyl acetate and lead citrate followed by examination with a transmission electron microscope. |
| Cell Assay |
Cell viability assay[3] Cell viability was assessed with MTT assay. To determine the effects of SN50 on enhancing the role of LY294002 on SGC7901 cells, cells were plated into 96-well microplates (7 × 103 cells/well) and cultured for 24 h. Then LY294002 (50 µmol/l), SN50 (18 µmol/l) and LY294002+SN50 were added to the culture medium and cell viability was assessed with MTT 24 h after drug treatment. Later MTT solution was added to the culture medium (5 mg/ml final concentration) 4 h before the end of treatment and the reaction was stopped by addition of 10% acidified SDS 100 µl. The absorbance value (A) at 570 nm was read using an automatic multiwell spectrophotometer. The percentage of cell death was calculated as follows: cell death (%) = (1 – A of experiment well/A of positive control well) × 100%. The 3-(4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT) assay was used to determine the cytotoxic effects of the drugs. Mitochondrial membrane potential was measured using the fluorescent probe JC-1. Hoechst 33258 staining was used to detect apoptosis and necrosis morphological changes after LY294002 and/or SN50 treatment. Expression of p53, PUMA and Beclin1 were determined with real-time polymerase chain reaction (RT-PCR) analysis. We used transmission electron microscopy to identify ultrastructural changes in SGC7901 cells after LY294002 and/or SN50 treatment[3]. |
| Animal Protocol |
The present study sought to first investigate the effect of a NF-κB inhibitor SN50, which inhibits NF-κB nuclear translocation, on cell death and behavioral deficits in our mice traumatic brain injury (TBI) models. Additionally, we tried to elucidate the possible mechanisms of the therapeutic effect of SN50 through NF-κB regulating apoptotic and inflammatory pathway in vivo. Encouragingly, the results showed that pretreatment with SN50 remarkably attenuated TBI-induced cell death (detected by PI labeling), cumulative loss of cells (detected by lesion volume), and motor and cognitive dysfunction (detected by motor test and Morris water maze). To analyze the mechanism of SN50 on cell apoptotic and inflammatory signaling pathway, we thus assessed expression levels of TNF-α, cathepsin B and caspase-3, Bid cleavage and cytochrome c release in SN50-pretreated groups compared with those in saline vehicle groups. The results imply that through NF-κB/TNF-α/cathepsin networks SN50 may contribute to TBI-induced extrinsic and intrinsic apoptosis, and inflammatory pathways, which partly determined the fate of injured cells in our TBI model.[1] Researchers evaluated the therapeutic efficacy of topical administration of SN50, an inhibitor of nuclear factor-kappaB, in a corneal alkali burn model in mice. An alkali burn was produced with 1 N NaOH in the cornea of C57BL/6 mice under general anesthesia. SN50 (10 microg/microl) or vehicle was topically administered daily for up to 12 days. The eyes were processed for histological or immunohistochemical examination after bromodeoxyuridine labeling or for semi-quantification of cytokine mRNA. Topical SN50 suppressed nuclear factor-kappaB activation in local cells and reduced the incidence of epithelial defects/ulceration in healing corneas. Myofibroblast generation, macrophage invasion, activity of matrix metalloproteinases, basement membrane destruction, and expression of cytokines were all decreased in treated corneas compared with controls. To elucidate the role of tumor necrosis factor (TNF)-alpha in epithelial cell proliferation, we performed organ culture of mouse eyes with TNF-alpha, SN50, or an inhibitor of c-Jun N-terminal kinase (JNK) and examined cell proliferation in healing corneal epithelium in TNF-alpha-/- mice treated with SN50. An acceleration of epithelial cell proliferation by SN50 treatment was found to depend on TNF-alpha/JNK signaling. In conclusion, topical application of SN50 is effective in treating corneal alkali burns in mice.[2] |
| References |
[1]. Therapeutic effect of SN50, an inhibitor of nuclear factor-κB, in treatment of TBI in mice. Neurol Sci. 2013 Mar;34(3):345-55. [2]. Therapeutic effect of topical administration of SN50, an inhibitor of nuclear factor-kappaB, in treatment of corneal alkali burns in mice. Am J Pathol. 2005 May;166(5):1393-403. [3]. SN50 enhances the effects of LY294002 on cell death induction in gastric cancer cell line SGC7901. Arch Med Sci. 2013 Dec 30;9(6):990-8. [4]. Inhibitor of nuclear factor-κB, SN50, attenuates lipopolysaccharide-induced lung injury in an isolated and perfused rat lung model. Transl Res. 2014 Mar;163(3):211-20. |
| Additional Infomation |
SN50 is a 26-amino acid peptide with formula C129H230N36O29S. It is a cell permeable inhibitor of NF-kappaB translocation. It has a role as a NF-kappaB inhibitor, a cardioprotective agent and an anti-inflammatory agent. NF-κB cell permeable inhibitory peptide (SN50) inhibits translocation of nuclear factor-κB (NF-κB) and production of inflammatory cytokines that are implicated in lipopolysaccharide (LPS)-induced lung injury (LPSLI). However, the protective effect of SN50 in LPSLI is unclear. We explored the cellular and molecular mechanisms of SN50 treatment in LPSLI. LPSLI was induced by intratracheal instillation of 10 mg/kg LPS using an isolated and perfused rat lung model. SN50 was administered in the perfusate 15 minutes before LPS was administered. Hemodynamics, lung histologic change, inflammatory responses, and activation of apoptotic pathways were evaluated. After LPSLI, increased pulmonary vascular permeability and lung weight gain was observed. The levels of interleukin (IL)-1β, tumor necrosis factor (TNF)-α, myeloperoxidase, and macrophage inflammatory protein-2 increased in bronchoalveolar lavage fluids. Lung-tissue expression of TNF-α, IL-1β, mitogen-activated protein kinases (MAPKs), caspase-3, p-AKT (serine-threonine kinase, also known as protein kinase B), and plasminogen activator inhibitor-1 (PAI-1) was greater in the LPS group compared with controls. Upregulation and activation of NF-κB was associated with increased lung injury in LPSLI. SN50 attenuated the inflammatory responses, including expression of IL-1β, TNF-α, myeloperoxidase, MAPKs, PAI-1, and NF-κB; downregulation of apoptosis indicated by caspase-3 and p-AKT expression was also observed. In addition, SN50 mitigated the increase in the lung weight, pulmonary vascular permeability, and lung injury. In conclusion, LPSLI is associated with inflammatory responses, apoptosis, and coagulation. NF-κB is an important therapeutic target in the treatment of LPSLI. SN50 inhibits translocation of NF-κB and attenuates LPSLI.[4] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~35.95 mM) H2O : ~50 mg/mL (~17.98 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 33.33 mg/mL (11.98 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.3595 mL | 1.7976 mL | 3.5952 mL | |
| 5 mM | 0.0719 mL | 0.3595 mL | 0.7190 mL | |
| 10 mM | 0.0360 mL | 0.1798 mL | 0.3595 mL |