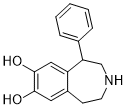
SKF38393 (also known as SKF-38393A) is a novel potent and selective dopamine D1 receptor agonist with IC50 of 110 nM, the (+)-enantiomer is the active isomer. Agonism of the D1DR by SKF 38393 is correlated to induced desynchronization of electroencephalographic activity in rats. SKF 38393 also demonstrated agonism of the serotonin SR-2C (5-HT1C receptor). SKF 38393 HCl is an activator of D5DR. In locally anesthetized, artificially respired, gallamine-treated rats, i.v. administration of SKF 38393 significantly altered dopamine cell activity. In these rats, firing rate increases and decreases were also observed.
Physicochemical Properties
| Molecular Formula | C16H17NO2 | |
| Molecular Weight | 255.32 | |
| Exact Mass | 255.126 | |
| Elemental Analysis | C, 75.27; H, 6.71; N, 5.49; O, 12.53 | |
| CAS # | 67287-49-4 | |
| Related CAS # | 67287-49-4;62717-42-4 (HCl);81702-42-3 (R-isomer HCl);62751-59-1 (R-isomer); 20012-10-6 (HBr); | |
| PubChem CID | 1242 | |
| Appearance | Typically exists as solid at room temperature | |
| Density | 1.209g/cm3 | |
| Boiling Point | 467.1ºC at 760mmHg | |
| Flash Point | 180.1ºC | |
| LogP | 2.704 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 1 | |
| Heavy Atom Count | 19 | |
| Complexity | 291 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | C1=CC=C(C=C1)C2CNCCC3=CC(=C(C=C32)O)O |
|
| InChi Key | JUDKOGFHZYMDMF-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H17NO2/c18-15-8-12-6-7-17-10-14(13(12)9-16(15)19)11-4-2-1-3-5-11/h1-5,8-9,14,17-19H,6-7,10H2 | |
| Chemical Name | 5-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine-7,8-diol | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Dopamine D1/D5 receptor | ||
| ln Vitro |
|
||
| ln Vivo |
SKF 38393(6 mg/kg, i.p.) prevents the scopolamine-induced impairment of performance of a T-maze working memory task. In adult male NMRI mice, SKF-38393 (1 μg/mouse) impaires context-dependent fear learning. Dopamine D(1)/D(5) receptor agonists may enhance cognition by mimicking dopamine's neurophysiological actions on the processes underlying learning and memory. The present study examined the task- and performance- dependence of the cognitive effects of a partial agonist at dopamine D(1)/D(5) receptors, SKF-38393 [(+/-)-1-phenyl-2,3,4,5-tetrahydro-(1H)-3-benzazepine-7,8-diol hydrobromide], in rats. Spatial working memory was assessed in a T-maze, spatial reference memory in a water maze and habituation learning in a novel environment, a hole board. The muscarinic acetylcholine receptor antagonist scopolamine (1.5 mg/kg, i.p.) was used to cause an impairment of performance of these learning tasks. Administration of SKF 38393 (6 mg/kg, i.p.) alone had no significant effect on spontaneous alternation in the T-maze, latency to escape to a hidden platform in the water maze or the habituation of spontaneous behaviour in the hole board. In contrast, in scopolamine-treated rats, whereas SKF 38393 prevented the scopolamine-induced deficit in the T-maze, it exacerbated the impairment in the water maze and did not significantly alter the disruption of habituation. These results suggest that dopamine D(1)/D(5) receptor activation has performance- and task-dependent effects on cognitive function [2]. |
||
| Enzyme Assay |
SKF 38393 hydrochloride is an agonist of D1 with IC50 of 110 nM. Iodinated SCH 23390, 125I-SCH 23982 (DuPont-NEN), was examined using quantitative autoradiography for its potency, selectivity, and anatomical and neuronal localization of binding to the dopamine D1 receptor in rat brain sections. 125I-SCH 23982 bound to D1 sites in the basal ganglia with very high affinity (Kd values of 55-125 pM), specificity (70-85% of binding was displaced by 5 microM cis-flupenthixol), and in a saturable manner (Bmax values of 65-176 fmol/mg protein). Specific 125I-SCH 23982 binding was displaced by the selective D1 antagonists SCH 23390 (IC50 = 90 pM) and cis-flupenthixol (IC50 = 200 pM) and the D1 agonist SKF 38393 (IC50 = 110 nM) but not by D2-selective ligands (I-sulpiride, LY 171555) or the S2 antagonist cinanserin. Compared with 3H-SCH 23390, the 5- to 10-fold greater affinity for the D1 site and 50-fold greater specific radioactivity of 125I-SCH 23982 makes it an excellent radioligand for labeling the D1 receptor. The concentrations of D1 sites were greatest in the medial substantia nigra and exceeded by over 50% the concentration of D1 sites in the lateral substantia nigra, caudoputamen, nucleus accumbens, olfactory tubercle, and entopeduncular nucleus. Lower concentrations of D1 sites were present in the internal capsule, dorsomedial frontal cortex, claustrum, and layer 6 of the neocortex. D1 sites were absent in the ventral tegmental area. Intrastriatal injections of the axon-sparing neurotoxin, quinolinic acid, depleted by 87% and by 46-58% the concentrations of displaceable D1 sites in the ipsilateral caudoputamen and medial and central pars reticulata of the substantia nigra, respectively. No D1 sites were lost in the lateral substantia nigra. Destruction of up to 94% of the mesostriatal dopaminergic projection with 6-hydroxydopamine did not reduce D1 binding nor, with one exception, increase striatal or nigral D1 receptor concentrations. 125I-SCH 23982 selectively labels D1 binding sites on striatonigral neurons with picomolar affinity, and these neurons contain the majority of D1 sites in rat brain[J Neurosci. 1987 Jan;7(1):213-222.]. |
||
| Animal Protocol |
|
||
| References |
[1]. Proc Natl Acad Sci U S A.1995 Mar 28;92(7):2446-50; [2]. Eur J Pharmacol.2007 Dec 22;577(1-3):71-7. |
||
| Additional Infomation |
1-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine-7,8-diol is a benzazepine that is 2,3,4,5-tetrahydro-3-benzazepine bearing a phenyl substituent at position 1 and two hydroxy substituents at positions 7 and 8. It is a benzazepine, a member of catechols and a secondary amino compound. A selective D1 dopamine receptor agonist used primarily as a research tool. Agonists of the dopamine D1/D5 receptors that are positively coupled to adenylyl cyclase specifically induce a slowly developing long-lasting potentiation of the field excitatory postsynaptic potential in the CA1 region of the hippocampus that lasts for > 6 hr. This potentiation is blocked by the specific D1/D5 receptor antagonist SCH 23390 and is occluded by the potentiation induced by cAMP agonists. An agonist of the D2 receptor, which is negatively coupled to adenylyl cyclase through G alpha i, did not induce potentiation. Although this slow D1/D5 agonist-induced potentiation is partially independent of N-methyl-D-aspartate receptors, it seems to share some steps with and is occluded by the late phase of long-term potentiation (LTP) produced by three repeated trains of nerve stimuli applied to the Schaffer collateral pathway. Similarly, the D1/D5 antagonist SCH 23390 attenuates the late phase of the LTP induced by repeated trains, and the D1/D5 agonist-induced potentiation is blocked by the protein synthesis inhibitor anisomycin. These results suggest that the D1/D5 receptor may be involved in the late, protein synthesis-dependent component of LTP in the hippocampal CA1 region, either as an ancillary component or as a mediator directly contributing to the late phase. [1] The effects of SKF-38393 were clearly performance-dependent: SKF 38393-induced changes in learning were revealed in performance-impaired scopolamine-treated rats and were absent in control rats, which showed good performance of the tasks. However poor performance was not a predictor of a facilitatory effect on learning in all tasks. This contrasts with the performance-dependent effects of full dopamine D1/D5 receptor agonists in normal rats learning a variety of tasks, where poor performance at long delays was improved and good performance, at short delays, was impaired (Floresco et al., 2001, Chudasama and Robbins, 2004, Hotte et al., 2005). Our findings indicate that poor performance on a task seems insufficient on its own to reveal facilitatory effects of dopamine D1/D5 receptor activation on learning. An additional, task-dependent, effect of SKF-38393 on performance seems likely to be due to different levels of dopaminergic activity being needed for optimal learning of the different tasks (Chudasama and Robbins, 2004). Thus a fixed dose of a drug may lead to over-stimulation of dopamine D1/D5 receptors adversely affecting performance in some tasks and optimal receptor stimulation enhancing performance of other tasks. Even though a partial agonist like SKF 38393 would be expected to be less liable to cause over-stimulation of dopamine D1/D5 receptors than a full agonist, given the present findings, the use of partial agonists may not avoid detrimental effects on certain types of learning and memory at a dose that enhances others.[2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9167 mL | 19.5833 mL | 39.1665 mL | |
| 5 mM | 0.7833 mL | 3.9167 mL | 7.8333 mL | |
| 10 mM | 0.3917 mL | 1.9583 mL | 3.9167 mL |