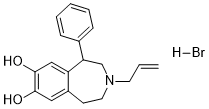
SKF-77434 HBr is a novel potent and selective dopamine D1-like receptor partial agonist. It is the N-Allyl analog ofSKF38393. The direct-acting dopamine D-1 agonists SKF 38393 [(+-)-7-bromo-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-be nzazepin e HCl], SKF 77434 [(+-)-7,8-dihydroxy-3-allyl-1-phenyl-2,3,4,5-tetrahydro-1H-benzazepine HCl] and SKF 75670 [3-methyl-7,8-dihydroxyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benza zep ine HBr] fully substituted for cocaine, whereas the peripherally active dopamine D-1 agonist fenoldopam did not. Of four dopamine D-2 agonists tested, only quinpirole fully substituted; the others (N-0434 [(+-)-2-(N-propyl-N-phenylethylamino)-5-hydroxytetralin], (-)-NPA [R(-)-propylnorapomorphine HCl] and SDZ 208-912 (N-[(8-)-2,6-dimethylergoline-8-yl]-2,2-dimethyl-propanamide)) produced very limited partial substitution (cocaine responding < 32%).
Physicochemical Properties
| Molecular Formula | C19H22BRNO2 | |
| Molecular Weight | 376.29 | |
| Exact Mass | 375.083 | |
| Elemental Analysis | C, 60.65; H, 5.89; Br, 21.23; N, 3.72; O, 8.50 | |
| CAS # | 300561-58-4 | |
| Related CAS # |
|
|
| PubChem CID | 11957703 | |
| Appearance | White to off-white solid powder | |
| LogP | 4.169 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 23 | |
| Complexity | 366 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | Br.C=CCN1CCC2=CC(=C(C=C2C(C2C=CC=CC=2)C1)O)O |
|
| InChi Key | JWQRAXTWDYUBFI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C19H21NO2.BrH/c1-2-9-20-10-8-15-11-18(21)19(22)12-16(15)17(13-20)14-6-4-3-5-7-14;/h2-7,11-12,17,21-22H,1,8-10,13H2;1H | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Dopamine D1 receptor |
| ln Vivo | Twelve rats were trained to press one lever after cocaine injection (3 mg/kg i.p.) and another lever after saline injection. Once rats were reliably discriminating cocaine from saline, other drugs were examined for their efficacies in substituting for cocaine. The dopamine uptake inhibitors WIN 35,428 [2-beta-carbomethoxy-3-beta-(4-fluorophenyl)tropane-1,5-naphthalene - disulfonate] and GBR 12909 (1-[2-bis(4-fluorophenyl)methoxy]ethyl]-4-[3- phenylpropyl]piperazine dihydrochloride) fully substituted for cocaine (cocaine responding > 80%), whereas the peripherally active cocaine methiodide and the 5-hydroxytryptamine uptake inhibitor fluoxetine did not substitute at all. Pentobarbital also failed to produce any cocaine-appropriate responding. Two selective norepinephrine uptake inhibitors were tested: tomoxetine fully substituted for the 3-mg/kg dose of cocaine and nisoxetine approached full substitution (79.7% cocaine responding). The direct-acting dopamine D-1 agonists SKF 38393 [(+-)-7-bromo-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-be nzazepin e HCl], SKF 77434 [(+-)-7,8-dihydroxy-3-allyl-1-phenyl-2,3,4,5-tetrahydro-1H-benzazepine HCl] and SKF 75670 [3-methyl-7,8-dihydroxyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benza zep ine HBr] fully substituted for cocaine, whereas the peripherally active dopamine D-1 agonist fenoldopam did not. Of four dopamine D-2 agonists tested, only quinpirole fully substituted; the others (N-0434 [(+-)-2-(N-propyl-N-phenylethylamino)-5-hydroxytetralin], (-)-NPA [R(-)-propylnorapomorphine HCl] and SDZ 208-912 (N-[(8-)-2,6-dimethylergoline-8-yl]-2,2-dimethyl-propanamide)) produced very limited partial substitution (cocaine responding < 32%)[1]. |
| Animal Protocol |
E¤ects of dopamine agonists on ethanol self-administration [1] Figure 1 shows that the Þve dopamine agonists tested decreased rates of responding for ethanol (R-6Br-APB, F3,21 = 8.54, P < 0.001; SKF 82958, F4,20 = 13.15, P < 0.001; SKF 81297, F3,18 = 14.20, P < 0.001; SKF 38393, F4,16 = 9.94, P < 0.001; SKF 77434, F3,18 = 9.63, P < 0.001). In contrast to the other dopamine agonists, which did not produce statistically signiÞcant e¤ects on water self-administration (SKF 82958, F4,20 = 2.28; SKF 38393, F4,16 = 0.13; SKF 77434, F3,18 = 0.32), R-6Br-APB increased responding for water at the dose of 0.3 mg/kg (F3,21 = 3.32, P < 0.05) (Table 1). ANOVA also indicated a signiÞcant e¤ect of SKF 81297 dose on water selfadministration (F3,18 = 3.57, P < 0.05); however, Dunnetts tests did not reveal signiÞcant di¤erences from control values. |
| References | J Pharmacol Exp Ther.1994Sep;270(3):1041-8. |
| Additional Infomation | N-allyl-1-phenyl-2,3,4,5-tetrahydro-3-benzazepine-7,8-diol hydrobromide is a hydrobromide salt prepared from N-allyl-1-phenyl-2,3,4,5-tetrahydro-3-benzazepine-7,8-diol and one equivalent of hydrogen bromide. Selective dopamine D1-like receptor partial agonist (IC50 values are 19.7 and 2425 nM for binding to D1-like and D2-like receptors respectively). Centrally active following systemic administration in vivo. It has a role as a dopamine agonist and a prodrug. It contains a N-allyl-1-phenyl-2,3,4,5-tetrahydro-3-benzazepinium-7,8-diol(1+). |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6575 mL | 13.2876 mL | 26.5752 mL | |
| 5 mM | 0.5315 mL | 2.6575 mL | 5.3150 mL | |
| 10 mM | 0.2658 mL | 1.3288 mL | 2.6575 mL |