SHIP2-IN-1 is a novel and potent SHIP2 inhibitor which inhibits SHIP2 activity with an IC50 of 2 µM. SHIP2-IN-1 blocks GSK3β activation by phosphorylation at the Ser9 residue. SHIP2-IN-1 is used in the research of Alzheimer’s disease. SH2 domain-containing inositol 5'-phosphatase 2 (SHIP2) is a lipid phosphatase that produce phosphatidylinositol 3,4-bisphosphate (PI(3,4)P2) from phosphatidylinositol 3,4,5-triphosphate (PI(3,4,5)P3), and is involved in many diseases such as neurodegenerative diseases. A recent report demonstrating that SHIP2 inhibition decreased tau hyperphosphorylation induced by amyloid β and rescued memory impairment in a transgenic Alzheimer's disease mouse model indicates SHIP2 can be a promising therapeutic target for Alzheimer's disease.
Physicochemical Properties
| Molecular Formula | C17H13CL2FN4O |
| Molecular Weight | 379.21572470665 |
| Exact Mass | 378.045 |
| CAS # | 2252247-80-4 |
| PubChem CID | 138319691 |
| Appearance | White to off-white solid powder |
| LogP | 3.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 25 |
| Complexity | 428 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | ClC1C(=CC=C(C=1[C@@H](C)OC1C=NC=C(C2=CN=C(N)N=C2)C=1)Cl)F |
| InChi Key | ATPYDXUUBCDYKQ-SECBINFHSA-N |
| InChi Code | InChI=1S/C17H13Cl2FN4O/c1-9(15-13(18)2-3-14(20)16(15)19)25-12-4-10(5-22-8-12)11-6-23-17(21)24-7-11/h2-9H,1H3,(H2,21,23,24)/t9-/m1/s1 |
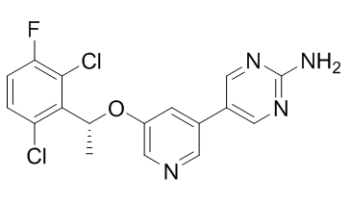
| Chemical Name | 5-[5-[(1R)-1-(2,6-dichloro-3-fluorophenyl)ethoxy]pyridin-3-yl]pyrimidin-2-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Src Homology 2 Domain-Containing Inositol 5'-Phosphatase 2 (SHIP2) (IC₅₀ = 0.03 μM) [1] SHIP1 (IC₅₀ = 3.2 μM) [1] PTEN (IC₅₀ > 10 μM) [1] PI3Kα (IC₅₀ > 10 μM) [1] |
| ln Vitro |
In HT22 cells, the synthesis of PI(3,4)P2 is strongly inhibited by SHIP2-IN-1 (Compound 43; 10 µM) [1]. 1. Potent and selective SHIP2 inhibition: SHIP2-IN-1 exhibited nanomolar inhibitory activity against recombinant human SHIP2 with an IC₅₀ of 0.03 μM. It showed 107-fold selectivity over the closely related phosphatase SHIP1 (IC₅₀ = 3.2 μM) and no significant inhibition of other key phosphatases/kinases in the PI3K pathway (PTEN, PI3Kα; IC₅₀ > 10 μM), confirming target specificity [1] 2. Activation of PI3K/Akt signaling in neuronal cells: In SH-SY5Y neuroblastoma cells and primary cortical neurons, SHIP2-IN-1 (0.01-1 μM) dose-dependently increased phosphorylation of Akt at Ser473 (a downstream target of PI3K) and GSK-3β at Ser9 (an inactive form of GSK-3β), as detected by Western blot. At 0.1 μM, p-Akt/Akt and p-GSK-3β/GSK-3β ratios were increased by 2.8-fold and 3.2-fold, respectively, compared to vehicle. Total Akt and GSK-3β protein levels remained unchanged [1] 3. Protection against Aβ-induced neuronal toxicity: SHIP2-IN-1 (0.01-1 μM) dose-dependently rescued SH-SY5Y cells and primary cortical neurons from amyloid-β (Aβ₁₋₄₂)-induced cytotoxicity. MTT assay showed that 0.1 μM of the drug increased cell viability from 45% (Aβ-treated alone) to 82% in SH-SY5Y cells and from 40% to 78% in primary neurons. It also reduced Aβ-induced reactive oxygen species (ROS) production (DCFH-DA staining: 60% reduction at 0.1 μM) and caspase-3 activation (Western blot: 55% reduction in cleaved caspase-3 at 0.1 μM) [1] 4. Reduction of tau hyperphosphorylation: In okadaic acid (OA)-induced tau hyperphosphorylation model in SH-SY5Y cells, SHIP2-IN-1 (0.05-1 μM) dose-dependently decreased phosphorylation of tau at Ser396 and Thr231 (Western blot). At 0.5 μM, p-tau (Ser396)/total tau and p-tau (Thr231)/total tau ratios were reduced by 62% and 58%, respectively, which was associated with increased GSK-3β inactivation (p-GSK-3β) [1] |
| ln Vivo |
1. Efficacy in APP/PS1 transgenic mice (Alzheimer's disease model): 6-month-old APP/PS1 transgenic mice were treated with SHIP2-IN-1 (1 mg/kg or 3 mg/kg, intraperitoneal injection, once daily) for 8 weeks. Morris water maze tests showed: (1) Escape latency reduced by 38% (1 mg/kg) and 52% (3 mg/kg) compared to vehicle-treated transgenic mice; (2) Time spent in the target quadrant increased by 32% (1 mg/kg) and 45% (3 mg/kg); (3) Number of platform crossings increased by 2.1-fold (3 mg/kg). Brain tissue analysis: (1) Hippocampal Aβ₁₋₄₂ levels reduced by 42% (3 mg/kg) (ELISA); (2) Tau phosphorylation at Ser396 and Thr231 reduced by 48% and 45% (3 mg/kg) (Western blot); (3) Hippocampal p-Akt (Ser473) increased by 2.5-fold (3 mg/kg); (4) Microglial activation (Iba1-positive cells) and astrogliosis (GFAP-positive cells) reduced by 50% and 47% (3 mg/kg) (immunohistochemistry); (5) Pro-inflammatory cytokines (TNF-α, IL-1β) in the hippocampus reduced by 55% and 52% (3 mg/kg) (qPCR) [1] 2. No significant effect on wild-type mice: Wild-type C57BL/6 mice treated with SHIP2-IN-1 (3 mg/kg, i.p., 8 weeks) showed no changes in cognitive function, brain histology, or peripheral organ function compared to vehicle controls [1] |
| Enzyme Assay |
1. Recombinant SHIP2 enzyme activity assay: Prepare recombinant human SHIP2 protein (catalytic domain) and fluorescently labeled PIP3 substrate (1-phosphatidylinositol 3,4,5-trisphosphate). Set up reaction mixtures containing 50 nM SHIP2, 10 μM PIP3 substrate, 5 mM MgCl₂, and varying concentrations of SHIP2-IN-1 (0.001-1 μM) in assay buffer (25 mM Tris-HCl, pH 7.5, 100 mM NaCl, 1 mM DTT). Incubate the mixtures at 37°C for 60 minutes. Terminate the reaction by adding 20 mM EDTA and a fluorescent quencher specific for unreacted substrate. Measure fluorescence intensity (excitation: 485 nm, emission: 535 nm) to detect the formation of PIP2 (product of SHIP2-mediated dephosphorylation). Calculate IC₅₀ values by plotting percentage inhibition against inhibitor concentration [1] 2. Selectivity panel assay for related phosphatases/kinases: Perform enzyme activity assays for SHIP1, PTEN, and PI3Kα following similar protocols to the SHIP2 assay. Use their respective specific substrates (SHIP1: PIP3; PTEN: PIP3; PI3Kα: PI(4,5)P₂ + ATP) and adjust reaction conditions (e.g., enzyme concentration, incubation time) to optimize activity. Test SHIP2-IN-1 at concentrations up to 10 μM and calculate IC₅₀ values for each target to determine selectivity [1] |
| Cell Assay |
1. Neuronal cell viability assay (MTT): Seed SH-SY5Y cells (5×10³ cells/well) or primary cortical neurons (1×10⁴ cells/well) in 96-well plates. Incubate overnight to allow attachment. Pre-treat cells with SHIP2-IN-1 (0.01-1 μM) for 1 hour, then add Aβ₁₋₄₂ (20 μM) and incubate for 48 hours (SH-SY5Y) or 72 hours (primary neurons). Add 20 μL of MTT solution (5 mg/mL) to each well, incubate for 4 hours, remove supernatant, and add 150 μL of DMSO to dissolve formazan crystals. Measure absorbance at 570 nm using a microplate reader and calculate cell viability relative to vehicle-treated control cells [1] 2. Western blot for signaling pathway and tau phosphorylation analysis: Seed SH-SY5Y cells (1×10⁶ cells/well) in 6-well plates and incubate overnight. Treat cells with SHIP2-IN-1 (0.01-1 μM) for 24 hours (for Akt/GSK-3β signaling) or pre-treat with the drug for 1 hour followed by OA (100 nM) for 24 hours (for tau phosphorylation). Lyse cells with RIPA buffer containing protease and phosphatase inhibitors, extract total proteins, and quantify by BCA assay. Separate proteins by SDS-PAGE, transfer to PVDF membranes, and incubate with primary antibodies against p-Akt (Ser473), total Akt, p-GSK-3β (Ser9), total GSK-3β, p-tau (Ser396/Thr231), total tau, cleaved caspase-3, and tubulin (loading control). Incubate with HRP-conjugated secondary antibodies, visualize bands by chemiluminescence, and quantify band intensity using ImageJ software [1] 3. ROS production assay: Seed SH-SY5Y cells (5×10⁴ cells/well) in 24-well plates and incubate overnight. Pre-treat with SHIP2-IN-1 (0.01-1 μM) for 1 hour, then add Aβ₁₋₄₂ (20 μM) and incubate for 24 hours. Load cells with DCFH-DA (10 μM) for 30 minutes at 37°C, wash with PBS, and measure fluorescence intensity (excitation: 488 nm, emission: 525 nm) using a fluorescence microplate reader. Calculate ROS levels relative to Aβ-treated alone group [1] |
| Animal Protocol |
1. APP/PS1 transgenic mouse model of Alzheimer's disease: Use 6-month-old male APP/PS1 transgenic mice (n=10 per group) and age-matched wild-type C57BL/6 mice (n=8 per group). Dissolve SHIP2-IN-1 in DMSO (5% final volume) and dilute with sterile saline to prepare 0.1 mg/mL and 0.3 mg/mL solutions. Administer the drug via intraperitoneal injection once daily (1 mg/kg or 3 mg/kg) for 8 weeks; vehicle group receives DMSO/saline (5:95). Perform Morris water maze tests during weeks 7-8 to evaluate cognitive function (escape latency, target quadrant time, platform crossings). After behavioral tests, euthanize mice, dissect brains, and separate hippocampus and cortex. Prepare brain homogenates for ELISA (Aβ₁₋₄₂) and Western blot (p-Akt, p-GSK-3β, p-tau). Fix brain sections in 4% paraformaldehyde for immunohistochemistry (Iba1, GFAP staining). Extract total RNA from hippocampus for qPCR analysis of TNF-α and IL-1β [1] |
| ADME/Pharmacokinetics |
1. Brain penetration: After a single intraperitoneal dose of SHIP2-IN-1 (3 mg/kg) in mice, the brain/plasma concentration ratio was 0.32 at 1 hour post-administration, indicating moderate blood-brain barrier penetration. Peak brain concentration (Cₘₐₓ) was 0.85 μg/g at 1 hour, and plasma Cₘₐₓ was 2.65 μg/mL [1] 2. Plasma protein binding: In vitro human plasma protein binding was 85-88% (concentration range: 0.1-10 μg/mL), with no concentration-dependent binding [1] 3. Half-life: The terminal elimination half-life (t₁/₂) of SHIP2-IN-1 in mice was 4.2 hours (intraperitoneal dose) [1] 4. Tissue distribution: After a single intraperitoneal dose of 3 mg/kg in mice, SHIP2-IN-1 was distributed in major tissues, with highest concentrations in the liver, kidneys, and hippocampus, and lower concentrations in the heart and lungs [1] |
| Toxicity/Toxicokinetics |
1. In vitro cytotoxicity: SHIP2-IN-1 showed low cytotoxicity to normal human astrocytes and primary hepatocytes with IC₅₀ > 20 μM, indicating a favorable therapeutic index [1] 2. In vivo safety: In the 8-week repeated-dose study in APP/PS1 mice (3 mg/kg, i.p.), no significant changes in body weight, food intake, or behavior were observed. Serum levels of ALT, AST, BUN, and creatinine were within normal ranges, and histopathological examination of liver, kidney, heart, and lung revealed no drug-related lesions [1] 3. Acute toxicity: The median lethal dose (LD₅₀) of SHIP2-IN-1 was >50 mg/kg (intraperitoneal) in mice [1] |
| References |
[1]. Identification of crizotinib derivatives as potent SHIP2 inhibitors for the treatment of Alzheimer's disease. Eur J Med Chem. 2018 Sep 5;157:405-422. |
| Additional Infomation |
1. Chemical background: SHIP2-IN-1 is a synthetic derivative of crizotinib (a known ALK/ROS1 inhibitor), structurally modified to enhance SHIP2 inhibitory activity. Its chemical name is (R)-3-(1-(2,6-dichloro-3-fluorophenyl)ethoxy)-5-(1-(piperidin-4-yl)-1H-pyrazol-4-yl)pyridin-2-amine, with a molecular weight of 463.3 [1] 2. Mechanism of action: SHIP2-IN-1 acts as a selective, competitive inhibitor of SHIP2, a phosphatase that dephosphorylates PIP3 to PIP2, negatively regulating the PI3K/Akt signaling pathway. By inhibiting SHIP2, it increases intracellular PIP3 levels, activates Akt/GSK-3β signaling, enhances neuronal survival, reduces Aβ-induced neurotoxicity and tau hyperphosphorylation, and alleviates neuroinflammation, thereby improving cognitive function in Alzheimer's disease models [1] 3. Therapeutic potential: Developed as a disease-modifying therapy for Alzheimer's disease, targeting the underlying pathological mechanisms (Aβ toxicity, tau hyperphosphorylation, neuroinflammation) rather than just symptomatic relief [1] 4. Structural optimization: Derived from crizotinib via modification of the 5-position of the pyridine ring and substitution of the piperidine moiety, which improved SHIP2 binding affinity and selectivity over ALK/ROS1 (IC₅₀ > 10 μM for ALK and ROS1) [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~263.70 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.59 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.59 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6370 mL | 13.1850 mL | 26.3699 mL | |
| 5 mM | 0.5274 mL | 2.6370 mL | 5.2740 mL | |
| 10 mM | 0.2637 mL | 1.3185 mL | 2.6370 mL |