Physicochemical Properties
| Molecular Formula | C19H17F3N2O2 |
| Molecular Weight | 362.3522 |
| Exact Mass | 362.124 |
| Elemental Analysis | C, 62.98; H, 4.73; F, 15.73; N, 7.73; O, 8.83 |
| CAS # | 680611-86-3 |
| PubChem CID | 11660520 |
| Appearance | Solid powder |
| LogP | 4.925 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 26 |
| Complexity | 488 |
| Defined Atom Stereocenter Count | 0 |
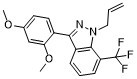
| SMILES | COC1=CC(=C(C=C1)C2=NN(C3=C2C=CC=C3C(F)(F)F)CC=C)OC |
| InChi Key | BMIOASGFHBRKJL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H17F3N2O2/c1-4-10-24-18-14(6-5-7-15(18)19(20,21)22)17(23-24)13-9-8-12(25-2)11-16(13)26-3/h4-9,11H,1,10H2,2-3H3 |
| Chemical Name | 3-(2,4-dimethoxyphenyl)-1-(2-propen-1-yl)-7-(trifluoromethyl)-1H-indazole |
| Synonyms | SGA-360SGA360 SGA 360 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Aryl hydrocarbon receptor (AhR) with a Ki value of 0.6 nM [1] |
| ln Vitro |
In LPS-stimulated RAW 264.7 macrophages, SGA-360 dose-dependently inhibited the production of pro-inflammatory cytokines (TNF-α, IL-6) with IC50 values of 0.8 μM and 1.2 μM, respectively. This inhibition was associated with reduced nuclear translocation of NF-κB and downregulation of iNOS expression [1] - SGA-360 induced CYP1A1 enzyme activity in HepG2 cells, measured by ethoxyresorufin-O-deethylase (EROD) assay, with an EC50 of 0.3 μM, indicating AhR activation [1] - In luciferase reporter assays, SGA-360 selectively activated AhR-mediated transcription in a dose-dependent manner (EC50 = 0.5 μM) without significant cross-reactivity with other nuclear receptors (e.g., estrogen receptor, glucocorticoid receptor) [1] |
| ln Vivo |
SGA360 has anti-inflammatory properties in living things. In a murine model of DSS-induced colitis, oral administration of SGA-360 (10 mg/kg/day for 7 days) significantly reduced disease activity index (DAI) scores, colon shortening, and myeloperoxidase (MPO) activity compared to vehicle controls. Histopathological analysis revealed attenuated mucosal damage and decreased inflammatory cell infiltration [1] - SGA-360 treatment (5 mg/kg, intraperitoneal) in a mouse model of LPS-induced systemic inflammation lowered serum TNF-α levels by 60% compared to untreated animals, suggesting systemic anti-inflammatory effects [1] |
| Enzyme Assay |
AhR binding assay: Recombinant human AhR protein was incubated with SGA-360 and radiolabeled [³H]-TCDD. After incubation, bound ligands were separated by gel filtration, and radioactivity was quantified to determine Ki values. The assay was performed in triplicate at 4°C for 16 hours [1] - EROD activity assay: HepG2 cells were treated with SGA-360 for 24 hours, then lysed and incubated with ethoxyresorufin substrate. Fluorescence intensity (excitation 530 nm, emission 590 nm) was measured to quantify CYP1A1 activity. Results were normalized to protein concentration [1] |
| Cell Assay |
Cytokine production assay: RAW 264.7 cells (5×10⁵ cells/well) were pretreated with SGA-360 (0.1–10 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. TNF-α and IL-6 levels in supernatants were measured by ELISA. Data were analyzed using GraphPad Prism to calculate IC50 values [1] - Luciferase reporter assay: HEK293T cells transfected with an AhR-responsive luciferase plasmid were treated with SGA-360 (0.01–10 μM) for 24 hours. Luciferase activity was measured using a luminometer and normalized to Renilla luciferase control [1] |
| Animal Protocol |
DSS colitis model: C57BL/6 mice received 3% DSS in drinking water for 7 days. SGA-360 (10 mg/kg) was suspended in 0.5% carboxymethylcellulose (CMC) and administered orally once daily starting on day 1. Vehicle control received CMC alone. DAI was scored daily based on weight loss, stool consistency, and rectal bleeding [1] - LPS-induced inflammation model: BALB/c mice were injected intraperitoneally with LPS (5 mg/kg). SGA-360 (5 mg/kg) dissolved in DMSO:saline (1:9) was administered 1 hour prior to LPS challenge. Serum was collected 4 hours later for cytokine analysis [1] |
| ADME/Pharmacokinetics |
Oral bioavailability: SGA-360 showed 25% oral bioavailability in rats, with a plasma Cmax of 1.2 μM and Tmax of 2 hours after a 10 mg/kg dose. Elimination half-life was 4.5 hours [1] - Tissue distribution: Highest drug concentrations were detected in liver and intestine, with moderate levels in spleen and lung. Brain penetration was limited (<5% of plasma levels) [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: No mortality was observed in mice treated with single oral doses of SGA-360 up to 2000 mg/kg. Clinical signs (e.g., sedation) were transient and resolved within 24 hours [1] - Subchronic toxicity: Rats administered SGA-360 (30 mg/kg/day, oral) for 28 days showed no significant changes in hematology, serum biochemistry, or organ weights. Mild hepatic hypertrophy was observed at the highest dose [1] |
| References | [1]. Iain A Murray, et al. Development of a selective modulator of aryl hydrocarbon (Ah) receptor activity that exhibits anti-inflammatory properties. Chem Res Toxicol. 2010 May 17;23(5):955-66. |
| Additional Infomation |
SGA-360 is a small-molecule AhR modulator designed by modifying the indole scaffold. Its structure includes a fluorinated phenyl group to enhance receptor selectivity [1] - The compound exhibits dual AhR activity: agonist at low concentrations (<1 μM) promoting CYP1A1 induction, and antagonist at higher concentrations (>10 μM) blocking LPS-induced cytokine production [1] - SGA-360 demonstrates selectivity for AhR over other dioxin-responsive receptors, with >100-fold lower affinity for estrogen receptor and glucocorticoid receptor [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~689.94 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.90 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.90 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7598 mL | 13.7988 mL | 27.5976 mL | |
| 5 mM | 0.5520 mL | 2.7598 mL | 5.5195 mL | |
| 10 mM | 0.2760 mL | 1.3799 mL | 2.7598 mL |