SBFI-26 (SBFI 26; SBFI26) is a novel, potent, specific and competitive inhibitor of fatty acid binding proteins FABP5 and FABP7 with anti-nociceptive and anti-inflammatory activities. It inhibits FABP5/7 with Kis of 0.9 µM and 0.4 µM, respectively.
Physicochemical Properties
| Molecular Formula | C28H22O4 |
| Molecular Weight | 422.47 |
| Exact Mass | 422.1518 |
| CAS # | 1541207-06-0 |
| Related CAS # |
1541209-75-9 (RR isomer) 1541207-06-0 (SS isomer) |
| PubChem CID | 1002969 |
| Appearance | White to light yellow solid powder |
| LogP | 5.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 32 |
| Complexity | 636 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | C([C@@H]1[C@@H](C2C=CC=CC=2)[C@@H](C(=O)O)[C@@H]1C1C=CC=CC=1)(=O)OC1=CC=CC2C=CC=CC1=2 |
| InChi Key | NVOKBONTLOAJKA-VEYUFSJPSA-N |
| InChi Code | InChI=1S/C28H22O4/c29-27(30)25-23(19-11-3-1-4-12-19)26(24(25)20-13-5-2-6-14-20)28(31)32-22-17-9-15-18-10-7-8-16-21(18)22/h1-17,23-26H,(H,29,30)/t23-,24-,25-,26-/m1/s1 |
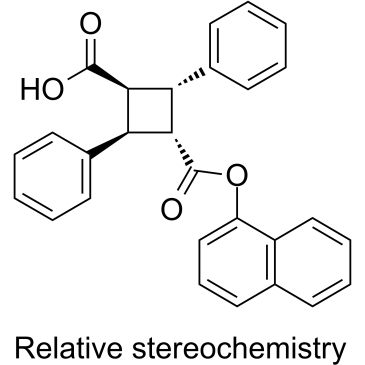
| Chemical Name | (1R,2R,3R,4R)-3-((naphthalen-1-yloxy)carbonyl)-2,4-diphenylcyclobutane-1-carboxylic acid |
| Synonyms | SBFI 26; SBFI-26; SBFI-26; CHEMBL4281879; (S)-SBFI-26; 1541207-06-0; (1s,2s,3s,4s)-3-{[(Naphthalen-1-Yl)oxy]carbonyl}-2,4-Diphenylcyclobutane-1-Carboxylic Acid; 8KS; SCHEMBL15590298; SCHEMBL16866271; SBFI26 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
|
|
| ln Vitro |
|
|
| ln Vivo |
|
|
| Enzyme Assay |
FAAH enzyme assays[1] Enzyme assays measuring the hydrolysis of [14C]AEA in the presence of SB-FI-26 or the FAAH inhibitor URB597 were carried out in HeLa homogenates expressing rat FAAH as described. High throughput fluorescence displacement assay with NBD-stearate[1] FABP5 was purified and delipidated as described previously. FABP5 (30 µg), NBD-stearate (1 µM), and a competitor test compound were incubated in 30 mM Tris-HCl, 100 mM NaCl buffer (pH 7.6). Competitors included arachidonic acid, BMS309403, 48 test compounds from ChemDiv library, SB-FI-26 and SB-FI-49. The initial assay was run with buffer (30 mM Tris-HCl buffer), negative controls (buffer and NBD-stearate), positive controls (buffer, NBD-stearate, FABP5), and experimental wells with a variable test compound added (arachidonic acid or one of the 48 test compounds) at 10 µM. Test compounds that produced high inhibition and proved statistically significant were then added to the fluorescent assay at 10 µM and tested in triplicate to verify their activity. The most effective test compound and BMS309403 were measured in increasing concentrations (0.01–50 µM), as were the SB-FI-26 and γ-truxillic acid 1-naphthyl ester, which were discovered following the test. The fluorescent assays were tested in the wells of Microtest 96-well Assay Plates, Optilux and loss of fluorescence intensity was measured with a FLUOstar OPTIMA spectrofluorometer set to excitation and emission wavelengths of 460 nm and 544 nm, respectively. For the most effective test compounds, IC50 values were calculated with GraphPad Prism. GraphPad Prism was also used to determine the Ki of these select competitors from the equation Ki = IC50/(1+([NBD-stearate]/Kd)). The Kd of NBD-stearate for FABP5 had been determined previously through incubating FABP5 with increasing concentrations of NBD-stearate. One site binding analysis in GraphPad Prism indicated that the Kd of NBD-stearate for FABP5 was 0.16 µM |
|
| Cell Assay |
PPAR transactivation[1] PPARα and PPARγ transactivation assays were performed in HeLa cells exactly as described. Briefly, cells were transfected with the PPAR reporter system, incubated with GW7647, rosiglitazone, or SB-FI-26 for 6 hrs, followed by measurement of luciferase and β-galactosidase activity using a luminometer as described. |
|
| Animal Protocol |
Carrageenan-induced paw edema and thermal hyperalgesia[1] Paw edema was induced by injecting 1% λ-carrageenan (20 µl, in sterile saline) into the plantar surface of the left hind paw and a control solution of saline into the right hind paw using a 27 gauge needle. Paw diameters were measured before carrageenan injection and 4 hours after injection using digital electronic calipers (Fisher) and expressed to the nearest ±0.01 mm. SB-FI-26(20 mg/kg, i.p.) was dissolved in ethanol∶emulphor∶saline (1∶1∶18), requiring sonication and gentle heating for solubilization, and administered 45 min prior to injection of carrageenan. The cannabinoid receptor antagonists, rimonabant and SR144528 (3 mg/kg, i.p.), in ethanol∶emulphor∶saline (1∶1∶18), were injected 15 min before the FABP inhibitor. Edema is reported as the change in paw diameter at 4 hr over the baseline. Changes in paw diameter of saline-injected contralateral paws were negligible. Thermal hyperalgesia measured the latency to withdraw the paw from a focused beam of radiant heat applied to the plantar surface of the hind paw using a Hargreaves plantar apparatus (Ugo Basile) set at an intensity of 3.0. For each mouse, the average latencies consisted of three trials spaced at least 5 minutes apart. The mice were habituated to the test chamber for 30 min. The cutoff time was set at 30 sec. |
|
| References |
[1].Targeting fatty acid binding protein (FABP) anandamide transporters - a novel strategy for development of anti-inflammatory and anti-nociceptive drugs. PLoS One. 2012;7(12):e50968. [2].The Antinociceptive Agent SBFI-26 Binds to Anandamide Transporters FABP5 and FABP7 at Two Different Sites. Biochemistry. 2017 Jul 11;56(27):3454-3462 |
|
| Additional Infomation |
Fatty acid binding proteins (FABPs), in particular FABP5 and FABP7, have recently been identified by us as intracellular transporters for the endocannabinoid anandamide (AEA). Furthermore, animal studies by others have shown that elevated levels of endocannabinoids resulted in beneficial pharmacological effects on stress, pain and inflammation and also ameliorate the effects of drug withdrawal. Based on these observations, we hypothesized that FABP5 and FABP7 would provide excellent pharmacological targets. Thus, we performed a virtual screening of over one million compounds using DOCK and employed a novel footprint similarity scoring function to identify lead compounds with binding profiles similar to oleic acid, a natural FABP substrate. Forty-eight compounds were purchased based on their footprint similarity scores (FPS) and assayed for biological activity against purified human FABP5 employing a fluorescent displacement-binding assay. Four compounds were found to exhibit approximately 50% inhibition or greater at 10 µM, as good as or better inhibitors of FABP5 than BMS309403, a commercially available inhibitor. The most potent inhibitor, γ-truxillic acid 1-naphthyl ester (ChemDiv 8009-2334), was determined to have K(i) value of 1.19±0.01 µM. Accordingly a novel α-truxillic acid 1-naphthyl mono-ester (SB-FI-26) was synthesized and assayed for its inhibitory activity against FABP5, wherein SB-FI-26 exhibited strong binding (K(i) 0.93±0.08 µM). Additionally, we found SB-FI-26 to act as a potent anti-nociceptive agent with mild anti-inflammatory activity in mice, which strongly supports our hypothesis that the inhibition of FABPs and subsequent elevation of anandamide is a promising new approach to drug discovery. Truxillic acids and their derivatives were also shown by others to have anti-inflammatory and anti-nociceptive effects in mice and to be the active component of Chinese a herbal medicine (Incarvillea sinensis) used to treat rheumatism and pain in humans. Our results provide a likely mechanism by which these compounds exert their effects.[1] Human FABP5 and FABP7 are intracellular endocannabinoid transporters. SBFI-26 is an α-truxillic acid 1-naphthyl monoester that competitively inhibits the activities of FABP5 and FABP7 and produces antinociceptive and anti-inflammatory effects in mice. The synthesis of SBFI-26 yields several stereoisomers, and it is not known how the inhibitor binds the transporters. Here we report co-crystal structures of SBFI-26 in complex with human FABP5 and FABP7 at 2.2 and 1.9 Å resolution, respectively. We found that only (S)-SBFI-26 was present in the crystal structures. The inhibitor largely mimics the fatty acid binding pattern, but it also has several unique interactions. Notably, the FABP7 complex corroborates key aspects of the ligand binding pose at the canonical site previously predicted by virtual screening. In FABP5, SBFI-26 was unexpectedly found to bind at the substrate entry portal region in addition to binding at the canonical ligand-binding pocket. Our structural and binding energy analyses indicate that both R and S forms appear to bind the transporter equally well. We suggest that the S enantiomer observed in the crystal structures may be a result of the crystallization process selectively incorporating the (S)-SBFI-26-FABP complexes into the growing lattice, or that the S enantiomer may bind to the portal site more rapidly than to the canonical site, leading to an increased local concentration of the S enantiomer for binding to the canonical site. Our work reveals two binding poses of SBFI-26 in its target transporters. This knowledge will guide the development of more potent FABP inhibitors based upon the SBFI-26 scaffold.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3670 mL | 11.8352 mL | 23.6703 mL | |
| 5 mM | 0.4734 mL | 2.3670 mL | 4.7341 mL | |
| 10 mM | 0.2367 mL | 1.1835 mL | 2.3670 mL |