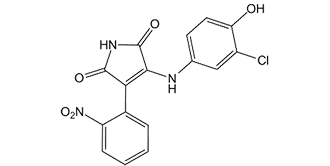
SB415286 is a novel, potent, selective, cell permeable and ATP-competitive GSK3α (glycogen synthase kinase-3alpha) inhibitor with potential anti-inflammatory activity. It has an equal inhibitory effect on GSK3α and an IC50/Ki of 78 nM/31 nM. In human liver cells, SB-415286 reduced GSK-3β activity, increased glycogen synthesis, and induced the expression of a reporter gene controlled by catenin-LEF/TCF in HEK293 cells. It can stop the cell death brought on by suppressed PI3k pathway activity in primary neurons. Additional research revealed that SB-415286's decreased GSK3β activity could prevent the rapamycin-mediated down-regulation of cyclin D1, cell cycle arrest, and chemosensitivity. A multifunctional serine/threonine kinase called glycogen synthase kinase-3 (GSK-3) is crucial for the necrosis and apoptosis of cardiomyocytes.
Physicochemical Properties
| Molecular Formula | C16H10CLN3O5 |
| Molecular Weight | 359.7207 |
| Exact Mass | 359.03 |
| Elemental Analysis | C, 53.42; H, 2.80; Cl, 9.85; N, 11.68; O, 22.24 |
| CAS # | 264218-23-7 |
| Related CAS # | 264218-23-7 |
| PubChem CID | 4210951 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 595.8±50.0 °C at 760 mmHg |
| Flash Point | 314.1±30.1 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.746 |
| LogP | 2.06 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 25 |
| Complexity | 617 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | ClC1=C(C([H])=C([H])C(=C1[H])N([H])C1C(N([H])C(C=1C1=C([H])C([H])=C([H])C([H])=C1[N+](=O)[O-])=O)=O)O[H] |
| InChi Key | PQCXVIPXISBFPN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H10ClN3O5/c17-10-7-8(5-6-12(10)21)18-14-13(15(22)19-16(14)23)9-3-1-2-4-11(9)20(24)25/h1-7,21H,(H2,18,19,22,23) |
| Chemical Name | 3-(3-chloro-4-hydroxyanilino)-4-(2-nitrophenyl)pyrrole-2,5-dione |
| Synonyms | SB-415286; SB415286; SB 415286 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
hGSK-3α (IC50 = 77.5 nM); hGSK-3β (IC50 = 77.5 nM) Glycogen Synthase Kinase 3 (GSK3), including GSK3β and GSK3α. For GSK3β, the IC₅₀ value was 10 nM; for GSK3α, the IC₅₀ value was 100 nM. No inhibitory activity against other kinases (e.g., cdk2, erk1, p38) was detected at concentrations up to 1 μM [1] |
| ln Vitro |
SB 415286 (SB-415286) inhibits human GSK-3α with an IC50 of 77.5 nM, and a Ki of 30.75 nM. SB-415286 stimulates glycogen synthesis in the Chang human liver cell line with EC50 of 2.9 μM. SB-415286 stimulates glycogen synthase activity in Chang human liver cells. In HEK293 cells, SB-415286 activates the transcription of a reporter gene controlled by the β-catenin-LEF/TCF pathway[1]. SB 415286 (SB-415286, 5-44 μM) attenuates B65 cell loss mediated by 1 mM H2O2. SB-415286 (5-44 μM) causes a significant dose-dependent decrease in the fluorescence intensity of DCF, and attenuates B65 ROS production as mediated by 1 mM H2O2. SB-415286 (5-44 μM) also attenuates ROS production in CGN mediated by 1 mM H2O2[2]. SB-415286 (50 µM) induces a substantial suppression of immunoprecipitated GSK3 activity by 97%[3]. 1. In Chinese Hamster Ovary (CHO) cells stably expressing human insulin receptors, treatment with SB415286 (1 μM, 24 hours) increased glycogen synthesis by ~2.2-fold (measured via [¹⁴C]-glucose incorporation) and activated glycogen synthase (GS) — the phosphorylation of GS at Ser⁶⁴1 (inhibitory site) was reduced by ~60% (Western blot). The drug also potentiated insulin-induced glycogen synthesis: in the presence of 1 nM insulin, SB415286 (1 μM) further enhanced glycogen accumulation by ~1.5-fold [1] 2. In B65 rat neuroblastoma cells, pretreatment with SB415286 (0.1 μM, 1 μM, 10 μM, 1 hour before H₂O₂ exposure) dose-dependently protected against hydrogen peroxide (H₂O₂, 200 μM)-induced cell death. At 1 μM, cell viability (MTT assay) increased from 35% (H₂O₂ alone) to 78% (~123% protection). The drug reduced H₂O₂-induced apoptosis: TUNEL-positive cells decreased by ~55% at 1 μM, and caspase-3 activity (fluorometric assay) was suppressed by ~60% at 1 μM. It also lowered intracellular reactive oxygen species (ROS) levels: DCFH-DA staining showed a ~45% reduction in ROS at 1 μM [2] 3. In primary rat cortical neurons (cultured for 10 days), SB415286 (1 μM, 10 μM, 1 hour pre-H₂O₂) similarly mitigated H₂O₂ (100 μM)-induced neurotoxicity. At 10 μM, neuron survival (NeuN immunostaining) increased from 40% to 82%, and ROS production was reduced by ~50% [2] 4. In differentiated L6 rat skeletal muscle myotubes, SB415286 (1 μM, 4 hours) increased glucose transport by ~1.8-fold (measured via [³H]-2-deoxyglucose uptake). It enhanced insulin-stimulated glucose transport: combined with 10 nM insulin, glucose uptake was ~2.3-fold higher than insulin alone. Western blot analysis showed that SB415286 (1 μM) reduced GSK3β phosphorylation at Ser⁹ (a marker of GSK3β inhibition) by ~70% [3] |
| ln Vivo | SB 415286 (10 mg/kg twice daily) administration decreases the intensity and severity of the rat colonic inflammation caused by trinitrobenzene sulphonic acid (TNBS), as well as the loss of body weight, which is linked to the downregulation of NF-B activity, which is involved in the production of proinflammatory mediators. In vivo growth of Neuro-2A cells in nude mice is markedly slowed down by SB 415286 treatment at 1 mg/kg. |
| Enzyme Assay |
GSK-3 kinase activity is measured, in the presence or absence of SB-216763 or SB-415286, in a reaction mixture containing final concentrations of: 1 nM human GSK-3α or rabbit GSK3α; 50 mM MOPS pH 7.0; 0.2 mM EDTA; 10 mM Mg-acetate; 7.5 mM β-mercaptoethanol; 5% (w/v) glycerol; 0.01% (w/v) Tween-20; 10% (v/v) DMSO; 28 μM GS-2 peptide substrate. The glycogen synthase region that the GS-2 peptide sequence corresponds to is one that GSK-3 phosphorylates. The addition of 0.34 μCi [33P]γ-ATP (IC50 determinations) or 2.7 μCi [33P]γ-ATP (Ki determinations). starts the assay. The total ATP concentration is 10 μM (based on IC50 calculations) or 0 to 45 μM (based on Ki calculations). After 30 minutes of room temperature incubation, the assay is terminated by adding a third of the assay volume of 2.5% (v/v) H3PO4 containing 21 mM ATP. After being spotted onto P30 phosphocellulose mats, samples are washed six times in 0.5% (v/v) H3PO4 before being analyzed. In sample bags containing Wallac betaplate scintillation fluid, the filter mats are sealed. By counting the mats in a Wallac microbeta scintillation counter, one can determine the amount of 33P incorporated into the substrate peptide[1]. 1. GSK3β kinase activity assay: Recombinant human GSK3β (5 ng) was incubated with a synthetic peptide substrate (sequence: YRRAAVPPSPSLSRHSSPHQpSEDEEE, 50 μM) in reaction buffer containing 20 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mM DTT, and 10 μM [γ-³²P]-ATP. SB415286 (0.1 nM-1 μM) was added, and the mixture was incubated at 30°C for 30 minutes. The reaction was terminated by spotting 20 μL of the mixture onto phosphocellulose paper, which was washed 3 times with 1% phosphoric acid to remove unincorporated [γ-³²P]-ATP. Radioactivity was measured via liquid scintillation counting, and the IC₅₀ for GSK3β was calculated from the dose-response curve [1] 2. GSK3α kinase activity assay: The protocol was identical to the GSK3β assay, except recombinant human GSK3α (5 ng) was used. The IC₅₀ for GSK3α was determined using the same concentration range of SB415286 and data analysis method [1] 3. Kinase selectivity assay: For other kinases (cdk2, erk1, p38), recombinant enzymes (5-10 ng) were incubated with their respective peptide substrates, [γ-³²P]-ATP, and SB415286 (0.1 nM-1 μM) in kinase-specific reaction buffers. Radioactivity was measured as described above, and no inhibitory activity was detected for these kinases [1] |
| Cell Assay |
B65 cells are used after 24 h of in vitro culture. CGN are used 7-8 days after in vitro creation. One hour before adding H2O2, lithium and SB-415286 are added to the neuronal preparation at the exact concentrations (50 M to 1 mM) after being dissolved in culture media and DMSO, respectively. The MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium] method is used to measure the loss in cell viability. MTT is added to the cells at a final concentration of 250 M, and after 1 hour of incubation, a dark blue formazan product is produced as a result of MTT's reduction. Following the removal of the media, the cells are dissolved in dimethylsulfoxide. Utilizing a microplate reader, the amount of formazan produced is determined by the change in absorbency at 595 nm. Results are presented as percentages of viability. The absorbency measured from non-treated cells is taken to be 100%[2]. 1. CHO cell glycogen synthesis assay: CHO cells (stably expressing human insulin receptors) were seeded in 24-well plates and cultured in DMEM + 10% FBS until 80% confluence. Cells were serum-starved for 16 hours, then treated with SB415286 (0.1 μM-1 μM) ± insulin (1 nM) for 24 hours. [¹⁴C]-glucose (0.5 μCi/mL) was added for the final 4 hours of treatment. Cells were washed with cold PBS, lysed with 10% TCA, and glycogen was precipitated overnight at 4°C. Precipitated glycogen was washed with ethanol, dissolved in water, and radioactivity was measured via liquid scintillation counting to quantify glycogen synthesis [1] 2. B65 cell H₂O₂ toxicity assay: B65 rat neuroblastoma cells were seeded in 96-well plates at 5×10³ cells/well and cultured in DMEM + 10% FBS for 24 hours. Medium was replaced with serum-free DMEM, and SB415286 (0.1 μM-10 μM) was added 1 hour before H₂O₂ (200 μM). After 24 hours, MTT reagent (0.5 mg/mL) was added, incubated at 37°C for 4 hours, dissolved in DMSO, and absorbance was measured at 570 nm to calculate viability. For apoptosis detection, cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and stained with TUNEL reagent; TUNEL-positive cells were counted under a fluorescence microscope [2] 3. Primary cortical neuron H₂O₂ protection assay: Cortices from E18 rat embryos were dissociated and plated at 1×10⁴ cells/well in 96-well plates (poly-L-lysine-coated). Cells were cultured in Neurobasal medium + B27 for 10 days. On day 10, SB415286 (1 μM-10 μM) was added 1 hour before H₂O₂ (100 μM). After 24 hours, cells were fixed and stained with NeuN antibody (neuron-specific marker); viable neurons were counted via immunofluorescence [2] 4. L6 myotube glucose transport assay: L6 cells were seeded in 24-well plates and differentiated into myotubes by culturing in DMEM + 2% horse serum for 7 days. Myotubes were serum-starved for 4 hours, then treated with SB415286 (1 μM) ± insulin (10 nM) for 4 hours. [³H]-2-deoxyglucose (0.1 μCi/mL) was added for 10 minutes, and cells were washed with cold PBS containing 200 μM phloretin. Cells were lysed with 0.1% SDS, and radioactivity was measured to determine glucose uptake [3] |
| Animal Protocol |
Male Wistar rats with acute colitis provoked by trinitrobenzene sulphonic acid (TNBS) ~1 mg/kg Administered subcutaneously twice daily |
| Toxicity/Toxicokinetics |
In vitro, SB415286 (up to 10 μM) showed no cytotoxicity in CHO cells, B65 cells, primary cortical neurons, or L6 myotubes (viability >90% vs. control, MTT assay) [1], [2], [3] |
| References |
[1]. Selective small molecule inhibitors of glycogen synthase kinase-3 modulate glycogen metabolism and gene transcription. Chem Biol. 2000 Oct;7(10):793-803. [2]. Neuroprotective effects of SB-415286 on hydrogen peroxide-induced cell death in B65 rat neuroblastoma cells and neurons. Int J Dev Neurosci. 2008 May-Jun;26(3-4):269-76. [3]. Use of lithium and SB-415286 to explore the role of glycogen synthase kinase-3 in the regulation of glucose transport and glycogen synthase. Eur J Biochem. 2003 Sep;270(18):3829-38. |
| Additional Infomation |
SB 415286 is a member of the class of maleimides carrying 3-chloro-4-hydroxyphenylamino and 2-nitrophenyl substituents at positions 3 and 4 respectively. It has a role as an EC 2.7.11.26 (tau-protein kinase) inhibitor, an antioxidant, a neuroprotective agent and an apoptosis inducer. It is a member of maleimides, a member of phenols, a member of monochlorobenzenes, a substituted aniline, a secondary amino compound and a C-nitro compound. 3-(3-chloro-4-hydroxyphenylamino)-4-(4-nitrophenyl)-1H-pyrrole-2,5-dione has been reported in Penicillium with data available. 1. SB415286 is a selective, ATP-competitive inhibitor of GSK3, with higher potency against GSK3β (IC₅₀=10 nM) than GSK3α (IC₅₀=100 nM). Its selectivity for GSK3 over other kinases reduces the risk of off-target effects [1] 2. The neuroprotective effects of SB415286 against H₂O₂-induced damage are mediated by inhibiting GSK3-dependent apoptosis (reducing caspase-3 activation) and lowering intracellular ROS production, suggesting potential applications in neurodegenerative diseases associated with oxidative stress [2] 3. In metabolic tissues (skeletal muscle, CHO cells), SB415286 improves glucose metabolism by promoting glucose transport and glycogen synthesis, and it potentiates insulin signaling — these effects support its potential as a therapeutic agent for type 2 diabetes [1], [3] 4. Unlike lithium (a non-selective GSK3 inhibitor), SB415286 does not affect other enzymes targeted by lithium (e.g., inositol monophosphatase), making it a more specific tool for studying GSK3 function [3] |
Solubility Data
| Solubility (In Vitro) |
DMSO: 72~100 mg/mL (200.2~278 mM) Ethanol: ~72 mg/mL (~200.2 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.95 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.95 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7799 mL | 13.8997 mL | 27.7994 mL | |
| 5 mM | 0.5560 mL | 2.7799 mL | 5.5599 mL | |
| 10 mM | 0.2780 mL | 1.3900 mL | 2.7799 mL |