Physicochemical Properties
| Molecular Formula | C5H5NOS |
| Molecular Weight | 127.1643 |
| Exact Mass | 127.009 |
| CAS # | 1121-30-8 |
| Related CAS # | 15922-78-8 (hydrochloride salt) |
| PubChem CID | 1570 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.43g/cm3 |
| Boiling Point | 253.8ºC at 760mmHg |
| Flash Point | 107.3ºC |
| Vapour Pressure | 0.00275mmHg at 25°C |
| Index of Refraction | 1.732 |
| LogP | 1.454 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 8 |
| Complexity | 162 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | YBBJKCMMCRQZMA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C5H5NOS/c7-6-4-2-1-3-5(6)8/h1-4,7H |
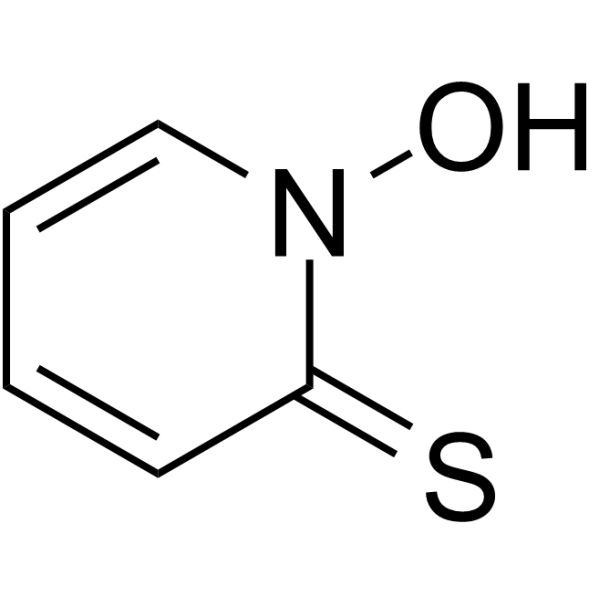
| Chemical Name | 1-hydroxypyridine-2-thione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Following oral ingestion, only the pyrithione moiety is absorbed. Less than 1% of administered zinc pyrithione is absorbed from the skin. Radioabeled Zn pyrithione administered to rats, rabbits and monkeys, either orally or via intraperitoneal injection were absorbed into circulatin to extent of 80-90%. The major route of elimination from the body after oral administration in rat was urine, with S-glucuronides of 2-mercaptopyridine-N-oxide being the major metabolites and 2-mercaptopyridine-N-oxide as the minor metabolite. Following oral administration, the majority of zinc is eliminated in the feces. Upon dermal administration, >90% of recovery was obtained from washings of the application site of pigs. Urinary excretion was 3% in animals with intact skin. Metabolism / Metabolites After oral administration in rabbits, rats, monkeys, and dogs, pyrithione zinc is biotransformed into 2-pyridinethiol 1-oxide S-glucuronide and 2-pyridinethiol S-glucuronide. |
| References |
[1]. Zinc pyrithione inhibits yeast growth through copper influx and inactivation of iron-sulfur proteins. Antimicrob Agents Chemother. 2011 Dec;55(12):5753-60. [2]. In vitro studies with 1-hydroxy-2(1H) pyridinethione. Proc Soc Exp Biol Med. 1953 Jan;82(1):122-4. |
| Additional Infomation |
Pyrithione is a pyridinethione that is pyridine-2(1H)-thione in which the hydrogen attached to the nitrogen is replaced by a hydroxy group. It is a Zn(2+) ionophore; the zinc salt is used as an antifungal and antibacterial agent. It has a role as an ionophore. It is a pyridinethione and a monohydroxypyridine. It is a tautomer of a pyridine-2-thiol N-oxide. Pyrithione zinc, or zinc pyrithione or zinc pyridinethione, is a coordination complex consisted of pyrithione ligands chelated to zinc (2+) ions via oxygen and sulfur centers. In the crystalline state, it exists as a centrosymmetric dimer. Due to its dynamic fungistatic and bacteriostatic properties, pyrithione zinc is used to treat dandruff and seborrheic dermatitis. Dandruff is a common scalp disease affecting >40% of the world's adult population, and may be caused by fungi such as Malassezia globosa and M. restricta. Pyrithione zinc is commonly found as an active ingredient in OTC antidandruff topical treatments such as shampoos. It mediates its action by increasing the cellular levels of copper, and damaging iron-sulfur clusters of proteins essential for fungal metabolism and growth. Due to low solubility, pyrithione zinc released from the topical formulations is deposited and retained relatively well onto the target skin surfaces. Other uses of pyrithione zinc include additive in antifouling outdoor paints and algaecide. While its use has been approved in the early 1960's by the FDA, safety and effectiveness of pyrithione zinc has been reported for decades. It is not shown to have any significant estrogenic activity according to the in vivo and in vitro assays. Pyrithione has been reported in Marsypopetalum modestum with data available. Pyrithione is a fungistatic and antimicrobial derivative of aspergillic acid. Although the exact mechanism of action remains to be fully elucidated, pyrithione appears to interfere with membrane transport ultimately leading to a loss of metabolic control. See also: Pyrithione Zinc (active moiety of); Pyrithione Sodium (has salt form). Drug Indication Indicated for the treatment of dandruff and seborrheic dermatitis. Mechanism of Action Inhibition of fungal growth by pyrithione zinc is linked to increased copper uptake and cellular levels of copper, which is demonstrated by decreased CTR1-lacZ expression and slightly increased CUP1-lacZ expression in affected microorganisms. The coordination complex of pyrithione zinc dissociates, and pyrithione ligand forms a CuPT complex from available extracellular copper in the target organism. Pyrithione acts as an ionophore, interacting nonspecifically with the plasma membrane to shuttle copper into the cell, and facilitates copper transport across intracellular membranes. Copper may be shuttled into the mitochondria. Copper inactivates iron-sulfur (Fe-S) cluster-containing proteins via a mechanism similar to that described for copper-induced growth inhibition in bacteria. Decreased activity of Fe-S proteins leads to inhibition of fungal metabolism and fungal growth. Pyrithione zinc has been shown to slightly increase the levels of zinc. Pharmacodynamics Pyrithione zinc has a broad antimicrobial spectrum of activity, including fungi, gram-positive and gram-negative bacteria. Pyrithione zinc is effective against Malassezia and all other fungi, especially the Malassezia species found on scalp. In patients with dandruff, treatment with pyrithione zinc reduced the amount of fungus on the scalp, which reduces the amount of free fatty acids, thereby reducing scalp flaking and itch. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~983.01 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (16.36 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (16.36 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (16.36 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.8641 mL | 39.3205 mL | 78.6411 mL | |
| 5 mM | 1.5728 mL | 7.8641 mL | 15.7282 mL | |
| 10 mM | 0.7864 mL | 3.9321 mL | 7.8641 mL |