Physicochemical Properties
| Molecular Formula | C20H16O5 |
| Molecular Weight | 336.34 |
| Exact Mass | 336.099 |
| CAS # | 18642-23-4 |
| PubChem CID | 5281806 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 458.8±34.0 °C at 760 mmHg |
| Melting Point | 290-292° |
| Flash Point | 231.3±25.7 °C |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.689 |
| LogP | 5.03 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 25 |
| Complexity | 554 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O1C2C([H])=C(C([H])=C([H])C=2C2C(=O)OC3C([H])=C(C(C([H])([H])/C(/[H])=C(\C([H])([H])[H])/C([H])([H])[H])=C([H])C=3C1=2)O[H])O[H] |
| InChi Key | YABIJLLNNFURIJ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H16O5/c1-10(2)3-4-11-7-14-17(9-15(11)22)25-20(23)18-13-6-5-12(21)8-16(13)24-19(14)18/h3,5-9,21-22H,4H2,1-2H3 |
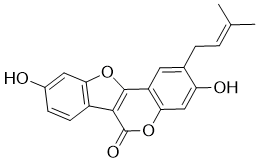
| Chemical Name | 3,9-dihydroxy-2-(3-methylbut-2-enyl)-[1]benzofuro[3,2-c]chromen-6-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Psoralen treatment (10, 15, 20, and 25 μM; 24 hours) was sensitive to clear breast cancer cell (BCC) populations (ALDH- cells, ALDH+ cells, and commercial BSCS), with an IC50 ranging from 18 to 21 μM. On the other hand, MCF-12A cells treated with psoralidin (30 μM; 24 hours) were able to significantly induce ALDH- cells, ALDH+ cells, and commercial BCSC[2]. Psoralidin's effects on treating ALDH- and ALDH+ cells in AIDS [2] |
| ln Vivo | Psoralen (5 mg/kg) reduces inflammation in BALB/c infrared-irradiated lungs by modifying the expression of pro-inflammatory cytokines that are crucial in inflammation [1]. |
| Cell Assay |
Cell viability assay [2] Cell Types: ALDH- cells, ALDH+ cells, commercial breast cancer stem cells (BSCS) and normal mammary epithelial cells (MCF-12A) Tested Concentrations: 10, 15 , 50 and 25. μM Incubation Duration: 24 hrs (hours) Experimental Results: IC50 for ALDH- cells, ALDH+ cells, commercial BCSC was 18 to 21 μM. Apoptosis analysis [2] Cell Types: ALDH- cells, ALDH+ cells and commercial BCSC Tested Concentrations: 20 and 30 μM Incubation Duration: 24 hrs (hours) Experimental Results: No obvious apoptosis was observed in all three cell types after 20 μM treatment death induction. However, at 30 μMin, 53.60%, 44.1%, and 45.9% of ALDH- cells, ALDH+ cells, and commercial BCSCs were apoptotic, respectively. |
| Animal Protocol |
Animal/Disease Models: balb/c (Bagg ALBino) mouse[1] Doses: 5 mg/kg Route of Administration: intraperitoneal (ip) injection; 30 minutes before and 1 hour after IR irradiation (20 Gy). Experimental Results: Anti-inflammatory effects on mice irradiated with infrared rays. |
| References |
[1]. Psoralidin, a dual inhibitor of COX-2 and 5-LOX, regulates ionizing radiation (IR)-induced pulmonary inflammation.Biochem Pharmacol. 2011 Sep 1;82(5):524-34. [2]. Silencing NOTCH signaling causes growth arrest in both breast cancer stem cells and breast cancer cells.Br J Cancer. 2013 Nov 12;109(10):2587-96. |
| Additional Infomation |
Psoralidin is a member of the class of coumestans that is coumestan substituted by hydroxy groups at positions 3 and 9 and a prenyl group at position 2 respectively. It has a role as a plant metabolite and an estrogen receptor agonist. It is a member of coumestans, a polyphenol and a delta-lactone. It is functionally related to a coumestan. Psoralidin has been reported in Phaseolus lunatus, Dolichos trilobus, and other organisms with data available. See also: Cullen corylifolium fruit (part of). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~148.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.08 mg/mL (6.18 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9732 mL | 14.8659 mL | 29.7318 mL | |
| 5 mM | 0.5946 mL | 2.9732 mL | 5.9464 mL | |
| 10 mM | 0.2973 mL | 1.4866 mL | 2.9732 mL |