Physicochemical Properties
| Exact Mass | 461.105 |
| Elemental Analysis | C, 54.66; H, 4.37; F, 4.12; N, 9.11; O, 20.80; S, 6.95 |
| CAS # | 123447-62-1 |
| PubChem CID | 65947 |
| Appearance | Typically exists as off-white to light yellow solids at room temperature |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 633.2±65.0 °C at 760 mmHg |
| Melting Point | 211-214°C |
| Flash Point | 336.8±34.3 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.721 |
| LogP | 3.27 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 32 |
| Complexity | 931 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | PWNMXPDKBYZCOO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H20FN3O6S/c1-10-16(31-21(29)30-10)9-23-3-5-24(6-4-23)15-8-14-12(7-13(15)22)18(26)17(20(27)28)19-25(14)11(2)32-19/h7-8,11H,3-6,9H2,1-2H3,(H,27,28) |
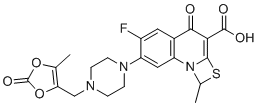
| Chemical Name | 6-fluoro-1-methyl-7-[4-[(5-methyl-2-oxo-1,3-dioxol-4-yl)methyl]piperazin-1-yl]-4-oxo-1H-[1,3]thiazeto[3,2-a]quinoline-3-carboxylic acid |
| Synonyms | Prulifloxacin;NM441; Pruvel; Quisnon; NM-441; Prulifloxacin [INN]; Sword; NM 441; NM-441 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluoroquinolone antibiotic |
| ln Vitro | Minimum inhibitory concentrations (MICs) and mutant prevention concentrations (MPCs) of prulifloxacin against 30 strains of Escherichia coli isolated from urinary tract infections as well as the 'biological cost' related to acquisition of resistance to the same drug in 10 uropathogenic E. coli were assessed. In terms of MIC(90), prulifloxacin was more potent than ciprofloxacin and levofloxacin. Prulifloxacin produced lower or equal MPC values than the other two fluoroquinolones (93.3% and 73.3% compared with levofloxacin and ciprofloxacin, respectively). Compared with susceptible strains, prulifloxacin-resistant mutants showed a reduced rate of growth (ranging from 20.0% to 98.0% in different culture media and incubation conditions) and a decreased fitness index (ranging from 0.959 to 0.999). They were also impaired in their ability to adhere to uroepithelial cells and urinary catheters (11.7-66.4% and 16.3-78.3% reduction, respectively) and showed a lower surface hydrophobicity (51.2-76.0%). They were more susceptible to ultraviolet irradiation (30.6-93.8% excess mortality), showed increased resistance to colicins and diminished transfer of plasmids (<1-8.5x10(-8) vs. 3.3x10(-7)-2.4x10(-4)). Synthesis of haemolysin and type I fimbriae and production of flagella were also adversely affected. This study demonstrates a strict relationship between acquisition of prulifloxacin resistance and loss of important virulence traits. In this transition, E. coli pays a severe biological cost that entails a general reduction of fitness, thus compromising competition with susceptible wild-type strains in the absence of the drug[3]. |
| ln Vivo | The peak plasma concentration of NM394 was 2.39 μg/ml following oral administration of NM441 at a dose of 20 mg/kg to dogs, as opposed to 0.63 μg/ml when NM394 was given alone. For treating Staphylococcus aureus-caused systemic infections, NM441 is twice as effective as ciprofloxacin and as effective as ofloxacin. In terms of efficacy against streptococcal infections, NM441 outperforms ofloxacin by two to three times and ciprofloxacin by five times [1]. |
| Animal Protocol | NM441 is a lipophilic prodrug of a new thiazeto-quinoline carboxylic acid derivative NM394, and when it is administered orally it is readily absorbed and hydrolyzed to its parent compound. After oral administration of NM441 at a dose of 20 mg/kg to dogs, the peak concentration of NM394 in plasma was 2.39 micrograms/ml, whereas it was 0.63 micrograms/ml for NM394 administered alone. The in vivo activity of NM441 was compared with those of ciprofloxacin, ofloxacin, and enoxacin in mouse protection studies. NM441 was as effective as ofloxacin and was twice as effective as ciprofloxacin against systemic infection with Staphylococcus aureus. Against infections with streptococci, NM441 was two to three times as effective as ofloxacin and five times as effective as ciprofloxacin. Against infection with Escherichia coli, NM441 was as effective as ciprofloxacin and ofloxacin, but against infections with Klebsiella pneumoniae, Serratia marcescens, and Pseudomonas aeruginosa, NM441 was two to four times as effective as ciprofloxacin and ofloxacin. NM441 was three to seven times as effective as enoxacin in systemic infections. Against urinary tract infections with E. coli, NM441 reduced the number of bacterial CFU per gram of kidney by 1 to 2 log10 more and, with P. aeruginosa, by 1 to 6 log10 more than did ciprofloxacin and ofloxacin. Against respiratory tract infections with K. pneumoniae, NM441 was as effective as ofloxacin and was twice as effective as ciprofloxacin[1]. |
| References |
[1]. M Ozaki, et al. In vivo evaluation of NM441, a new thiazeto-quinoline derivative. Antimicrob Agents Chemother. 1991 Dec;35(12):2496-9. [2]. Guillem Prats, et al. Prulifloxacin: a new antibacterial fluoroquinolone. Expert Rev Anti Infect Ther. 2006 Feb;4(1):27-41. [3]. In vitro activity of prulifloxacin against Escherichia coli isolated from urinary tract infections and the biological cost of prulifloxacin resistance. Int J Antimicrob Agents . 2007 Jun;29(6):679-87. [4]. Correlation among the toxicity profiling (28-days repeated oral dose toxicity), toxicokinetics and tissue distribution data of ulifloxacin, the active metabolite of prulifloxacin in Wistar albino rats. Environ Toxicol Pharmacol . 2012 Sep;34(2):588-607. |
| Additional Infomation |
Prulifloxacin is a quinolone antibiotic and a fluoroquinolone antibiotic. Prulifloxacin has been investigated for the treatment of Urinary Tract Infection. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~12.5 mg/mL (~27.09 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (2.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (2.71 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |