Physicochemical Properties
| Molecular Formula | C10H19NO4 |
| Molecular Weight | 217.26216 |
| Exact Mass | 217.131 |
| CAS # | 17298-37-2 |
| PubChem CID | 107738 |
| Appearance | White to off-white solid powder |
| Vapour Pressure | 0mmHg at 25°C |
| Index of Refraction | 1.65 |
| LogP | 0.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 15 |
| Complexity | 227 |
| Defined Atom Stereocenter Count | 0 |
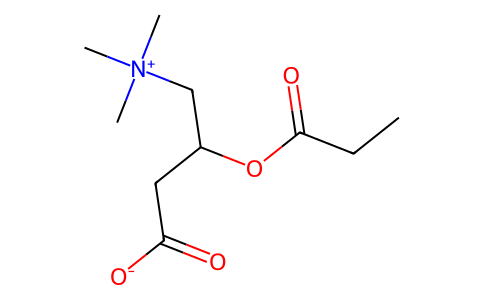
| SMILES | CCC(OC(CC([O-])=O)C[N+](C)(C)C)=O |
| InChi Key | UFAHZIUFPNSHSL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H19NO4/c1-5-10(14)15-8(6-9(12)13)7-11(2,3)4/h8H,5-7H2,1-4H3 |
| Chemical Name | 3-propanoyloxy-4-(trimethylazaniumyl)butanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Most (54-86%) dietary carnitine is absorbed in the small intestine and enters the bloodstream. The kidneys efficiently conserve carnitine, so even carnitine-poor diets have little impact on the body's total carnitine content. Rather than being metabolized, excess carnitine is excreted in the urine as needed via the kidneys to maintain stable blood concentrations. /Carnitine/ L-Carnitine and acylcarnitine esters are present in all tissues. In most tissues and cells, they are present in higher concn than in the circulation ... L-carnitine and acetyl-L-carnitine are concn in most tissues via the high-affinity, Na+-dependent organic cation transporter OCTN2 ... OCTN2 binds acetyl-L-carnitine and propionyl-L-carnitine with comparable affinity. This protein is highly expressed in heart, placenta, skeletal muscle, kidney, pancreas, testis, and epididymis and weakly expressed in brain, lung, and liver ... /Acylcarnitine esters/ ... At a filtered load of 50 umol/L, the efficiency of L-carnitine and acylcarnitine ester reabsorption is 90 to 98% /in kidneys/. However, as the filtered load of L-carnitine incr, as, eg after consumption of a dietary supplement or after iv infusion, the efficiency of reabsorption declines rapidly ... Clearance of acylcarnitine esters is often higher than that of nonesterified L-carnitine /in kidneys/ ... Under conditions of rapid intracellular synth of acylcarnitine esters or direct accumulation from the circulation ... a higher proportion of acylcarnitine esters in urine compared to that in the circulation /is achieved/ ... Kidneys may be substantially involved in the regulation of circulating acylcarnitine ester concn. /Aacylcarnitine ester/ ... A concn ratio of acylcarnitine esters/nonesterified L-carnitine of 0.4 or greater in plasma is ... considered abnormal ... |
| Toxicity/Toxicokinetics |
Interactions Cyclosporine (CyA) is an immunosuppressive agent used after solid organ transplantation, but its clinical use is limited by side effects, the most important of which is nephrotoxicity. ... A previous work ... demonstrated that L-propionylcarnitine (L-PC), a propionyl ester of L-carnitine, is able to prevent CyA-induced acute nephrotoxicity reducing lipid peroxidation in the isolated and perfused rat kidney. CyA administration was associated with a dose dependent increase in renovascular resistance prevented by a pretreatment with L-PC. The aim of the present study was to confirm L-PC protective effect ... in an in vivo rat model. Chronic nephrotoxicity study was carried out for 28 days. L-PC was administered (ip 25 mg/kg bw) since the first day, while CyA treatment was performed for the last 21 days (by oral administration 25 mg/kg bw). ... L-PC was able to significantly lower blood pressure in CyA treated animals and to prevent CyA induced decrease in creatinine clearance. Moreover renal tissue analysis revealed that L-PC was able to reduce lipid hydroperoxide content and morphological abnormalities associated to chronic CyA administration. In conclusion our study demonstrated for the first time in vivo that L-PC protects against functional and tissue damage associated to chronic CyA administration. Nephrotoxicity has represented the major limitation in the use of cyclosporine A (CyA). The structural abnormalities at the level of the proximal tubular cells are necrosis, vacuolization and lipid droplets, as well as CyA-induced glomerular afferent arteriole constriction and granular juxtaglomerular cell hyperplasia. The mode of action of vasoconstriction is not well known, but there appears to be substantial impairment of endothelial cell function leading to enhanced release of vasoconstrictors such as endothelin and thromboxane. L-propionylcarnitine (PC), one of the most potent analogues of carnitine, is able to correct and to prevent alterations in endothelial membrane permeability and it has been identified in the kidney of various animal species. To investigate a possible reduction of CyA-induced nephrotoxocity ... the effects of a pretreatment with PC before administering several doses of CyA /were examined/ n an isolated and perfused rat kidney. The histological findings showed that the perfusion with PC reduces the vasoconstrictive effect of CyA on the glomerular capillaries and preserves the tubular epithelium. The ratio of the diameter between the glomerular capillary tuft and Bowman's capsule was higher, while at the tubular level the ratio internal-diameter/diameter evaluated at the level of the basal membrane was lower in PC + CyA perfused kidneys than in only CyA perfused ones. The final value of perfusion pressure was lower in PC + CyA perfused kidneys than in only CyA perfused ones, confirming the histological findings. The release induced by CyA of alanine aminopeptidase (AAP) and N-acetyl-glucosaminidase (NAG), markers of tubular damage, was significantly reduced by pretreatment with PC. These data suggest that the pretreatment with PC reduces the CyA-induced nephrotoxicity in an isolated and perfused rat kidney. The purpose of this study is to evaluate the ability of propionyl-L-carnitine, a carnitine derivative to prevent cardiac dysfunction induced by erucic acid and streptozotocin treatment in rats. Rats were fed for 10 days with normal or 10% erucic-acid-enriched diet with or without propionyl-L-carnitine injected intraperitoneally (1 mM/kg daily). Another group of rats was injected for 8-10 weeks with streptozotocin (65 mg/kg) with or without propionyl-L-carnitine intraperitoneally injected at the same dosage. Thereafter the animals were sacrificed and the hearts isolated and perfused aerobically. When isovolumic measurements of left ventricular function were applied, there was no difference in mechanical activity between treated and control hearts. On the contrary, when pressure-volume curves were determined in the paced hearts, the pressure developed by hearts from erucic acid-treated or diabetic rats was reduced. Propionyl-L-carnitine always produced positive inotropy. This was true for the control-saline treated rats that received the drug, as well as for the hearts isolated from cardiomyopathic animals. These data suggest that propionyl-L-carnitine, when given chronically, is able to overcome myocardial dysfunction caused either from erucic acid treatment or diabetes. In this study, the possible mechanisms whereby propionyl-l-carnitine (PLC) could protect against adriamycin (ADR)-induced cardiomyopathy were carried out. Administration of ADR (3 mg/kg) ip, every other day over a period of 2 weeks) resulted in a significant two-fold increase in serum levels of creatine phosphokinase, lactate dehydrogenase and glutamic oxaloacetic transaminase, whereas daily administration of PLC (250 mg/kg), ip for 2 weeks) induced non-significant change. Daily administration of PLC to ADR-treated rats resulted in complete reversal of ADR-induced increase in cardiac enzymes except lactate dehydrogenase which was only reversed by 66%. In cardiac tissue homogenate, ADR caused a significant 53% increase in malonedialdehyde (MDA) and a significant 50% decrease in reduced glutathione (GSH) levels, whereas PLC induced a significant 33% decrease in MDA and a significant 41% increase in GSH levels. Daily administration of PLC to ADR-treated rats completely reversed the increase in MDA and the decrease in GSH induced by ADR to the normal levels. In rat heart mitochondria isolated 24 h after the last dose, ADR induced a significant 48% and 42% decrease in(14)CO(2)released from the oxidation of [1-(14)C]palmitoyl-CoA and [1-(14)C]palmitoylcarnitine, respectively, whereas PLC resulted in a significant 66% and 54% increase in the oxidation of both substrates, respectively. Interestingly, administration of PLC to ADR-treated rats resulted in complete recovery of the ADR-induced decrease in the oxidation of both substrates. In addition, in rat heart mitochondria, the oxidation of [1-(14)C]pyruvate, [1-(14)C]pyruvate and [1-(14)C]octanoate were not affected by ADR and/or PLC treatment. Moreover, ADR caused severe histopathological lesions manifested as toxic myocarditis which is protected by PLC. Worth mentioning is that PLC had no effect on the antitumor activity of ADR in solid Ehrlich carcinoma. Results from this study suggest that: (1) in the heart, PLC therapy completely protects against ADR-induced inhibition of mitochondrial beta -oxidation of long-chain fatty acids; (2) PLC has and/or induces a powerful antioxidant defense mechanism against ADR-induced lipid peroxidation of cardiac membranes; and finally (3) PLC has no effect on the antitumor activity of ADR. For more Interactions (Complete) data for PROPIONYL-L-CARNITINE (8 total), please visit the HSDB record page. |
| Additional Infomation |
O-propanoylcarnitine is an O-acylcarnitine compound having propanoyl as the acyl substituent. It has a role as an analgesic, an antirheumatic drug, a cardiotonic drug, a peripheral nervous system drug and a human metabolite. It is functionally related to a propionic acid. Propionylcarnitine has been reported in Drosophila melanogaster, Homo sapiens, and other organisms with data available. Mechanism of Action L-Propionylcarnitine, a propionyl ester of L-carnitine, increases the intracellular pool of L-carnitine. It exhibits a high affinity for the enzyme carnitine acetyltransferase (CAT) and, thus, is readily converted into propionyl-coenzyme A and free carnitine. It has been reported that L-propionylcarnitine possesses a protective action against heart ischemia-reperfusion injury;... To obtain a better insight into the antiradical mechanism of L-propionylcarnitine, the present research analyzed the superoxide scavenging capacity of L-propionylcarnitine and its effect on linoleic acid peroxidation. In addition, the effect of L-propionylcarnitine against DNA cleavage was estimated using pBR322 plasmid. ... L-propionylcarnitine showed a dose-dependent free-radical scavenging activity. In fact, it was able to scavenge superoxide anion, to inhibit the lipoperoxidation of linoleic acid, and to protect pBR322 DNA from cleavage induced by H2O2 UV-photolysis. Therapeutic Uses L-Carnitine, acetyl-L-carnitine, and/or propionyl-L-carnitine may be used for replacement therapy to restore normal carnitine concn and/or a normal nonesterified-to-esterified carnitine ratio ... /EXPL THER/ The aim of this double-blind, placebo-controlled, dose titration, multicenter trial was to assess the efficacy and safety of propionyl-carnitine in intermittent claudication. ... After a 2-week preliminary period to assess maximal walking distance, 245 patients were randomly assigned to receive propionyl-L-carnitine (n = 118) or placebo (n = 127). The initial oral dose of 500 mg twice daily was increased at 2-month intervals to 2 g/day and then to 3 g/day in patients showing improvement in treadmill performance < 30% over baseline. Efficacy analysis was conducted for the 214 patients who completed the 24 weeks of treatment by comparing the effect of placebo and propionyl-L-carnitine on day 180. ... Analysis of variance showed a significant improvement of 73 +/- 9% (mean +/- SE) in maximal walking distance with propionyl-L-carnitine (n = 99) compared with 46 +/- 6% for placebo (n = 115, p = 0.03). For distance walked at onset of claudication, propionyl-L-carnitine showed about double the improvement of placebo; however, the difference was not statistically significant. There were no changes in electrocardiographic and routine biochemical and hematologic tests that would indicate an adverse effect of propionyl-L-carnitine. Adverse events requiring drug discontinuation (11 in the propionyl-L-carnitine group, 3 in the placebo group) were unrelated to study medication. The dose titration design of the study also provided information on the dose-response relation. Slightly less than 67% of patients were expected to improve their maximal walking distance by at least 30%, assuming 2 g/day of propionyl-L-carnitine (95% confidence interval 0.51 to 0.70). The response rate during the entire titration course was significantly in favor of propionyl-L-carnitine compared with placebo. ... /EXPL THER/ Propionyl-l-carnitine (PLC) is a naturally occurring compound that has been considered for the treatment of many forms of cardiomyopathies. /EXPL THER/ Propionyl-L-carnitine is a carnitine derivative that has a high affinity for muscular carnitine transferase, and it increases cellular carnitine content, thereby allowing free fatty acid transport into the mitochondria. ... The results of phase-2 studies in chronic heart failure patients showed that long-term oral treatment with propionyl-L-carnitine improves maximum exercise duration and maximum oxygen consumption over placebo and indicated a specific propionyl-L-carnitine effect on peripheral muscle metabolism. A multicenter trial on 537 patients showed that propionyl-L-carnitine improves exercise capacity in patients with heart failure, but preserved cardiac function. For more Therapeutic Uses (Complete) data for PROPIONYL-L-CARNITINE (13 total), please visit the HSDB record page. Drug Warnings Propionyl-L-carnitine stimulates a better efficiency of the Krebs cycle during hypoxia by providing it with a very easily usable substrate, propionate, which is rapidly transformed into succinate without energy consumption (anaplerotic pathway). Alone, propionate cannot be administered to patients in view of its toxicity. ... the efficacy and safety of oral vitamin E and propionyl-L-carnitine, separately or in combination, /were compared/ for the treatment of Peyronie's disease. ... A total of 236 men (mean age 43.4 years) with Peyronie's disease were randomly assigned to 4 groups. Group 1 (58 men) received 300 mg vitamin E orally twice daily. Group 2 (59) received 1 gm propionyl-L-carnitine orally twice daily, and group 3 (60) received 300 mg vitamin E and 1 gm propionyl-L-carnitine orally twice daily. Group 4 (control group, 59 men) received a similar regimen of placebo during the 6-month treatment period. . ... This study did not show significant improvement in pain, curvature or plaque size in patients with PD treated with vitamin E, propionyl-L-carnitine, or vitamin E plus propionyl-L-carnitine compared with those treated with placebo. Publication |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6028 mL | 23.0139 mL | 46.0278 mL | |
| 5 mM | 0.9206 mL | 4.6028 mL | 9.2056 mL | |
| 10 mM | 0.4603 mL | 2.3014 mL | 4.6028 mL |