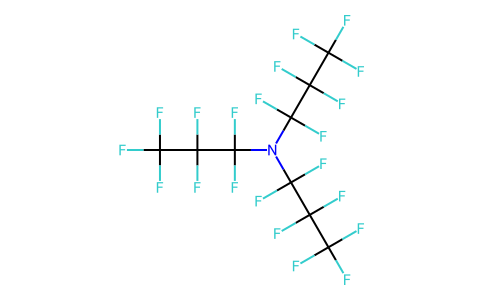
Physicochemical Properties
| Molecular Formula | C9NF21 |
| Molecular Weight | 521.0694 |
| Exact Mass | 520.969 |
| CAS # | 338-83-0 |
| PubChem CID | 67645 |
| Appearance | Colorless to light yellow liquid(Density:1.822 g/cm3) |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 130.0±35.0 °C at 760 mmHg |
| Flash Point | 32.4±25.9 °C |
| Vapour Pressure | 9.9±0.2 mmHg at 25°C |
| Index of Refraction | 1.269 |
| LogP | 15.38 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 31 |
| Complexity | 555 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | C(C(F)(F)F)(C(F)(F)N(C(C(C(F)(F)F)(F)F)(F)F)C(C(C(F)(F)F)(F)F)(F)F)(F)F |
| InChi Key | JAJLKEVKNDUJBG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C9F21N/c10-1(11,4(16,17)18)7(25,26)31(8(27,28)2(12,13)5(19,20)21)9(29,30)3(14,15)6(22,23)24 |
| Chemical Name | 1,1,2,2,3,3,3-heptafluoro-N,N-bis(1,1,2,2,3,3,3-heptafluoropropyl)propan-1-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The decline of the concentration of perfluorochemicals (PFC) after a single injection of three different doses was studied in the circulation of rats. The doses used amounted to 4.4, 10 and 14 g/kg body weight of Fluosol-DA, an emulsion of 7 parts of perfluorodecalin (FDC) and 3 parts of perfluorotripropylamine (FTPA). This also allowed testing of the composition of the emulsion remaining in the circulation and of that found in the liver. After two days a decrease of the half life from 34.0 +/- 0.7 to 17.1 +/- 4.3 h was found within the circulation at the highest dose. At the same time a change in the composition of the emulsion in the blood stream occurred, favouring the fraction of FTPA. FTPA increased from 28.3 +/- 1.4 to 54.4 +/- 8.1% on the fourth day. Whereas in the cells of the liver PFC droplets may be broken up, freed from their surfactant layer and handled according to their individual components, for PFC in the blood stream an unchanged composition should be assumed. Both results, the decreasing half life and the change in composition of the circulating emulsion may best be explained by a shrinking and instability of the emulgator film, showing the necessity for development of a superior surfactant. /Fluosol-DA/ The effect of an emulsion of perfluorochemicals (PFC) (7 parts perfluorodecalin and 3 parts perfluorotripropylamine, 4.4 g PFC/kg body weight) on organ function was determined. Whereas maximal storage of PFC was reached in the spleen as early as 12 h after PFC administration, the liver attained a maximal PFC content only after 2 days. Biological Half-Life Organ retention of the blood substitute component, perfluorotripropylamine (FTPA)... /was examined/. Various dosages of an emulsion of FTPA were administered to five rats. At intervals up to 86 weeks after infusion, (19F) MRI was used to measure the amount of FTPA in liver and spleen. The data were fit to both linear and exponential elimination models, and organ retention half-lives were calculated. The exponential half-lives for combined liver and spleen FTPA ranged from 110 to 190 days. Linear half-lives ranged from 175 to 300 days. /In anemic patients/ the half-life of /fluosol-DA/ was 24.3+-4.3 hr. |
| Additional Infomation |
Perfluorotripropylamine is an organofluorine compound. It has a role as a blood substitute. It is functionally related to a tripropylamine. Therapeutic Uses Blood substitute. /Exptl Ther/ Emulsions of perfluorocarbons (PFCs) have been tested as blood substitutes. /Exptl Ther/ Eight male rabbits were divided into the test (n=5) and control (n=3) groups. Each underwent intrajejunal, ip, and iv (artery, portal vein) catheter placements along with ligation of the duodenum and the terminal ileum under general anesthesia. The test group received oxygen-saturated perfluorotripropylamine (FTPA), and the control group received oxygen desaturated FTPA. The oxygen delivery was assessed by serial blood gas measurements before and after the admin of FTPA. The admin of oxygen-saturated FTPA significantly increased the partial pressure of oxygen within both the arterial and the portal venous blood (PaO2, PpVO2) without significant changes in PCO2 values. Oxygen desaturated FTPA failed to show any effects on blood gas values. Compared with oxygen desaturated FTPA, oxygen-saturated FTPA increased PaO2, PpVO2, and oxygen saturation (artery, portal vein) significantly at some, but not all of the time-points measured. The intraabdominal admin of saturated FTPA improved both the portal venous and the arterial oxygenation. This new mode of oxygenation may be helpful as an adjunct to conventional oxygen delivery systems. /Exptl Ther/ Perfluorodecalin and perfluorotripropylamine which have N2 solubility coefficients of 28.4 and 35.7 ml/dl, respectively, were used for treatment of decompression sickness in this study. Rats with chronically implanted venous catheters were held for 30 min at 800 kPa (7 bar, 8 ATA) by introducing compressed air into a chamber in which they were kept; a relatively short period of decompression followed (200 kPa/min). Immediately thereafter injections of the perfluorochemicals (PFCs) in a dose of 10 g/kg were given, controls received saline in the same volume or remained without treatment. An observation period of 2 h followed; after this time the incidence of death amongst the experimental animals (as compared with controls tested by the chi 2-test) showed that PFC treatment increased the likelihood of survival. Probit-log time relationship for the incidence of death also revealed a significant decrease in lethality in treated rats 30 min after the end of decompression. The mean lethal times Lt50 differed significantly, too. A still greater effect might be expected if the PFC emulsion were deprived of its normal nitrogen content by oxygenation before administration. Under the conditions of the present experiments PFCs produced an improvement in N2 exhalation at least in terms of the survival rate after compression followed by a very short decompression time. For more Therapeutic Uses (Complete) data for PERFLUAMINE (7 total), please visit the HSDB record page. Drug Warnings Emulsions of perfluorocarbons (PFCs) have been tested as blood substitutes. However, evidence exists that there is long-term retention of some PFCs by the organs of the reticuloendothelial system (RES). Since PFCs have a profound influence on several important neutrophil functions, patients receiving PFC should be monitored closely for possible infectious complications. It was concluded that the data in this select group of patients refusing blood products suggest that, after blood loss, ... /fluosol-DA/ is unnecessary in moderate anemia and ineffective in severe anemia. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9191 mL | 9.5956 mL | 19.1913 mL | |
| 5 mM | 0.3838 mL | 1.9191 mL | 3.8383 mL | |
| 10 mM | 0.1919 mL | 0.9596 mL | 1.9191 mL |