Physicochemical Properties
| Molecular Formula | C5H9NO4 |
| Molecular Weight | 147.1293 |
| Exact Mass | 147.053 |
| CAS # | 6893-26-1 |
| Related CAS # | D-Glutamic acid-d5;14341-88-9 |
| PubChem CID | 23327 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 333.8±32.0 °C at 760 mmHg |
| Melting Point | 200-202ºC |
| Flash Point | 155.7±25.1 °C |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.522 |
| LogP | -1.43 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 10 |
| Complexity | 145 |
| Defined Atom Stereocenter Count | 1 |
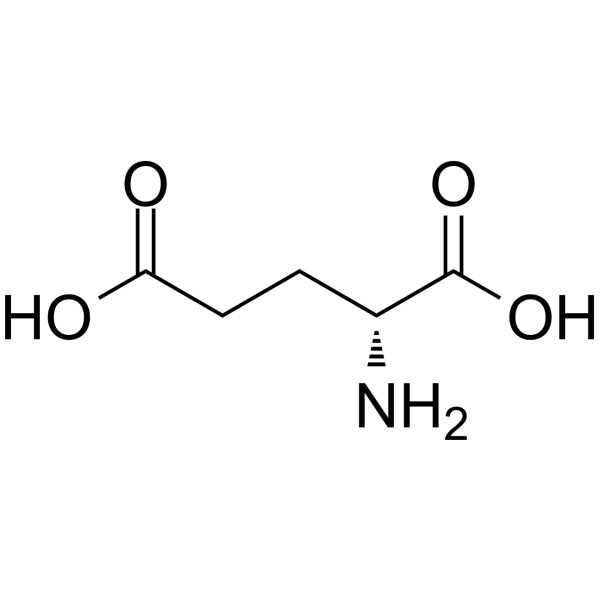
| SMILES | C(CC(=O)O)[C@H](C(=O)O)N |
| InChi Key | WHUUTDBJXJRKMK-GSVOUGTGSA-N |
| InChi Code | InChI=1S/C5H9NO4/c6-3(5(9)10)1-2-4(7)8/h3H,1-2,6H2,(H,7,8)(H,9,10)/t3-/m1/s1 |
| Chemical Name | (2R)-2-aminopentanedioic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Various d-amino acids, such as D-serine, D-aspartic acid (D-Asp) and D-glutamic acid (D-Glu), are commonly prevalent in animals including humans and are currently considered unique Physiologically active compounds and/or biomarkers [1]. D-[Asp/Glu] (4 mg/mL) suppresses the binding of IgE to peanut (75%), but D-Glu and D-Asp show no inhibitory effect. IgE is specific for D-[Asp/Glu] and may have the potential to eliminate IgE or diminish IgE binding to peanut allergen [2]. |
| ln Vivo | At the moment, D-glutamate is being studied as a regulator of hormone secretion and neuronal transmission. D-aspartate oxidase is the only enzyme that metabolizes it in mammals [1]. Following intraperitoneal administration, D-glutamate is transformed into n-pyrrolidone carboxylic acid, while L-glutamate is catabolized by α-ketoglutarate. In the cecum, the methyl carbon of acetate is created from the carbon 2 of D- and L-glutamic acid. The transformation of D-glutamic acid into n-pyrrolidone carboxylic acid is catalyzed by the kidney and liver of rats [3]. |
| References |
[1]. Changes in D-aspartic acid and D-glutamic acid levels in the tissues and physiological fluids of mice with various D-aspartate oxidase activities. J Pharm Biomed Anal. 2015 Dec 10;116:47-52. [2]. IgE binding to peanut allergens is inhibited by combined D-aspartic and D-glutamic acids. Food Chem. 2015 Jan 1;166:248-53. [3]. The metabolism of D- and L- glutamic acid in the rat. J Biol Chem. 1961 Feb;236:365-9. |
| Additional Infomation |
D-glutamic acid is an optically active form of glutamic acid having D-configuration. It has a role as an Escherichia coli metabolite and a mouse metabolite. It is a D-alpha-amino acid and a glutamic acid. It is a conjugate acid of a D-glutamate(1-). It is an enantiomer of a L-glutamic acid. Glutamic acid is the most common excitatory neurotransmitter in the central nervous system. D-Glutamic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). D-glutamic acid has been reported in Pisum sativum with data available. A non-essential amino acid naturally occurring in the L-form. Glutamic acid is the most common excitatory neurotransmitter in the CENTRAL NERVOUS SYSTEM. See also: Glutamic Acid (annotation moved to); Glutamic Acid Hydrochloride (annotation moved to). |
Solubility Data
| Solubility (In Vitro) |
H2O : ~13 mg/mL (~88.36 mM) DMSO :< 1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 10 mg/mL (67.97 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.7967 mL | 33.9836 mL | 67.9671 mL | |
| 5 mM | 1.3593 mL | 6.7967 mL | 13.5934 mL | |
| 10 mM | 0.6797 mL | 3.3984 mL | 6.7967 mL |