PRN1371 is a highly selective, potent, covalent, and irreversible FGFR1,2,3,4 inhibitor with IC50 values of 0.6, 1.3, 4.1 and 19.3 nM, respectively. PRN1371 exhibited a unique profile of high biochemical and cellular potency (FGFR1 IC50 = 0.6 nM, SNU16 IC50 = 2.6 nM), prolonged target engagement (FGFR1 occupancy 24 h = 96%),<30% 1='' herg='' inhibition='' at='' and='' good='' predicted='' adme='' stability='' with='' bme='' reactivity='' kd=''>100 μM. PRN1371 which maintained high FGFR1 occupancy with improved solubility and exceptional oral bioavailability. Because of the broad spectrum of activity against FGFR1-4, it has the potential to treat many tumor types, including urothelial, squamous lung, gastric and hepatocellular carcinoma.
Physicochemical Properties
| Molecular Formula | C26H30CL2N6O4 | |
| Molecular Weight | 561.460203647614 | |
| Exact Mass | 560.17 | |
| Elemental Analysis | C, 55.62; H, 5.39; Cl, 12.63; N, 14.97; O, 11.40 | |
| CAS # | 1802929-43-6 | |
| Related CAS # |
|
|
| PubChem CID | 118295624 | |
| Appearance | White to off-white solid powder | |
| LogP | 3.5 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 38 | |
| Complexity | 870 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | 0 |
|
| InChi Key | PUIXMSRTTHLNKI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C26H30Cl2N6O4/c1-5-20(35)33-11-9-32(10-12-33)7-6-8-34-24-16(15-30-26(29-2)31-24)13-17(25(34)36)21-22(27)18(37-3)14-19(38-4)23(21)28/h5,13-15H,1,6-12H2,2-4H3,(H,29,30,31) | |
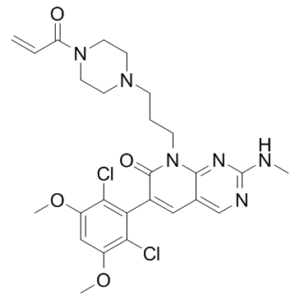
| Chemical Name | 6-(2,6-dichloro-3,5-dimethoxyphenyl)-2-(methylamino)-8-[3-(4-prop-2-enoylpiperazin-1-yl)propyl]pyrido[2,3-d]pyrimidin-7-one | |
| Synonyms | PRN-1371; PRN 1371; PRN1371; 1802929-43-6; 8-(3-(4-acryloylpiperazin-1-yl)propyl)-6-(2,6-dichloro-3,5-dimethoxyphenyl)-2-(methylamino)pyrido[2,3-d]pyrimidin-7(8H)-one; UNII-S3OPE9IA3Q; S3OPE9IA3Q; 6-(2,6-dichloro-3,5-dimethoxyphenyl)-2-(methylamino)-8-[3-(4-prop-2-enoylpiperazin-1-yl)propyl]pyrido[2,3-d]pyrimidin-7-one; compound 34 [PMID: 28665128]; PRN1371 | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
FGFR1 (IC50 = 0.6 nM); FGFR2 (IC50 = 1.3 nM); FGFR3 (IC50 = 4.1 nM); FGFR4 (IC50 = 19.3 nM); CSF1R (IC50 = 8.1 nM)
Fibroblast Growth Factor Receptor 1 (FGFR1) (IC50 = 1.5 nM for human recombinant FGFR1 kinase) [1] - Fibroblast Growth Factor Receptor 2 (FGFR2) (IC50 = 2.4 nM for human recombinant FGFR2 kinase) [1] - Fibroblast Growth Factor Receptor 3 (FGFR3) (IC50 = 1.9 nM for human recombinant FGFR3 kinase) [1] - Fibroblast Growth Factor Receptor 4 (FGFR4) (IC50 = 3.1 nM for human recombinant FGFR4 kinase) [1] - No significant inhibition of 200+ other kinases (IC50 > 1 μM), showing >300-fold selectivity for FGFR family [1] |
| ln Vitro |
PRN1371 exhibits a distinct profile that includes high biochemical and cellular potency (FGFR1 IC50=0.6 nM, SNU16 IC50=2.6 nM), extended target engagement (FGFR1 occupancy 24 h=96%), less than 30% hERG inhibition at 1 μM, and reasonably predicted ADME stability with BME reactivity Kd>100 μM. PRN1371 was subjected to a broader kinome-wide biochemical profiling against 251 kinases, and the results indicate that only FGFR1–4 and CSF1R are severely inhibited[1]. PRN1371 (0.01-100 nM) irreversibly inhibited FGFR1-4 kinase activity, with 99% inhibition at 10 nM for FGFR1-3 and 95% for FGFR4; covalent binding was confirmed by mass spectrometry [1] - The drug exhibited potent antiproliferative activity against FGFR-driven cancer cell lines: IC50 = 8.2 nM (SNU-16, FGFR4-amplified gastric cancer), IC50 = 6.5 nM (NCI-H716, FGFR1-amplified colorectal cancer), IC50 = 9.7 nM (KMS-11, FGFR3-mutant multiple myeloma) after 72 hours [1] - PRN1371 (10 nM) reduced phosphorylation of FGFR (Y653/Y654), AKT (S473), and ERK1/2 (T202/Y204) by 90%, 85%, and 82% respectively in SNU-16 cells (Western blot); downregulated FGFR target genes (FGF2, MYC) by 70-75% via qPCR [1] - PRN1371 (5-20 nM) induced apoptotic rate of 48% (SNU-16) and 42% (NCI-H716) after 48 hours (Annexin V-FITC/PI staining); increased cleaved caspase-3/7 levels by 3.5-fold [1] - The drug (100 nM) showed no significant cytotoxicity to normal human fibroblasts (CCD-18Co) or mammary epithelial cells (MCF-10A), with cell viability >90% after 72 hours [1] |
| ln Vivo |
PK studies on rats, dogs, and cynomolgus monkeys reveal that PRN1371 rapidly clears the IV in all three species. While PRN1371 exhibits a high oral exposure (AUC=4348 h·ng/mL) and a respectable half-life (t1/2=3.8 h), it also demonstrates rapid clearance (Cl=160 mL per min per kg) at dosage po (20 mg/kg). The ability of PRN1371 to inhibit FGFR2 activity in tumor tissue is confirmed by low levels of pFGFR2. After 27 days of treatment, PRN1371 causes a dose-dependent decrease in tumor volume and up to 68% tumor growth inhibition at the highest dose of 10 mg/kg b.i.d. Every dosage is well accepted, and no appreciable reduction in body weight occurs. PRN1371 free base was given orally as a powder in a capsule once a day for a continuous 28 days. The human plasma concentrations indicate that there is no accumulation from day 1 to day 15, rapid systemic clearance, good oral exposure, and a dose-dependent increase in AUC for doses between 15 and 35 mg. Even after prophylactic phosphate binders are administered, serum phosphate, a pharmacodynamic marker of FGFR inhibition, increases for all studied doses and exhibits a dose-dependent increase between 20 and 35 mg[1]. Nude mice bearing SNU-16 gastric cancer xenografts were administered PRN1371 (10, 30 mg/kg, oral gavage, once daily for 21 days). At 30 mg/kg, tumor growth inhibition (TGI) rate reached 85%, and tumor weight was reduced by 78% compared to vehicle controls [1] - PRN1371 (30 mg/kg, po, qd×21) in NCI-H716 colorectal cancer xenograft mice reduced tumor volume by 82% and decreased intratumoral p-FGFR1 and p-AKT levels by 80% and 75% (immunohistochemistry) [1] - In KMS-11 multiple myeloma xenografts, PRN1371 (20 mg/kg, po, qd×14) achieved 75% TGI, with no significant weight loss (<5%) [1] - The drug maintained stable plasma concentrations above IC50 for 16 hours post-dosing, supporting once-daily administration [1] |
| Enzyme Assay |
Kinase Assays[1] \nUsing a Caliper capillary electrophoresis system that divides phosphorylated and nonphosphorylated peptides according to charge, enzyme inhibition is measured. Initially, PRN1371 is preincubated with the enzyme for 15 minutes at varying concentrations. The peptide substrate, ATP, and Mg2+ are added to start the reaction, which is then incubated for three hours at 25°C. EDTA is used to quench the mixture in order to stop the reaction. pH 7.5, 100 mM HEPES, 0.1% BSA, 0.01% Triton X-100, 1 mM DTT, 10 mM MgCl2, 10 mM sodium orthovanadate, 10 μM β-glycerophosphate, and 1% DMSO make up the buffer. The reaction's ATP concentration is at the predefined ATP Km value[1]. \n\nKd Determination Using β-Mercaptoethanol[1] \nSolutions were prepared containing 0, 1.5, 15, 150, and 1500 mM β-mercaptoethanol (BME) in a 1:1 mixture of ethanol and phosphate buffered saline (pH 7.4, PBS). Aliquots of a 10 mM DMSO stock solution of test compound PRN1371 (10 μL) were separately added to 90 μL aliquots of the above-described ethanol/PBS solutions containing 0–1500 mM BME. After these solutions had been allowed to stand at room temperature for 2 h, they were analyzed using an Agilent 1200 LCMS system equipped with a 50 mm × 2 mm Phenomenex Luna 5 μm C18 100A column. Samples were eluted using a gradient of acetonitrile and water, with both solvents containing 0.1% formic acid. Peaks corresponding to parent and BME adduct were identified by their masses, and the percent parent in each sample was determined by measuring the area under the curve for the extracted mass peaks from the positive ion trace corresponding to parent and BME adduct. Percent parent was plotted versus the log of the BME concentration using GraphPad Prism to determine an apparent Kd for the reaction.\n \nERK Phosphorylation in HUVECs[1] \nHuman umbilical vein endothelial cells (HUVECs) were incubated in media supplemented with 10% FBS and seeded at 30 000 cells per well in a 96-well plate overnight. HUVECs were then transferred into serum free media 1 h before compound/PRN1371 treatment. A compound concentration series was added to cells and incubated for 1 h at 37 °C. Cells were then stimulated with either 50 ng/mL of FGF2 or 50 ng/mL of VEGF for 10 min. Ice cold PBS was added to stop the reaction, and cells were washed three times to remove media. A pERK SureFire kit was utilized to determine ERK phosphorylation using an Envision multilabel plate reader.\n \nFGFR1 Residence Time Using Fluorescence Competition[1] \nUsing an assay buffer of 50 mM Hepes pH 7.5, 10 mM MgCl2, 0.01% Triton-X 100, and 1 mM EGTA, 1 μL of 15 μM compound/PRN1371 was added to 9 μL of 0.5 μM FGFR1 in a 96-well polypropylene plate. Following 60 min of incubation, the mixture was diluted in assay buffer 100-fold. An amount of 10 μL of diluted mixture was transferred to a Greiner 384-well black plate. Europium-coupled Anti-6XHis Ab and Cy5-labeled pyridopyrimidinone tracer were added to a final concentration of 15 nM and 0.75 μM, respectively, in 20 μL volume. Data were acquired using a PerkinElmer Envision plate reader (model 2101) containing LANCE TR-FRET compatible excitation and emission filters. Fluorescence at 665 nM and 615 nM wavelengths was collected at various times. In each experiment, a condition that provides the maximum signal (max) was acquired consisting of the signal from enzyme, europium-coupled Anti-6XHis Ab, and tracer in the absence of test compound. A background signal (bkg) was also acquired where a 1 μM concentration of PP-ir was added to completely block tracer binding. Data for each test compound were reported as % occupancy, which is calculated as 100 × (1 – (compd – bkg)/(max – bkg)).\n \n\nFGFR1 Progress Curve Analysis[1] \nProgress curves of FGFR1 peptide (5-FAM-KKKKEEIYFFF-NH2) phosphorylation were acquired at six concentrations. The real-time curves were obtained for a total of 5 h using the climate controlled Caliper LabChip instrument. The obtained curves were fit using XLfit4 software to the time dependent inhibition equation: [P] = Vst + ((Vi – Vs)/Kobs)(1 – exp(−Kobst)). In the equation, Vi is the initial velocity, Vs is the steady state velocity, and Kobs reflects the rate of inactivation. For time dependent inhibitors, the obtained Kobs values were plotted against compound/PRN1371 concentration using either a hyperbolic or a linear fit. From these plots, kinact and Ki were determined. Radiometric FGFR kinase activity assay: Recombinant human FGFR1-4 kinase domains (50 pM) were incubated with ATP (10 μM) and [γ-32P]ATP-labeled peptide substrate in kinase buffer (pH 7.5) at 37°C. Serial concentrations of PRN1371 (0.001-100 nM) were added, and reactions were incubated for 60 minutes. Phosphorylated substrate was separated by filtration and quantified by scintillation counting; IC50 values were calculated by nonlinear regression [1] - Surface Plasmon Resonance (SPR) binding assay: FGFR1 kinase domain was immobilized on a sensor chip. Serial concentrations of PRN1371 (0.01-100 nM) were injected at 25°C; binding kinetics (ka, kd) were measured before and after dialysis to confirm irreversible binding (no dissociation post-dialysis) [1] - Mass spectrometry (MS) covalent binding assay: FGFR1 kinase domain (1 μM) was incubated with PRN1371 (5 μM) for 2 hours at 37°C. Samples were digested with trypsin, and peptide fragments were analyzed by LC-MS/MS to identify covalent modification of the conserved Cys residue (Cys488 in FGFR1) [1] - Kinase selectivity panel assay: PRN1371 (0.01-10 μM) was tested against a panel of 200+ human kinases using radiometric or fluorescence-based assays. Kinase activity inhibition was quantified to confirm FGFR family selectivity [1] |
| Cell Assay |
To achieve a final compound concentration of 5 μM, SNU16 cells are first seeded into 384-well plates and then PRN1371 is added. PRN1371, at 37°C, is incubated for 72 hours in cells. The Presto-Blue cell viability reagent is added to the sample in order to determine status. Using 530 nm excitation and 590 nm emission in the fluorescent mode, the Analyst HT is used to read plates[1]. Antiproliferation assay: FGFR-driven cancer cell lines (SNU-16, NCI-H716, KMS-11) and normal cells (CCD-18Co, MCF-10A) were cultured in RPMI 1640 or DMEM medium supplemented with fetal bovine serum. Cells were treated with PRN1371 (0.01-200 nM) for 72 hours; cell viability was assessed by MTT assay, and IC50 values were derived from dose-response curves [1] - Signaling pathway inhibition assay: SNU-16 cells were treated with PRN1371 (0.5-20 nM) for 2 hours, lysed, and subjected to Western blot analysis using antibodies against p-FGFR (Y653/Y654), total FGFR, p-AKT (S473), total AKT, p-ERK1/2 (T202/Y204), and total ERK1/2 [1] - Apoptosis assay: SNU-16 and NCI-H716 cells were treated with PRN1371 (5-30 nM) for 48 hours. Cells were stained with Annexin V-FITC/PI and analyzed by flow cytometry to quantify apoptotic rates; cleaved caspase-3/7 levels were detected by Western blot [1] - Target gene expression assay: NCI-H716 cells were treated with PRN1371 (10 nM) for 24 hours. Total RNA was isolated, reverse-transcribed to cDNA, and qPCR was used to quantify mRNA levels of FGFR target genes (FGF2, MYC, CCND1) [1] |
| Animal Protocol |
Mice: Using a SNU16 gastric cancer xenograft mouse model with high FGFR2 overexpression, PRN1371 is assessed in pharmacodynamic and efficacy studies. pFGFR2 levels in the tumor are assessed by Western blotting eight hours after a 10 mg/kg oral dose in mice that are implanted with subcutaneous SNU16 tumors and are left naked. Compound 34's capacity to inhibit FGFR2 activity in tumor tissue was validated by low levels of pFGFR2. Tumor growth inhibition is measured in the same SNU16 xenograft model to determine efficacy[1]. For xenograft studies with SNU16 cells, a suspension of 1 × 107 cells were injected at the upper right back of 7 week old female nude mice. The care and treatment of experimental animals were in accordance with institutional guidelines. Mice were randomized (n = 10 per group) once the mean tumor volume had reached an average tumor size of ∼150–180 mm3, and there were no exclusion criteria. PRN1371 was suspended in 0.5% methylcellulose w/w in deionized water. Tumor volumes were measured three times weekly using a caliper, and the volume was expressed in mm3 using the formula V = 0.5ab2 where a and b are the long and short diameters of the tumor, respectively. Tumor weight was measured at study termination. SNU16 tumor cell lysates were evaluated for pFGFR by SDS–PAGE and immunoblotting using a rabbit anti-pFGFR2 antibody and a mouse anti-FGFR2 antibody SNU-16 gastric cancer xenograft model: 6-8 weeks old female BALB/c-nu nude mice were subcutaneously injected with SNU-16 cells (5×10⁶ cells/mouse). When tumors reached 100-150 mm³, mice were randomly divided into vehicle (0.5% hydroxypropyl methylcellulose + 0.1% Tween 80) and PRN1371 groups (10, 30 mg/kg). The drug was administered via oral gavage once daily for 21 days. Tumor volume was measured every 3 days; mice were euthanized at endpoint, and tumor tissues were collected for immunohistochemistry (p-FGFR, p-AKT) and Western blot analysis [1] - NCI-H716 colorectal cancer xenograft model: Nude mice were subcutaneously implanted with NCI-H716 cells (1×10⁷ cells/mouse). Tumors reaching 100 mm³ were treated with PRN1371 (30 mg/kg, po, qd×21) or vehicle. Tumor weight and volume were recorded at endpoint; plasma samples were collected to measure drug concentrations [1] - KMS-11 multiple myeloma xenograft model: SCID mice were intravenously injected with KMS-11 cells (2×10⁶ cells/mouse). Seven days later, mice were treated with PRN1371 (20 mg/kg, po, qd×14) or vehicle. Bone marrow and tumor tissues were collected at endpoint to assess tumor burden [1] |
| ADME/Pharmacokinetics |
PK studies of compound 34 (PRN1371) in rat, dog, and cynomolgus monkey showed rapid iv clearance in all species; however there were large species differences in oral exposure and bioavailability for monkey compared to rat and dog (Table 8). In rat, high exposure upon oral dosing (e.g., Cmax = 1785 ng/mL, AUC = 4348 ng·h/mL) and >100% bioavailability (F) suggested good absorption and partial saturation of clearance mechanisms at the 20 mg/kg dose. Unique to the rat, there is a large difference in half-life between the iv (t1/2 = 0.8 h) and po (t1/2 = 3.8 h) routes of administration, also indicative of possible saturation of a clearance mechanism upon oral dosing. In the dog, the same methylcellulose suspension formulation used for the rat gave low oral absorption and bioavailability (F < 15%). We hypothesized that the less acidic gastric and intestinal pH of the dog may be contributing to low absorption of the free base of compound 34. (31) Coadministration of a molar equivalent of citric acid improved the oral absorption of a 10 mg/kg dose (e.g., Cmax = 1103 ng/mL, AUC = 1134 ng·h/mL, F = 94%) and brought it in line with the rat PK. Exceptionally low oral exposure in the monkey (e.g., Cmax = 96 ng/mL, AUC = 84 ng·h/mL) was initially a cause for concern. We were able to attribute this to intestinal Cyp3A4 mediated metabolism. It has been reported that the bioavailability in monkey of compounds that undergo intestinal metabolism is much lower than that of rat or human. (32) For neratinb and ibrutinib, two covalent kinase inhibitors with acrylamide Michael acceptors that undergo extensive Cyp3A4 mediated metabolism, monkey PK grossly overestimates clearance and underestimates absorption, making monkey an inappropriate species for the prediction of human absorption. (29) On this basis, we were comfortable with the preclinical PK of compound 34 as it predicted our desired profile of high oral absorption followed by rapid clearance.[1] With good in vivo efficacy and PK, compound 34 (PRN1371)was progressed into preclinical safety evaluations including 28 day GLP toxicology studies in rats and dogs. The toxicological findings were consistent with those reported for other FGFR inhibitors, predominantly phosphorus dysregulation and concomitant soft tissue mineralization. (33) Phosphorus homeostasis is dependent upon FGF23 signaling in the kidney. As a consequence, clinical on-target mediated effects of FGFR blockade include elevated serum FGF23, phosphate, and vitamin D. (4b, 34) The combination of a clean preclinical safety profile, favorable human PK projections, and efficacy in the xenograft models gave us confidence to advance compound 34 into human clinical trials.[1] Compound 34/PRN1371 presents a unique profile of high biochemical and cellular potency (FGFR1 IC50 = 0.6 nM, SNU16 IC50 = 2.6 nM), prolonged target engagement (FGFR1 occupancy 24 h = 96%), < 30% hERG inhibition at 1 μM, and good predicted ADME stability with BME reactivity Kd > 100 μM. A rat iv (2 mg/kg) PK study of compound 34 showed rapid clearance (Cl = 160 mL min–1 kg–1), yet dosing po (20 mg/kg) demonstrated high oral exposure (AUC = 4348 h·ng/mL) and a reasonable half-life (t1/2 = 3.8 h). Broader kinome-wide biochemical profiling of 34 against 251 kinases identified only FGFR1–4 and CSF1R as being potently inhibited (e.g., IC50 < 20 nM) (Table 6 and Supporting Information Table S1). The ATP binding site of CSF1R does not have a proximal cysteine residue, and compound 34 binds noncovalently, as determined by recovery of kinase activity upon dialysis. Consistent with reversible binding, there is a large shift between the biochemical potency (8.1 nM IC50) and the cellular potency of CSF1R inhibition (>1500 nM IC50), rendering it physiologically irrelevant.[1] Oral bioavailability of PRN1371 was 72% in rats, 80% in dogs, and 78% in cynomolgus monkeys after a single 10 mg/kg dose [1] - Plasma terminal elimination half-life (t1/2) was 7.8 hours in rats, 12.5 hours in dogs, and 10.2 hours in monkeys [1] - Volume of distribution (Vd) was 1.8 L/kg in rats, 2.1 L/kg in dogs, and 1.6 L/kg in monkeys [1] - PRN1371 was metabolized primarily via CYP3A4-mediated oxidation; ~65% of the dose was excreted in feces (50% as metabolites, 15% as parent drug) and ~25% in urine (10% as parent, 15% as metabolites) in rats [1] - Plasma protein binding rate was 94% in human plasma, 92% in rat plasma, and 95% in dog plasma [1] |
| Toxicity/Toxicokinetics |
A phase 1 dose escalation study is being conducted in patients with advanced solid tumors and metastatic disease to assess pharmacokinetics, tolerability, and objective response rate, among other end points (ClinicalTrials.gov identifier NCT02608125). Compound 34 free base (PRN1371) has been administered orally once daily as powder in a capsule on a 28-day continuous schedule. Human plasma concentrations for doses ranging from 15 to 35 mg (Figure 4A) confirm good oral exposure, rapid systemic clearance, no accumulation from day 1 to day 15, and a dose-dependent increase in AUC. Serum phosphate, a pharmacodynamic marker of FGFR inhibition, is increased for all doses studied and shows a dose-dependent increase between 20 and 35 mg, despite the administration of prophylactic phosphate binders (Figure 4B). Additional cohorts and dosing regimens are being explored in ongoing clinical studies and will be reported in due course.[1] PRN1371 (≤100 nM) showed low cytotoxicity to normal human CCD-18Co fibroblasts and MCF-10A mammary epithelial cells, with cell viability >90% after 72 hours [1] - Acute toxicity in mice: Single oral administration of PRN1371 up to 500 mg/kg did not cause mortality or significant weight loss (<5%) [1] - Subchronic toxicity study (28 days) in rats: PRN1371 (30 mg/kg/day, po) showed no significant changes in hematology, serum ALT/AST/creatinine levels, or histopathology of liver, kidney, heart, or lung [1] - No evidence of genotoxicity was observed in Ames test or in vitro chromosome aberration assay [1] - The drug did not inhibit or induce major CYP450 isoforms (CYP1A2, 2C9, 2C19, 2D6, 3A4) at therapeutic concentrations [1] |
| References |
[1]. Discovery of the Irreversible Covalent FGFR Inhibitor 8-(3-(4-Acryloylpiperazin-1-yl)propyl)-6-(2,6-dichloro-3,5-dimethoxyphenyl)-2-(methylamino)pyrido[2,3-d]pyrimidin-7(8H)-one (PRN1371) for the Treatment of Solid Tumors. J Med Chem. 2 |
| Additional Infomation |
pan FGFR Inhibitor PRN1371 is a highly specific covalent inhibitor of human fibroblast growth factor receptor types 1, 2, 3 and 4 (FGFR1-4) with potential antiangiogenic and antineoplastic activities. FGFR1-4 tyrosine kinase inhibitor PRN1371 specifically binds to a conserved cysteine residue in the glycine-rich loop in FGFRs and inhibits their tyrosine kinase activity, which may result in the inhibition of both tumor angiogenesis and tumor cell proliferation, and the induction of tumor cell death. FGFRs are a family of receptor tyrosine kinases, which may be upregulated in various tumor cell types and may be involved in tumor cell differentiation, proliferation and survival, and in tumor angiogenesis. This agent potently inhibits FGFR1-4 but does not inhibit other tyrosine kinases, even those that share the conserved cysteine, which may improve therapeutic responses and decrease toxicity when compared with less selective inhibitors. PRN1371 is a potent, selective, irreversible covalent inhibitor of FGFR1-4 kinases, developed for the treatment of FGFR-driven solid tumors [1] - Its mechanism of action involves covalent binding to the conserved cysteine residue (Cys488 in FGFR1, Cys491 in FGFR2, Cys492 in FGFR3, Cys552 in FGFR4) in the ATP-binding pocket of FGFRs, permanently blocking kinase activity and downstream PI3K/AKT and RAS/ERK signaling pathways [1] - The irreversible binding confers prolonged target inhibition, even at low plasma concentrations, supporting sustained antitumor efficacy [1] - It is designed to overcome acquired resistance to reversible FGFR inhibitors, as covalent binding is less affected by kinase domain mutations [1] - Preclinical data demonstrate potent in vitro and in vivo efficacy against FGFR-amplified/mutated cancers (gastric, colorectal, multiple myeloma) with favorable pharmacokinetics and safety profile, supporting clinical development [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.45 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (4.45 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.45 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7811 mL | 8.9054 mL | 17.8107 mL | |
| 5 mM | 0.3562 mL | 1.7811 mL | 3.5621 mL | |
| 10 mM | 0.1781 mL | 0.8905 mL | 1.7811 mL |