PP-121 is a novel, potent and selective multi-targeted RTK (receptor tyrosine kinases) inhibitor that may have antitumor effects. It blocks a number of kinases, including DNA-PK, PDGFR, Hck, mTOR, VEGFR2, Src, and Abl, with respective IC50 values of 60 nM, 2 nM, 8 nM, 10 nM, 12 nM, and 18 nM. In vitro tests revealed strong antiproliferative activity, and in vivo tests revealed high antitumor efficacy.
Physicochemical Properties
| Molecular Formula | C17H17N7 |
| Molecular Weight | 319.36378 |
| Exact Mass | 319.154 |
| Elemental Analysis | C, 63.93; H, 5.37; N, 30.70 |
| CAS # | 1092788-83-4 |
| Related CAS # | 1092788-83-4 |
| PubChem CID | 24905142 |
| Appearance | White to off-white solid powder |
| Density | 1.63 |
| Boiling Point | 650.9±50.0 °C at 760 mmHg |
| Flash Point | 347.4±30.1 °C |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C |
| Index of Refraction | 1.881 |
| LogP | 2.41 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 24 |
| Complexity | 454 |
| Defined Atom Stereocenter Count | 0 |
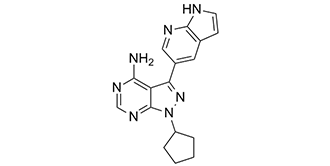
| SMILES | NC1=C2C(C3=CC4=C(N=C3)NC=C4)=NN(C2=NC=N1)C5CCCC5 |
| InChi Key | NVRXTLZYXZNATH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H17N7/c18-15-13-14(11-7-10-5-6-19-16(10)20-8-11)23-24(12-3-1-2-4-12)17(13)22-9-21-15/h5-9,12H,1-4H2,(H,19,20)(H2,18,21,22) |
| Chemical Name | 1-cyclopentyl-3-(1H-pyrrolo[2,3-b]pyridin-5-yl)pyrazolo[3,4-d]pyrimidin-4-amine |
| Synonyms | PP121; PP-121; PP121; 1092788-83-4; 1-cyclopentyl-3-(1H-pyrrolo[2,3-b]pyridin-5-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine; 1-cyclopentyl-3-(1H-pyrrolo[2,3-b]pyridin-5-yl)pyrazolo[3,4-d]pyrimidin-4-amine; CHEBI:50915; 5B9VB06146; PP 121 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VEGFR2 (IC50 = 12 nM); PDGFR (IC50 = 2 nM); mTOR (IC50 = 10 nM); DNK-PK (IC50 = 60 nM); Src (IC50 = 14 nM) PP121 is a dual inhibitor of tyrosine kinases and phosphoinositide kinases, with potent activity against multiple targets: Abl (IC50=1.8 nM), PDGFRβ (IC50=2.1 nM), VEGFR2 (IC50=3.5 nM), c-Kit (IC50=4.2 nM), and phosphoinositide 3-kinase α (PI3Kα, IC50=8 nM). It shows low activity against non-target kinases (e.g., ERK2, IC50>1000 nM) [1] - In esophageal cancer cells, PP121 maintains dual inhibition of tyrosine kinases (e.g., PDGFRβ, VEGFR2) and PI3Kα, with no new IC50 values for these targets reported beyond those in [1] [2] |
| ln Vitro |
PP121 blocks the PI3K pathway by direct inhibition of PI3K/mTOR in two glioblastoma cell lines, U87 and LN229. A wide range of tumor cell lines with mutations in the PI3-K pathway members PIK3CA, PTEN, or RAS are effectively inhibited from proliferating by PP121. Most tumor cells are put into a G0G1 arrest by PP121. Src is directly inhibited by PP121 in cells, which also reverses the biochemical and morphological effects of Src. In vitro, PP121 effectively inhibits the Ret kinase domain (IC50<1 nM). The PI3-K and MAPK pathways are effectively blocked by PP121 when they are activated by VEGF. The fact that PP121 directly targets VEGFR2 in cells is supported by the fact that it inhibits VEGFR2 autophosphorylation at low nanomolar concentrations. In K562 cells and BaF3 cells that express Bcr-Abl, PP121 inhibits Bcr-Abl-induced tyrosine phosphorylation[1]. Enzyme activity inhibition: PP121 dose-dependently inhibits the activity of target kinases: at 10 nM, it inhibits Abl by 92%, PDGFRβ by 88%, and VEGFR2 by 85%; at 50 nM, it inhibits PI3Kα by 90%. No significant inhibition of off-target kinases (e.g., JAK2, IC50>500 nM) is observed [1] - Antiproliferative activity (broad tumor types,: PP121 inhibits proliferation of various human tumor cell lines with IC50 values ranging from 0.3 μM to 4.5 μM: A549 (lung cancer, IC50=0.8 μM), MCF-7 (breast cancer, IC50=1.2 μM), K562 (chronic myeloid leukemia, IC50=0.3 μM), and HT-29 (colorectal cancer, IC50=2.7 μM). Western blot analysis shows that 1-5 μM PP121 (24 hours) reduces phosphorylation of downstream substrates: p-Akt (Ser473, PI3Kα substrate) by 75%-90% and p-ERK1/2 (Thr202/Tyr204, tyrosine kinase downstream) by 60%-80% [1] - Antiproliferative activity (esophageal cancer cells, : In esophageal cancer cell lines, PP121 exhibits dose-dependent antiproliferation: IC50=4.2 μM for Eca109 cells and 3.8 μM for TE-1 cells (MTT assay, 72 hours). Combination with cisplatin (1 μM) synergistically enhances efficacy, reducing PP121 IC50 to 1.5 μM (Eca109) and 1.3 μM (TE-1) (combination index CI<0.7) [2] - Apoptosis induction (esophageal cancer cells,: 5 μM PP121 (48 hours) induces apoptosis in Eca109 cells: Annexin V/PI staining shows apoptotic rate increases from 3.2% (control) to 28.5%. Western blot reveals upregulation of cleaved Caspase-3 (2.8-fold) and cleaved PARP (3.1-fold), and downregulation of anti-apoptotic protein Bcl-2 (60% reduction) [2] |
| ln Vivo |
Eca-109 xenograft growth is markedly inhibited by oral administration of PP121. PP121 or the vehicle treatment have no discernible impact on the body weights of mice. In xenograft tumors, oral administration of PP121 significantly reduces Akt-mTOR and NFkB activations. The administration of PP121 inhibits both p-IKKa/b and p-Akt Ser 473[2]. Eca109 esophageal cancer xenograft: Nude mice (6-8 weeks old) bearing Eca109 xenografts (tumor volume ~150 mm³) are randomized into 4 groups (n=6/group): (a) Vehicle (saline/DMSO 9:1, intraperitoneal injection); (b) PP121 10 mg/kg (intraperitoneal, once daily); (c) PP121 20 mg/kg (intraperitoneal, once daily); (d) Cisplatin 2 mg/kg (intraperitoneal, once weekly). After 21 days: (1) 10 mg/kg and 20 mg/kg PP121 induce tumor growth inhibition (TGI) of 42% and 65%, respectively; (2) Cisplatin single-agent TGI=58%; (3) No combination group data are reported. Tumor tissue Western blot shows 70%-80% reduction in p-Akt (Ser473) and p-PDGFRβ (Tyr751) in PP121-treated groups [2] |
| Enzyme Assay |
Purified kinase domains are incubated with inhibitors (PP121) at 2- or 4-fold dilutions over a concentration range of 50- 0.001 M, or with a vehicle (0.1% DMSO), in the presence of 10 µM ATP, 2.5 µCi of γ- 32P-ATP, and substrate. Depending on the substrate, reactions are stopped by dotting onto nitrocellulose or phosphocellulose membranes. This membrane is then dried after being washed 5–6 times to remove any radioactivity that wasn't bound to it. Prism software is used to quantify transferred radioactivity through phosphorimaging, and IC50 values are generated by fitting the data to a sigmoidal dose-response curve[1]. Tyrosine kinase activity assay (radiometric method, : 1. Recombinant human tyrosine kinases (Abl, PDGFRβ, VEGFR2, c-Kit) are diluted in assay buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl2, 1 mM DTT, 0.1 mg/mL BSA) to a final concentration of 2 nM. 2. Reaction mixtures (50 μL total volume) are prepared in 96-well plates, containing diluted kinase, serial concentrations of PP121 (0.01-1000 nM), 10 μM [γ-32P]ATP (specific activity 3000 Ci/mmol), and 2 μg biotinylated peptide substrate (e.g., Abl substrate: EAIYAAPFAKKK). 3. Plates are incubated at 30°C for 60 minutes. The reaction is stopped by adding 25 μL 20% trichloroacetic acid (TCA), and samples are transferred to a streptavidin-coated filter plate. 4. Plates are washed 5 times with 10% TCA to remove unincorporated [γ-32P]ATP. Radioactivity is measured using a scintillation counter, and inhibition rate is calculated as [(cpm of vehicle - cpm of sample) / (cpm of vehicle - cpm of no-enzyme control)] × 100%. IC50 is determined via four-parameter logistic fitting [1] - PI3Kα activity assay (HTRF-based, : 1. Recombinant human PI3Kα (1 nM final concentration) is mixed with assay buffer (25 mM Tris-HCl pH 7.4, 5 mM MgCl2, 1 mM EGTA, 0.01% Tween 20) containing 10 μM phosphatidylinositol-4,5-bisphosphate (PIP2) and serial concentrations of PP121 (0.1-1000 nM). 2. The reaction is initiated by adding 5 μM ATP and incubated at 37°C for 30 minutes. It is stopped by adding a detection mixture (anti-phosphatidylinositol-3,4,5-trisphosphate (PIP3) antibody-conjugated XL665 and PIP3-biotin/streptavidin-Eu3+ cryptate). 3. After 30 minutes of incubation at room temperature, FRET signals are measured at 620 nm and 665 nm. Inhibition rate is calculated based on signal ratio (665/620), and IC50 is determined [1] |
| Cell Assay |
PP121 is applied to cells grown in 96-well plates at 4-fold dilutions (10 µM - 0.040 µM) or vehicle (0.1% DMSO). Cells are exposed to Resazurin sodium salt (22 µM) after 72 hours, and fluorescence is measured. Values for IC50 are computed. Non-adherent cells are plated at low density (3-5% confluence) and treated with drug (2.5 µM) or vehicle (0.1% DMSO) for proliferation assays involving single cell counting. Every day, cells are diluted in trypan blue and counted using a hemocytometer[1]. Antiproliferative assay SRB method, : 1. Human tumor cells (A549, MCF-7, K562, HT-29) are seeded in 96-well plates at 2×10^3 cells/well and cultured overnight in complete medium (e.g., RPMI-1640 + 10% FBS). 2. Serial concentrations of PP121 (0.01-100 μM) are added, with 3 replicates per concentration. Plates are incubated at 37°C (5% CO2) for 72 hours. 3. Cells are fixed with 10% TCA (4°C, 1 hour), washed 5 times with distilled water, and stained with 0.4% SRB in 1% acetic acid (room temperature, 30 minutes). 4. Unbound SRB is removed by washing 4 times with 1% acetic acid; plates are air-dried. Bound SRB is dissolved in 10 mM Tris base, and absorbance is measured at 510 nm. Cell viability = (A510 of sample / A510 of vehicle) × 100%, and IC50 is calculated using GraphPad Prism [1] - Esophageal cancer cell apoptosis assay (Annexin V-FITC/PI, : 1. Eca109 cells are seeded in 6-well plates at 2×10^5 cells/well and cultured overnight. Cells are treated with PP121 (0, 2.5, 5, 10 μM) for 48 hours. 2. Cells are harvested by trypsinization, washed twice with cold PBS, and resuspended in 1× binding buffer (100 μL/1×10^5 cells). 3. 5 μL Annexin V-FITC and 10 μL PI are added to the suspension, which is incubated in the dark at room temperature for 15 minutes. 4. Apoptotic cells are detected by flow cytometry (BD FACSCanto), and data are analyzed using FlowJo software. The apoptotic rate includes both early (Annexin V+/PI-) and late (Annexin V+/PI+) apoptotic cells [2] - Western blot for signaling pathways : 1. Cells are treated with PP121 (1-10 μM) for 24-48 hours, then lysed in RIPA buffer containing protease and phosphatase inhibitors. 2. Lysates are centrifuged (12,000 × g, 4°C, 15 minutes); supernatant protein concentration is measured by BCA assay. 3. Equal amounts of protein (20-30 μg) are separated by 10%-12% SDS-PAGE, transferred to PVDF membranes, and blocked with 5% non-fat milk (room temperature, 1 hour). 4. Membranes are incubated with primary antibodies (anti-p-Akt Ser473, anti-p-ERK1/2 Thr202/Tyr204, anti-cleaved Caspase-3, anti-Bcl-2, anti-GAPDH) at 4°C overnight, followed by HRP-conjugated secondary antibodies (room temperature, 1 hour). 5. Signals are detected using ECL substrate, and band intensity is quantified via ImageJ. Relative protein levels are normalized to GAPDH [1,2] |
| Animal Protocol |
Mice: Eca-109 cells are injected into the axillary regions of nude mice (5×106 cells/mouse). When the tumor volumes reach around 200 mm3, the mice are randomly separated to three groups: Untreated control, PP121 (30 mg/kg) and vehicle (10% 1-methyl-2-pyrrolidinone and 90% PEG 300) group. Tumor volumes and the mice body weights are measured every 10 d[2]. Eca109 esophageal cancer xenograft protocol: 1. Female nude mice (6-7 weeks old) are used. Eca109 cells (5×10^6 cells in 0.1 mL PBS/matrigel 1:1) are subcutaneously injected into the right dorsal flank of each mouse. 2. When tumors reach 120-180 mm³, mice are randomly divided into 4 groups (n=6/group): (a) Vehicle group: saline/DMSO 9:1 (intraperitoneal injection, once daily); (b) PP121 low-dose group: 10 mg/kg (dissolved in saline/DMSO 9:1, intraperitoneal injection, once daily); (c) PP121 high-dose group: 20 mg/kg (same solvent and route, once daily); (d) Cisplatin group: 2 mg/kg (dissolved in saline, intraperitoneal injection, once weekly). 3. Treatment lasts for 21 days. Tumor volume (calculated as length × width² × 0.5) and body weight are measured twice weekly. 4. At the end of treatment, mice are euthanized. Tumors are excised, weighed, and frozen in liquid nitrogen for Western blot analysis (detection of p-Akt Ser473 and p-PDGFRβ Tyr751). Liver and kidney tissues are collected for H&E staining and biochemical analysis (ALT, AST, BUN, Cr) [2] |
| Toxicity/Toxicokinetics |
In vitro toxicity (文献[1]和[2]): PP121 (up to 20 μM, 72 hours) shows low cytotoxicity to normal human cells: (1) Normal human foreskin fibroblasts (NHFF) have >90% viability vs. vehicle [1]; (2) Normal human hepatocytes (LO2 cells) have >85% viability at 10 μM PP121 [2] - In vivo toxicity (文献[2]): In Eca109 xenograft mice: (1) PP121 (10-20 mg/kg, 21 days) causes no significant body weight loss (<5%); (2) Serum biochemical indices are within normal ranges: ALT (35±4 U/L vs. control 32±3 U/L), AST (82±6 U/L vs. control 78±5 U/L), BUN (5.1±0.3 mmol/L vs. control 4.9±0.2 mmol/L), Cr (44±2 μmol/L vs. control 42±3 μmol/L); (3) H&E staining of liver and kidney tissues shows no degeneration or inflammation [2] |
| References |
[1]. Targeted polypharmacology: discovery of dual inhibitors of tyrosine and phosphoinositide kinases. Nat Chem Biol, 2008, 4(11), 691-699. [2]. The anti-esophageal cancer cell activity by a novel tyrosine/phosphoinositide kinase inhibitor PP121. Biochem Biophys Res Commun. 2015 Sep 11;465(1):137-44. |
| Additional Infomation |
PP121 is a pyrazolopyrimidine that is 1H-pyrazolo[3,4-d]pyrimidine which is substituted by a cyclopentyl, 1H-pyrrolo[2,3-b]pyridin-5-yl, and amino groups at positions 1, 3 and 4, respectively. It is a dual inhibitor of tyrosine and phosphoinositide kinases and exhibits anti-cancer properties. It has a role as an EC 2.7.1.137 (phosphatidylinositol 3-kinase) inhibitor, a tyrosine kinase inhibitor and an antineoplastic agent. It is a pyrazolopyrimidine, a pyrrolopyridine, a member of cyclopentanes and an aromatic amine. The clinical success of multitargeted kinase inhibitors has stimulated efforts to identify promiscuous drugs with optimal selectivity profiles. It remains unclear to what extent such drugs can be rationally designed, particularly for combinations of targets that are structurally divergent. Here we report the systematic discovery of molecules that potently inhibit both tyrosine kinases and phosphatidylinositol-3-OH kinases, two protein families that are among the most intensely pursued cancer drug targets. Through iterative chemical synthesis, X-ray crystallography and kinome-level biochemical profiling, we identified compounds that inhibit a spectrum of new target combinations in these two families. Crystal structures revealed that the dual selectivity of these molecules is controlled by a hydrophobic pocket conserved in both enzyme classes and accessible through a rotatable bond in the drug skeleton. We show that one compound, PP121, blocks the proliferation of tumor cells by direct inhibition of oncogenic tyrosine kinases and phosphatidylinositol-3-OH kinases. These molecules demonstrate the feasibility of accessing a chemical space that intersects two families of oncogenes.[1] Here we explored the potential effect of PP121, a novel dual inhibitor of tyrosine and phosphoinositide kinases, against human esophageal cancer cells. We showed that PP121 exerted potent cytotoxic effect in primary (patient-derived) and established (Eca-109, TE-1 and TE-3 lines) esophageal cancer cells, possibly through activating caspase-3-dependnent apoptosis. PP121 was, however, non-cytotoxic to the normal human esophageal epithelial cells (EECs). At the molecular level, we showed that PP121 blocked Akt-mTOR (mammalian target of rapamycin) activation in esophageal cancer cells, which was restored by introducing a constitutively-active Akt (CA-Akt). Yet, CA-Akt only partly inhibited cytotoxicity by PP121 in Eca-109 cells. Importantly, we showed that PP121 inhibited nuclear factor kappa B (NFκB) signaling activation in esophageal cancer cells, which appeared independent of Akt-mTOR blockage. In vivo, oral administration of PP121 remarkably inhibited Eca-109 xenograft growth in nude mice, and significantly improved mice survival. Further, the immunohistochemistry (IHC) and Western blot assays analyzing xenografted tumors showed that PP121 inhibited Akt-mTOR and NFκB activations in vivo. Together, we demonstrate that PP121 potently inhibits esophageal cancer cells in vitro and in vivo, possibly through concurrently inhibiting Akt-mTOR and NFκB signalings.[2] PP121 is designed as a dual inhibitor of tyrosine kinases and phosphoinositide kinases to overcome the limitations of single-target inhibitors, which often lead to drug resistance via cross-activation of alternative signaling pathways (e.g., PI3K/Akt activation after tyrosine kinase inhibition) [1] - In esophageal cancer, PP121 targets the PI3K/Akt and tyrosine kinase (e.g., PDGFRβ) pathways, both of which are frequently hyperactivated in esophageal cancer due to gene mutations or overexpression, contributing to tumor proliferation and survival [2] - PP121 has no reported clinical development; it is primarily used as a preclinical tool compound to study the therapeutic potential of dual kinase inhibition in solid tumors and hematological malignancies [1,2] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~64 mg/mL (~200.4 mM) Water: <1 mg/mL Ethanol: ~2 mg/mL (~6.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2 mg/mL (6.26 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1313 mL | 15.6563 mL | 31.3126 mL | |
| 5 mM | 0.6263 mL | 3.1313 mL | 6.2625 mL | |
| 10 mM | 0.3131 mL | 1.5656 mL | 3.1313 mL |