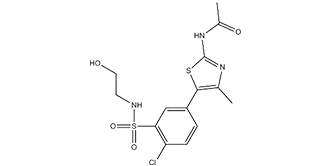
PIK-93, a phenylthiazole analog, is a novel, potent, and selective PI4K (PI4KIIIβ) inhibitor with antiviral effects (antienterovirus activity). It inhibits PI4K (PI4KIIIβ) with an IC50 of 19 nM and PI3Kα with an IC50 of 39 NM.
Physicochemical Properties
| Molecular Formula | C14H16N3O4S2CL |
| Molecular Weight | 389.87754 |
| Exact Mass | 389.027 |
| Elemental Analysis | C, 43.13; H, 4.14; Cl, 9.09; N, 10.78; O, 16.41; S, 16.45 |
| CAS # | 593960-11-3 |
| Related CAS # | 593960-11-3; |
| PubChem CID | 6852167 |
| Appearance | White to off-white solid powder |
| Density | 1.48 g/cm3 |
| Boiling Point | 611.8ºC at 760 mmHg |
| Flash Point | 323.8ºC |
| LogP | 4.122 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 24 |
| Complexity | 543 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC(NC1=NC(C)=C(S1)C2=CC=C(C(S(=O)(NCCO)=O)=C2)Cl)=O |
| InChi Key | JFVNFXCESCXMBC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H16ClN3O4S2/c1-8-13(23-14(17-8)18-9(2)20)10-3-4-11(15)12(7-10)24(21,22)16-5-6-19/h3-4,7,16,19H,5-6H2,1-2H3,(H,17,18,20) |
| Chemical Name | N-(5-(4-chloro-3-(N-(2-hydroxyethyl)sulfamoyl)phenyl)-4-methylthiazol-2-yl)acetamide |
| Synonyms | PIK-93; PIK93; PIK 93 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PI4KIIIβ (IC50 = 19 nM); PI4KIIIα (IC50 = 1.1 μM); p110γ (IC50 = 16 nM); p110α (IC50 = 39 nM); p110δ (IC50 = 120 nM); p110β (IC50 = 590 nM); PI3KC2β (IC50 = 140 nM); PI3KC2α (IC50 = 16 μM); hsVPS34 (IC50 = 320 nM); DNA-PK (IC50 = 64 nM); ATM (IC50 = 490 nM); mTORC1 (IC50 = 1.38 μM); ATR (IC50 = 17 μM) |
| ln Vitro | PIK-93 inhibits PI3Kγ and PI4KIIIβ, with IC50 values of 16 nM and 19 nM, respectively. PIK-93 also inhibits other members of PI3Ks, including PI3Kα, β, and δ, with IC50 values of 39 nM, 0.59 μM, and 0.12 μM, respectively. Even at a concentration of 10 M, PIK-93 has no discernible inhibitory effect on a panel of other kinases[1]. PIK-93 (0.5 M-1 M) decreases the consolidation and stability of the leading edge formed after treatment with uniform f-Met-Leu-Phe (fMLP) in differentiated HL60 (dHL60) cells. The fMLP-dependent accumulation of total F-actin is altered by PIK-93, but not in terms of quantity. PIK-93 increases cell turning frequency while decreasing the chemotactic index in fMLP gradients[2]. In COS-7 cells, PIK-93 (250 nM) successfully prevents the buildup of FL-Cer and the CERT-PH domain in the Golgi. The same concentration of PIK-93 also significantly reduces the ability of endogenous ceramide to transform into sphingomyelin when labeled with [3H]serine. These facts suggest that PI4KIIIβ plays a critical role in the regulation of spingomyelin synthesis as well as ceramide transport between the ER and Golgi[3]. PIK-93 (300 nM) inhibits net Ca2+ entry and carbachol-induced translocation of TRPC6 to the plasma membrane in T6.11 cells[4]. According to a recent study, PIK-93 has anti-enterovirus properties because it inhibits the replication of the poliovirus (PV) and the hepatitis C virus (HCV), with EC50 values of 0.14 µM and 1.9 µM, respectively[5]. |
| ln Vivo | N/A |
| Enzyme Assay | Standard TLC lipid kinase activity assays are used to calculate IC50 values. In order to carry out kinase reactions, a reaction mixture containing kinase, PIK-93 (2% DMSO final concentration), buffer (25 mM HEPES, pH 7.4, 10 mM MgCl2), and freshly sonicated phosphatidylinositol (100 µg/mL) must be prepared. A final concentration of 10 or 100 µM is used to start the reactions, and they are then allowed to continue for 20 min at room temperature. The starting material for the reactions is ATP containing 10 µCi of γ-32P-ATP. The addition of 105 µL 1N HCl and 160 l of CHCl3:MeOH (1:1) then ends the reactions in preparation for TLC analysis. The organic phase is transferred to a new tube using a gel loading pipette tip that has been CHCl3-precoated after the biphasic mixture has been vortexed and briefly centrifuged. In a 65:35 solution of n-propanol:1M acetic acid, this extract is spotted on TLC plates and developed for 3–4 hours. Following drying, the TLC plates are shown to a phosphorimager screen and quantitated. As a two-fold dilution from the highest concentration of 100 μM, kinase activity is typically assessed at 10–12 concentrations of PIK–93. |
| Cell Assay | For actin staining, dHL60 cells are preincubated in suspension with PIK-93 or vehicle for 40 min, centrifuged for 5 min at 2000 rpm at room temperature in a J6-B centrifuge, resuspended in mHBSS containing the appropriate agent at the same concentration, and then allowed to adhere to fibronectin-covered coverslips for 3 min. Rhodamine-phalloidin (10 units/mL) is used to stain cells that have been fixed in 3.7% PFA for 15 minutes. |
| Animal Protocol | N/A; |
| References |
[1]. A pharmacological map of the PI3-K family defines a role for p110alpha in insulin signaling. Cell. 2006 May 19;125(4):733-47 [2]. To stabilize neutrophil polarity, PIP3 and Cdc42 augment RhoA activity at the back as well as signals at the front. J Cell Biol. 2006 Jul 31;174(3):437-45 [3]. Phosphatidylinositol 4-kinase IIIbeta regulates the transport of ceramide between the endoplasmic reticulum and Golgi. J Biol Chem. 2006 Nov 24;281(47):36369-77 [4]. Involvement of phosphoinositide 3-kinase and PTEN protein in mechanism of activation of TRPC6 protein in vascular smooth muscle cells. J Biol Chem. 2012 May 18;287(21):17672-81 [5]. Phosphatidylinositol 4-kinase III beta is a target of enviroxime-like compounds for antipoliovirus activity. J Virol. 2011 Mar;85(5):2364-72 |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~78 mg/mL (200.1 mM) Water: <1 mg/mL Ethanol: <1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 4.55 mg/mL (11.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 45.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 30% PEG400+0.5% Tween80+5%Propylene glycol:30mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5649 mL | 12.8245 mL | 25.6489 mL | |
| 5 mM | 0.5130 mL | 2.5649 mL | 5.1298 mL | |
| 10 mM | 0.2565 mL | 1.2824 mL | 2.5649 mL |