Physicochemical Properties
| Molecular Formula | C18H23NO |
| Molecular Weight | 269.38 |
| CAS # | 83-98-7 |
| Related CAS # | Orphenadrine citrate;4682-36-4;Orphenadrine hydrochloride |
| PubChem CID | 4601 |
| Appearance | Light yellow to brown <25°C solid powder,>25°C liquid |
| Density | 1.0278 (rough estimate) |
| Boiling Point | 195ºC at 12 ATM |
| Melting Point | 25°C |
| Index of Refraction | 1.5740 (estimate) |
| LogP | 3.663 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 20 |
| Complexity | 260 |
| Defined Atom Stereocenter Count | 0 |
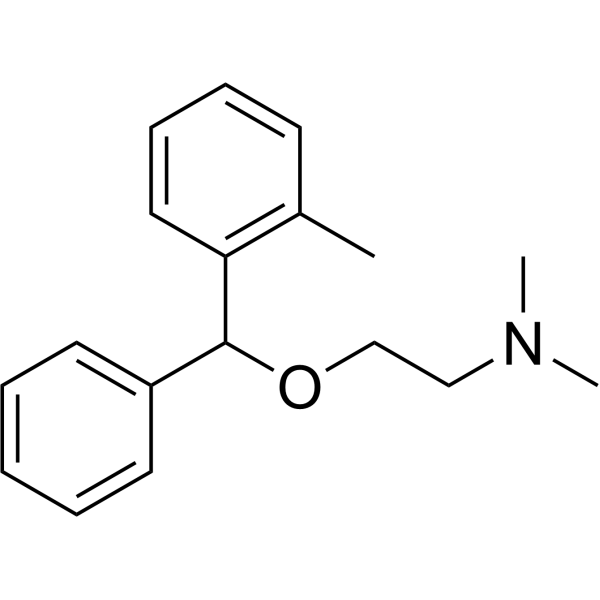
| SMILES | CC1=CC=CC=C1C(C2=CC=CC=C2)OCCN(C)C |
| Synonyms | (±)-Orphenadrine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | NMDA receptor[1]; CYP450 2B[2]; Cholinesterase (ChE)[3] |
| ln Vitro | Orphenadrine (30-300 μM) exhibits relatively fast concentration-dependent open channel blockade kinetics with a Koff of 0.013[1]. |
| ln Vivo | In a study of the tumor-promoting effects of orphenadrine, male rats were pretreated with a single intraperitoneal injection of N-diethylnitrosamine (DEN) for 2 weeks. Orphenadrine (0, 750, 1500 ppm; po; 6 wk) accelerated hepatocyte proliferation and induced liver tumor-promoting activity[2]. Orphenadrine (30 mg/kg; po; 3 d) protected rats from the effects of 3-nitropropionic acid (3-NPA) (30 mg/kg; 3 d), which resulted in increased expression of neuronal damage markers [(3)H]-PK 11195 and HSP27 in astrocytes[3]. |
| Cell Assay |
Cell Viability Assay[1] Cell Types: NMDA open-channel Tested Tested Concentrations: 30, 100 and 300 μM Incubation Duration: 5 seconds; with 200 μM NMDA Experimental Results: Nearly completely inhibited [3H]MK-801 binding at 100 μM. Exhibited relatively fast, Tested Tested Concentrations-dependent open channel blocking kinetics. |
| Animal Protocol |
Animal/Disease Models: Liver tumor model in male rats pre-treated by N-diethylnitrosamine[2] Doses: 0, 750, 1500 ppm Route of Administration: PO; for 6 weeks Experimental Results: Increased mRNA expression levels of Cyp2b1/2, Mrp2 and Cyclin D1. Increased microsomal reactive oxygen species (ROS) production and oxidative stress markers such as thiobarbituric acid-reactive substances and 8-hydroxydeoxyguanosine. |
| References |
[1]. Orphenadrine is an uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist: binding and patch clamp studies. J Neural Transm Gen Sect. 1995;102(3):237-46. [2]. Liver tumor promoting effect of orphenadrine in rats and its possible mechanism of action including CAR activation and oxidative stress. J Toxicol Sci. 2013;38(3):403-13. [3]. Orphenadrine prevents 3-nitropropionic acid-induced neurotoxicity in vitro and in vivo. Br J Pharmacol. 2001 Feb;132(3):693-702. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~371.22 mM; with ultrasonication) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.28 mM)(saturation unknown) in 10% DMSO 40% PEG300 5% Tween-80 45% Saline (add these co-solvents sequentially from left to right, and one by one),clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution and add it to 400 μL PEG300, mix well; then add 50 μL Tween-80 to the above system, mix well; then continue to add 450 μL of normal saline to make up to 1 mL. Preparation of normal saline: Dissolve 0.9 g of sodium chloride in ddH₂O and make up to 100 mL to obtain a clear and transparent normal saline solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.28 mM)(saturation unknown) in 10% DMSO 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one),clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution and add it to 900 μL of 20% SBE-β-CD saline solution and mix well. 2 g SBE-β-CD (sulfobutyl ether β-cyclodextrin) powder is diluted to 10 mL of saline and completely dissolved until clear and transparent. Solubility in Formulation 3: ≥ 2.5 mg/mL (9.28 mM)(saturation unknown) in 10% DMSO 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one),clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution and add it to 900 μL corn oil and mix well. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.7122 mL | 18.5611 mL | 37.1223 mL | |
| 5 mM | 0.7424 mL | 3.7122 mL | 7.4245 mL | |
| 10 mM | 0.3712 mL | 1.8561 mL | 3.7122 mL |