Physicochemical Properties
| Molecular Formula | C20H17CLN4O4 |
| Molecular Weight | 412.83 |
| Exact Mass | 412.093 |
| CAS # | 417717-04-5 |
| Related CAS # | O-Demethyl Lenvatinib hydrochloride |
| PubChem CID | 135566046 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.6±0.0 g/cm3 |
| Boiling Point | 644.1±0.0 °C at 760 mmHg |
| Flash Point | 343.4±0.0 °C |
| Vapour Pressure | 0.0±0.0 mmHg at 25°C |
| Index of Refraction | 1.739 |
| LogP | 4.23 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 29 |
| Complexity | 619 |
| Defined Atom Stereocenter Count | 0 |
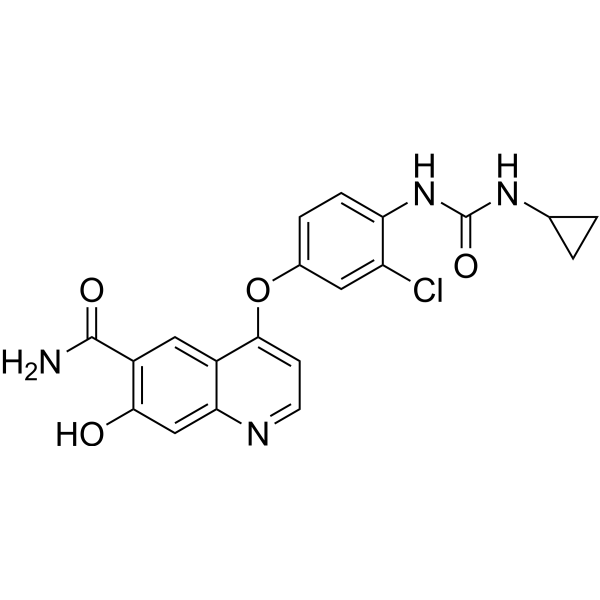
| SMILES | O=C(C1C(O)=CC2N=CC=C(C=2C=1)OC1C=C(Cl)C(NC(NC2CC2)=O)=CC=1)N |
| InChi Key | XEZZOIWZFIDBIQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H17ClN4O4/c21-14-7-11(3-4-15(14)25-20(28)24-10-1-2-10)29-18-5-6-23-16-9-17(26)13(19(22)27)8-12(16)18/h3-10,26H,1-2H2,(H2,22,27)(H2,24,25,28) |
| Chemical Name | 4-[3-chloro-4-(cyclopropylcarbamoylamino)phenoxy]-7-hydroxyquinoline-6-carboxamide |
| Synonyms | 417717-04-5; O-Demethyl Lenvatinib; Lenvatinib metabolite M2; 4-(3-chloro-4-(((cyclopropylamino)carbonyl)amino)phenoxy)-7-hydroxy-6-quinolinecarboxamide; UNII-269N24780P; 4-[3-chloro-4-(cyclopropylcarbamoylamino)phenoxy]-7-hydroxyquinoline-6-carboxamide; 269N24780P; 6-Quinolinecarboxamide, 4-(3-chloro-4-(((cyclopropylamino)carbonyl)amino)phenoxy)-7-hydroxy-; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Lenvatinib metabolite |
| References |
[1]. Lenvatinib: A Promising Molecular Targeted Agent for Multiple Cancers. Cancer Control. 2018 Jan-Dec;25(1):1073274818789361. [2]. Lenvatinib versus Bay 43-9006 in first-line treatment of patients with unresectable hepatocellularcarcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018 Mar 24;391(10126):1163-1173. |
| Additional Infomation |
Lenvatinib is a small-molecule tyrosine kinase inhibitor that inhibits vascular endothelial growth factor receptor (VEGFR1-3), fibroblast growth factor receptor (FGFR1-4), platelet-derived growth factor receptor α (PDGFRα), stem cell factor receptor (KIT), and rearranged during transfection (RET). These receptors are important for tumor angiogenesis, and lenvatinib inhibits tumor angiogenesis by inhibiting function of these receptors. Phase I trials of lenvatinib were conducted at the same time in Japan, Europe, and the United States, and tumor shrinkage effects were observed in thyroid cancer, endometrial cancer, melanoma, renal cell carcinoma, sarcoma, and colon cancer. Lenvatinib is a promising drug that has shown therapeutic effects against various solid tumors. Adverse events, such as hypertension, proteinuria, diarrhea, and delayed wound healing, can occur with lenvatinib treatment. Managing these adverse events is also important for the use of lenvatinib. In this mini-review article, we outline the current state, toxicity, and future prospects of lenvatinib toward thyroid cancer, hepatocellular carcinoma, renal cell carcinoma, and lung cancer. [1] Background: In a phase 2 trial, lenvatinib, an inhibitor of VEGF receptors 1-3, FGF receptors 1-4, PDGF receptor α, RET, and KIT, showed activity in hepatocellular carcinoma. We aimed to compare overall survival in patients treated with lenvatinib versus sorafenib as a first-line treatment for unresectable hepatocellular carcinoma. Methods: This was an open-label, phase 3, multicentre, non-inferiority trial that recruited patients with unresectable hepatocellular carcinoma, who had not received treatment for advanced disease, at 154 sites in 20 countries throughout the Asia-Pacific, European, and North American regions. Patients were randomly assigned (1:1) via an interactive voice-web response system-with region; macroscopic portal vein invasion, extrahepatic spread, or both; Eastern Cooperative Oncology Group performance status; and bodyweight as stratification factors-to receive oral lenvatinib (12 mg/day for bodyweight ≥60 kg or 8 mg/day for bodyweight <60 kg) or sorafenib 400 mg twice-daily in 28-day cycles. The primary endpoint was overall survival, measured from the date of randomisation until the date of death from any cause. The efficacy analysis followed the intention-to-treat principle, and only patients who received treatment were included in the safety analysis. The non-inferiority margin was set at 1·08. The trial is registered with ClinicalTrials.gov, number NCT01761266. Findings: Between March 1, 2013 and July 30, 2015, 1492 patients were recruited. 954 eligible patients were randomly assigned to lenvatinib (n=478) or sorafenib (n=476). Median survival time for lenvatinib of 13·6 months (95% CI 12·1-14·9) was non-inferior to sorafenib (12·3 months, 10·4-13·9; hazard ratio 0·92, 95% CI 0·79-1·06), meeting criteria for non-inferiority. The most common any-grade adverse events were hypertension (201 [42%]), diarrhoea (184 [39%]), decreased appetite (162 [34%]), and decreased weight (147 [31%]) for lenvatinib, and palmar-plantar erythrodysaesthesia (249 [52%]), diarrhoea (220 [46%]), hypertension (144 [30%]), and decreased appetite (127 [27%]) for sorafenib. Interpretation: Lenvatinib was non-inferior to sorafenib in overall survival in untreated advanced hepatocellular carcinoma. The safety and tolerability profiles of lenvatinib were consistent with those previously observed.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4223 mL | 12.1115 mL | 24.2230 mL | |
| 5 mM | 0.4845 mL | 2.4223 mL | 4.8446 mL | |
| 10 mM | 0.2422 mL | 1.2112 mL | 2.4223 mL |