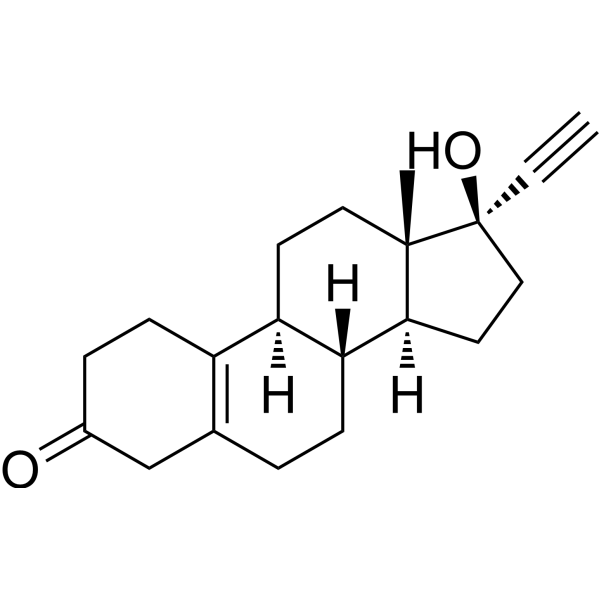
Physicochemical Properties
| Molecular Formula | C20H26O2 |
| Exact Mass | 298.193 |
| CAS # | 68-23-5 |
| PubChem CID | 6231 |
| Appearance | White to off-white solid powder |
| Density | 1.15g/cm3 |
| Boiling Point | 441.6ºC at 760 mmHg |
| Melting Point | 169-170ºC |
| Flash Point | 188.2ºC |
| Index of Refraction | 1.577 |
| LogP | 3.636 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 22 |
| Complexity | 606 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | C[C@]12CC[C@H]3[C@H]([C@@H]1CC[C@]2(C#C)O)CCC4=C3CCC(=O)C4 |
| InChi Key | ICTXHFFSOAJUMG-SLHNCBLASA-N |
| InChi Code | InChI=1S/C20H26O2/c1-3-20(22)11-9-18-17-6-4-13-12-14(21)5-7-15(13)16(17)8-10-19(18,20)2/h1,16-18,22H,4-12H2,2H3/t16-,17-,18+,19+,20+/m1/s1 |
| Chemical Name | (8R,9S,13S,14S,17R)-17-ethynyl-17-hydroxy-13-methyl-1,2,4,6,7,8,9,11,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-3-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ...Metabolized in women; 75% of an acute oral dose was excreted in 7-day urine and feces, with complex pattern of urinary metabolites. Metabolism / Metabolites It is generally considered that progestogens that are structurally related to norethisterone are pro-drugs and that their progestational activity is due to their conversion to norethisterone. After oral administration, norethisterone acetate and ethynodiol diacetate are rapidly converted to norethisterone by esterases during hepatic first-pass metabolism. Although less is known about the transformation of lynestrenol and norethynodrel, it appears that lynestrenol first undergoes hydroxylation at carbon 3 and then oxidation of the hydroxyl group to form norethisterone. Although there is no convincing evidence for the in-vivo transformation of norethynodrel to norethisterone, data from receptor binding tests and bioassays suggest that norethynodrel is also a pro-drug. The /metabolic/ products /of norethynodrel/ are eliminated as glucuronides and sulfates... ...Metabolic studies of norethynodrel in women have revealed the presence of the two 3-hydroxy epimers. The recovery of the two...was not large due to extensive further metabolism by routes such as hydroxylation, but keto reduction appears as the major initial pathway. Double-bond reduction is an important pathway for ... norethynodrel ... /gives/ rise to stereoisomers of 17alpha-ethynyl-5-estrane-3,17beta-diol as significant metabolites in women. From less polar fractions, there were isolated 17alpha- ethynylestr-5(10)-ene-3alpha,17beta-diol, 17alpha-ethynylestr-5 (10)-ene-3beta,17beta-diol, 17 alpha-ethynyl-5beta-estrane-3alpha,17beta-diol, and 17alpha-ethynyl-5 alpha-estrane-3beta, 17 beta-diol. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Norethynodrel is only available in the United States in combination oral contraceptive products. Based on the available evidence, expert opinion holds that nonhormonal methods are preferred during breastfeeding and progestin-only contraceptive are preferred over combined oral contraceptives in breastfeeding women, especially during the first 4 weeks postpartum. Those with an interest in a combination oral contraceptive should consult the record entitled, Contraceptives, Oral, Combined. ◉ Effects in Breastfed Infants Abnormal breast enlargement was reported in a 3-week-old male breastfed infant whose mother was taking an oral contraceptive containing norethynodrel 25 mg and mestranol 100 mcg. The contraceptive was the probable cause of breast enlargement, but most likely the estrogen component mestranol rather than norethynodrel caused the effect. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. However, progestins such as norethynodrel are not considered to suppress lactation. A study analyzed data from a prospective cohort study of U.S. women from May 2005 through June 2007. Women were followed from the third trimester of pregnancy throughout the first year postpartum. Data from the subset of women who intended to breastfeed for 3 months or longer postpartum during their third trimester of pregnancy and who were using a contraceptive at 3 months postpartum were analyzed (n = 1349). Women who intended to breastfeed for at least 4 months and were taking a progestin-only oral contraceptive, such as norethynodrel, were 3.15 times more likely to be breastfeeding (exclusive or nonexclusive) at 4 months than women who used a nonhormonal contraceptive. Women who said they would breastfeed for 3 to 4 months had 4-month breastfeeding rates equivalent to those using a nonhormonal contraceptive. These rates were much higher than those of women who were taking an estrogen-containing, combined oral contraceptive. |
| References |
[1]. 2 - The Clinical Pharmacology of Oestrogens and Progestagens. 1979,. |
| Additional Infomation |
Therapeutic Uses Contraceptives, Oral, Synthetic Norethynodrel indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception. /Former/ Norethynodrel is a progestogen derived from 19-nortestosterone. It is used in oral contraceptive agents and hormonal pregnancy tests (no longer available in the United States). EXPTL USE VET: Experimentally, it has been used with mestranol in swine to synchronize estrus after treatment is withdrawn. Anabolic, androgenic, and fetal masculinization effects are less than with norethindrone... Drug Warnings Norethynodrel exhibits a dose dependent suppression of lactation. Lower infant weight gain, decreased milk production, and decreased composition of nitrogen and protein content of human milk have been associated with similar synthetic progestogens and estrogen products ... . The magnitude of these changes is low. However, the changes in milk production and composition may be of nutritional importance in malnourished mothers. If breast feeding is desired, the lowest dose of oral contraceptives should be chosen. Monitoring of infant weight gain and the possible need for nutritional supplementation should be considered. Use of oral contraceptives is associated with an increased risk of several serious conditions including thromboembolism, stroke, myocardial infarction, liver tumor, gallbladder disease, visual disturbances, fetal abnormalities, and hypertension. Cigarette smoking increases the risk of serious adverse cardiovascular effects during oral contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes daily) and is markedly greater in women older than 35 years of age. Women who are receiving estrogen-progestin contraceptives should be strongly advised not to smoke. Women older than 35 years of age who smoke, and women with ischemic heart disease or a history of this disease, should not use estrogen-progestin contraceptives. Estrogen-progestin contraceptives should be used with caution in women with cardiovascular disease risk factors. /Estrogen-Progestin Combination/ The most frequent adverse effect of oral contraceptives is nausea. In addition, nausea has been reported in women using vaginal or transdermal estrogen-progestin contraceptives. The principal risk associated with currently recommended high-dose, postcoital estrogen-progestin combination regimens appears to be moderate to severe adverse GI effects including severe vomiting and nausea, which occur in 12-22 and 30-66%, respectively, of women receiving the short-course regimens and may limit compliance with, and effectiveness of, the regimens. In 2 prospective, randomized studies, nausea and vomiting were less common with a high-dose postcoital progestin-only regimen (0.75 mg levonorgestrel every 12 hours for 2 doses) than with a high-dose estrogen-progestin regimen (100 mcg ethinyl estradiol and 0.5 mg levonorgestrel every 12 hours for 2 doses). Other adverse GI effects include vomiting, abdominal cramps, abdominal pain, bloating, diarrhea, and constipation. Gingivitis and dry socket have also been reported. Changes in appetite and changes in weight also may occur. /Estrogen-Progestin Combination/ The most frequent dermatologic reaction to oral contraceptives is chloasma or melasma. Women who have had melasma during pregnancy appear to be most susceptible. Irregular brown macules may develop slowly on the face within 1 month to 2 years following initiation of oral contraceptive therapy. The macules fade more slowly than in melasma gravidarum and may be permanent. Acne may improve during oral contraceptive therapy because of decreased sebum production and depression of sebaceous gland activity; however, it may increase in severity during initial therapy and may develop in some women who have not previously had acne. Other dermatologic reactions include allergic rash, urticaria, erythema multiforme, erythema nodosum, hemorrhagic eruption, and pruritus. Hirsutism and alopecia have also occurred. Herpes gestationis and porphyria cutanea have reportedly been adversely affected in women receiving oral contraceptives. /Estrogen-Progestin Combination/ For more Drug Warnings (Complete) data for NORETHYNODREL (32 total), please visit the HSDB record page. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |