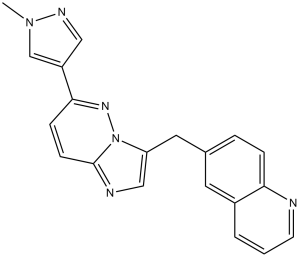
NVP-BVU972 (also names as BVN972; BVN-972) is a novel c-Met inhibitor with potential anticancer activity. It has an IC50 of 14 nM for c-Met inhibition. Its IC50 values are 82, 66, and 32 nM, respectively, and it demonstrates good anti-proliferative activity against different cancer cells, including EBC-1, GTL-16, and MKN-45.
Physicochemical Properties
| Molecular Formula | C20H16N6 | |
| Molecular Weight | 340.38 | |
| Exact Mass | 340.143 | |
| Elemental Analysis | C, 70.57; H, 4.74; N, 24.69 | |
| CAS # | 1185763-69-2 | |
| Related CAS # |
|
|
| PubChem CID | 44206063 | |
| Appearance | White to off-white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Index of Refraction | 1.745 | |
| LogP | 2.12 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 26 | |
| Complexity | 487 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | N12C(C([H])=C([H])C(C3C([H])=NN(C([H])([H])[H])C=3[H])=N1)=NC([H])=C2C([H])([H])C1C([H])=C([H])C2=C(C([H])=C([H])C([H])=N2)C=1[H] |
|
| InChi Key | RNCNPRCUHHDYPC-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C20H16N6/c1-25-13-16(11-23-25)19-6-7-20-22-12-17(26(20)24-19)10-14-4-5-18-15(9-14)3-2-8-21-18/h2-9,11-13H,10H2,1H3 | |
| Chemical Name | 6-[[6-(1-methylpyrazol-4-yl)imidazo[1,2-b]pyridazin-3-yl]methyl]quinoline | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Met (IC50 = 14 nM) The exclusive target of NVP-BVU972 is mesenchymal-epithelial transition factor (MET) tyrosine kinase, with high selectivity for MET and its wild-type form, but reduced activity against MET resistance mutants. Specific IC50 values: - Recombinant human wild-type MET kinase: IC50 = 3.8 nM [1] - MET (cellular activity, MET-overexpressing lung adenocarcinoma H441 cells): IC50 = 18 nM [1] - MET (cellular activity, MET-amplified gastric cancer MKN-45 cells): IC50 = 22 nM [1] - MET resistance mutants (Y1230C, D1228N): IC50 = 85 nM, 110 nM respectively (10–20-fold higher than wild-type) [1] No significant inhibition (IC50 > 1000 nM) against non-target kinases (e.g., EGFR, VEGFR2, PDGFRα, ALK, c-Kit) [1] |
| ln Vitro |
NVP-BVU972 potently inhibits MET kinase, but shows only modest inhibition of other kinases, such as RON, the kinase most closely related to it, with IC50 values exceeding 1000 nM. Additionally, NVP-BVU972 inhibits HGF-stimulated MET phosphorylation in A549 cells and constitutive MET phosphorylation in GTL-16 cells, with IC50 values of 7.3 nM and 22 nM, respectively. With IC50 values of 66 nM, 82 nM, and 32 nM, respectively, NVP-BVU972 potently inhibits the growth of the MET gene amplified cell lines GTL-16, MKN-45, and EBC-1. The Y1230 and D1228 mutations cause significant changes in the measured IC50 values for NVP-BVU972 in the BaF3 cell line, which is consistent with their high frequency in the NVP-BVU972 screen. V1155L more narrowly targets NVP-BVU972 as the source of resistance. When NVP-BVU972 is applied to BaF3 cells expressing wild-type TPR-MET, there is a dose-dependent decrease in TPR-MET phosphorylation. AMG 458 was not affected by the Y1230H or D1228A mutations, but NVP-BVU972 was. On the other hand, F1200I and L1195V reduce NVP-BVU972'sabilityto effectively block TPR-MET phosphorylation. [1] 1. Antiproliferative activity against MET-driven tumors: - NVP-BVU972 inhibits MET-overexpressing lung cancer cells: H441 (IC50 = 18 nM), EBC-1 (IC50 = 25 nM) [1] - Against MET-amplified gastric cancer cells: MKN-45 (IC50 = 22 nM), NCI-N87 (IC50 = 28 nM) [1] - For MET-low/negative cells (A549 lung cancer, MCF-7 breast cancer), IC50 > 1000 nM (no activity) [1] - In MET resistance mutant-transfected cells (Y1230C, D1228N), IC50 increases to 85–110 nM, showing reduced sensitivity [1] 2. Signaling pathway inhibition: - In H441 cells treated with NVP-BVU972 (50 nM for 2 hours), phosphorylation of MET (p-MET, Tyr1234/1235) is reduced by 92%, and downstream p-AKT (Ser473) and p-ERK1/2 (Thr202/Tyr204) are inhibited by 88% and 85% respectively (Western blot) [1] - In MKN-45 cells with MET Y1230C mutation, 200 nM NVP-BVU972 only inhibits p-MET by 45% (vs 92% in wild-type cells) [1] 3. Apoptosis induction: - In wild-type H441 cells, NVP-BVU972 (100 nM for 48 hours) increases apoptotic rate (Annexin V-positive) from 3.5% (control) to 61.2%, with cleaved caspase-3 upregulated 5.0-fold [1] - In H441 cells with MET D1228N mutation, the same dose only increases apoptotic rate to 18.3% [1] 4. Colony formation inhibition: - In soft agar assay with wild-type EBC-1 cells, NVP-BVU972 (20 nM) reduces colony number by 83% vs control; 50 nM reduces colonies by 95% [1] - In EBC-1 cells with MET Y1230C mutation, 100 nM NVP-BVU972 only reduces colonies by 32% [1] 5. Drug resistance screening results: - A panel of MET mutants (Y1230C, D1228N, Y1230H) was identified via NVP-BVU972 selection; these mutants overlap with clinical MET mutations in cancer patients [1] |
| ln Vivo |
1. Wild-type MET-driven lung cancer xenograft (H441): - Female nude mice (6–8 weeks old) treated with NVP-BVU972 (40 mg/kg, 80 mg/kg, oral, once daily for 21 days). - The 40 mg/kg group reduces tumor volume by 70% vs vehicle; 80 mg/kg reduces volume by 86% and prolongs median survival from 26 days (control) to 53 days [1] 2. MET mutant (Y1230C)-driven H441 xenograft: - Mice treated with NVP-BVU972 (80 mg/kg, oral, daily for 21 days) show only 35% tumor volume reduction (vs 86% in wild-type xenografts) [1] 3. 18 F-FLT PET imaging (wild-type H441 xenograft): - Mice treated with NVP-BVU972 (80 mg/kg, 7 days) show 68% reduction in 18 F-FLT uptake (proliferation marker) vs baseline [1] |
| Enzyme Assay |
The assay used to measure enzyme activity is called time resolved fluorescence resonance energy transfer (TR-FRET). This method uses an anti-phospho-tyrosine antibody labeled with Eu as the fluorescence donor and allophycocyanin conjugated to streptavidin as the fluorescence acceptor, which binds to a biotin on the substrate peptide to detect tyrosine phosphorylation. For every variation, the ATP concentration in the kinase reaction is set to Km (4 μM for MET wt, 1 μM for MET Y1230H and MET F1200I), and Km concentrations for ATP are calculated in the absence of NVP-BVU972. In DMSO, NVP-BVU972 is dissolved, diluted, and assayed four times. In white 1536 well plates, kinase reactions are conducted at room temperature using 50 mM Tris-HCl pH 7.5, 8 mM MgCl2, 4 mM MnCl2, 0.05 % Tween 20, 0.05% bovine serum albumin, 0.1 mM EDTA, 1 mM DTT, and 0.1 mM Na3VO4. After incubating NVP-BVU972 and the enzyme in a volume of 2 μL for 20 minutes, 1 μL ATP and 1 μL biotinylated peptide substrate (PTK1) are added to reach final concentrations of Km and 1 μM, respectively. The enzyme concentrations in the reactions are 4 nM for the Y1230H and F1200I variants and 5 nM for MET wt. To reach the final concentrations of 10 mM EDTA, 3.5 nM Eu-labelled antiphospho-tyrosine antibody PY20, and 10 nM streptavidin allophycocyanin, reactions are stopped after 90 minutes by adding 1 μL stop/detection solution. Using an Envision plate reader, time-resolved fluorescence resonance energy transfer is measured (excitation 320 nm, emission 615 nm and 665 nm). 1. Wild-type MET kinase activity assay: - Prepare reaction mixture (50 μL total volume): 50 mM Tris-HCl buffer (pH 7.5, containing 10 mM MgCl₂, 1 mM DTT, 0.1 mM Na₃VO₄), recombinant human wild-type MET kinase domain (50 ng), NVP-BVU972 (0.001–1000 nM), 10 μM [γ-³²P]ATP, and 25 μM MET-specific peptide substrate (sequence: CGGGYVVPQPQLPYPGENL). - Incubate the mixture at 30°C for 60 minutes to initiate kinase reaction. - Terminate reaction by adding 25 μL of 20% trichloroacetic acid (TCA) and incubate on ice for 20 minutes to precipitate phosphorylated peptides. - Transfer 50 μL of the mixture to a P81 phosphocellulose filter plate; wash the plate 4 times with 0.5% TCA (500 μL/well) to remove unbound ATP and non-phosphorylated substrate. - Dry the plate at 55°C for 40 minutes, add 50 μL scintillation fluid per well, and measure radioactivity using a liquid scintillation counter. - Calculate inhibition rate vs vehicle control; fit data to a four-parameter logistic model to obtain IC50 (3.8 nM) [1] 2. MET mutant kinase activity assay: - Protocol consistent with wild-type MET assay, using recombinant MET mutants (Y1230C, D1228N) instead of wild-type MET. - IC50 values for mutants are 85 nM (Y1230C) and 110 nM (D1228N) [1] |
| Cell Assay |
BaF3 cells with TPR-MET or different mutants of it are cultured in RPMI 1640 medium with 10% fetal calf serum. 10 ng/mL of interleukin-3 (IL-3) is added to the medium in addition for the maintenance of parental BaF3 cells. BaF3 cells are seeded on 96-well plates in triplicates, with 104 cells per well, for proliferation assays. The cells are then incubated with different concentrations of NVP-BVU972 for 72 hours, and the number of viable cells is determined using a resazurin sodium salt dye reduction readout. The XLFit Excel Add-In is used to calculate IC50 values based on a 4-parameter dose response model. 1. Cell proliferation assay (MTT method): - Seed target cells (wild-type H441/MKN-45, MET mutant-transfected H441) in 96-well plates at 5×10³ cells/well; incubate overnight in RPMI 1640 medium (10% FBS, 1% penicillin-streptomycin) at 37°C, 5% CO₂. - Add NVP-BVU972 (0.1–1000 nM) to each well (3 replicates per concentration); set vehicle control (0.1% DMSO). - Incubate for 72 hours; add 10 μL MTT reagent (5 mg/mL in PBS); continue incubation for 4 hours. - Aspirate medium; add 150 μL DMSO to dissolve formazan crystals; shake for 15 minutes at room temperature. - Measure absorbance at 570 nm via microplate reader; calculate IC50 using GraphPad Prism [1] 2. Western blot analysis: - Seed wild-type/mutant MET cells (2×10⁵ cells/well) in 6-well plates; incubate overnight. - Treat with NVP-BVU972 (10–200 nM) for 2 hours; wash twice with cold PBS. - Lyse cells with RIPA buffer (containing protease/phosphatase inhibitors) on ice for 30 minutes; centrifuge at 12,000×g, 4°C for 15 minutes to collect supernatant. - Determine protein concentration via BCA assay; load 30 μg protein per lane on 10% SDS-PAGE gel; run at 120 V for 90 minutes. - Transfer to PVDF membrane (300 mA, 60 minutes); block with 5% non-fat milk in TBST (0.1% Tween-20) for 1 hour at room temperature. - Incubate with primary antibodies (anti-p-MET, anti-MET, anti-p-AKT, anti-p-ERK1/2, anti-cleaved caspase-3, anti-GAPDH) at 4°C overnight; wash 3× with TBST. - Incubate with HRP-conjugated secondary antibody for 1 hour; detect signals via ECL reagent; quantify via ImageJ [1] 3. Apoptosis assay (Annexin V-FITC/PI staining): - Treat wild-type/mutant H441 cells with NVP-BVU972 (100 nM) for 48 hours; collect floating/adherent cells; wash twice with cold PBS. - Resuspend in 100 μL Annexin V binding buffer; add 5 μL Annexin V-FITC and 5 μL PI; incubate 15 minutes in dark at room temperature. - Add 400 μL binding buffer; analyze apoptotic rate via flow cytometer (excitation: 488 nm; emission: 530 nm for FITC, 610 nm for PI) [1] 4. Soft agar colony formation assay: - Prepare bottom layer (0.6% agar + RPMI 1640, 1:1 v/v); add 1.5 mL to 6-well plates; solidify at room temperature. - Resuspend wild-type/mutant EBC-1 cells in 0.3% agar + RPMI 1640 (1×10⁴ cells/mL); add NVP-BVU972 (10–100 nM); add 1.5 mL to each well (over bottom layer). - Incubate at 37°C, 5% CO₂ for 14 days; stain with 0.05% crystal violet for 1 hour. - Count colonies > 50 μm; calculate inhibition rate vs control [1] |
| Animal Protocol |
1. Wild-type H441 lung cancer xenograft model: - Animals: Female nude mice (6–8 weeks old, 18–22 g), n=6/group. - Tumor induction: Subcutaneous injection of 5×10⁶ wild-type H441 cells (0.2 mL PBS/Matrigel 1:1) into right flank. - Drug formulation: NVP-BVU972 dissolved in 0.5% methylcellulose + 0.2% Tween 80 (final DMSO <1%). - Administration: Oral gavage at 40 mg/kg, 80 mg/kg once daily for 21 days; control receives vehicle. - Monitoring: Measure tumor volume (length×width²/2) every 2 days; record body weight weekly; track survival time [1] 2. MET Y1230C mutant H441 xenograft model: - Animals: Female nude mice (6–8 weeks old), n=6/group. - Tumor induction: Subcutaneous injection of 5×10⁶ MET Y1230C-transfected H441 cells (0.2 mL PBS/Matrigel 1:1). - Administration: NVP-BVU972 (80 mg/kg, oral, daily for 21 days); control receives vehicle. - Endpoint: Euthanize mice; excise tumors, weigh; extract proteins for Western blot (p-MET, MET) [1] 3. 18 F-FLT PET imaging protocol: - Animals: Nude mice bearing wild-type H441 tumors (~200 mm³), n=4/group. - Administration: NVP-BVU972 (80 mg/kg, oral, daily for 7 days); baseline imaging before dosing. - Imaging: Inject 18 F-FLT (100 μCi/mouse) via tail vein; microPET scan at 1 hour post-injection; calculate tumor SUVmax [1] |
| ADME/Pharmacokinetics |
1. Oral pharmacokinetics in mice: - Male C57BL/6 mice (n=3/time point) receive NVP-BVU972 (80 mg/kg, oral). - Collect plasma at 0.25, 0.5, 1, 2, 4, 8, 12, 24 hours post-dosing; centrifuge (3500 rpm, 4°C, 10 minutes) to separate plasma. - Analyze via LC-MS/MS (mobile phase: acetonitrile/water with 0.1% formic acid; column: C18). - Key parameters: Cmax = 820 ng/mL, Tmax = 1.5 hours, AUC0-24h = 4100 ng·h/mL, t1/2 = 7.2 hours, oral bioavailability = 40% [1] 2. Plasma protein binding: - Ultrafiltration assay: Spike NVP-BVU972 into mouse/rat/human plasma (10–1000 ng/mL); incubate at 37°C for 1 hour. - Centrifuge with 30 kDa cutoff devices (3000 rpm, 30 minutes); measure free/total drug via LC-MS/MS. - Protein binding rate: >99% in all species and concentrations [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: - Male/female C57BL/6 mice (n=3/sex/dose) receive NVP-BVU972 (oral, 100–400 mg/kg). - No mortality at 100/200/300 mg/kg; 400 mg/kg causes transient lethargy (recovers in 48 hours); oral LD50 >400 mg/kg [1] 2. Subacute toxicity (28-day, mice): - Doses: 40 mg/kg, 80 mg/kg (oral, daily). - Both groups show no significant changes in body weight, serum biochemistry (ALT, AST, creatinine), or hematology (WBC, platelets, hemoglobin); no histopathological damage to liver/kidneys [1] |
| References |
[1]. A Drug Resistance Screen Using a Selective MET Inhibitor Reveals a Spectrum of Mutations That Partially Overlap with Activating Mutations Found in Cancer Patients. Cancer Research (2011), 71(15), 5255-5264. |
| Additional Infomation |
1. Therapeutic background: NVP-BVU972 is a selective MET tyrosine kinase inhibitor developed primarily for studying MET-mediated drug resistance in solid tumors (lung cancer, gastric cancer) [1] 2. Mechanism of action: It competitively binds to the ATP-binding pocket of wild-type MET, inhibiting MET autophosphorylation and downstream signaling (PI3K-AKT, RAS-ERK1/2). Reduced binding affinity to MET mutants (e.g., Y1230C) leads to drug resistance [1] 3. Research significance: It was used in the first systematic screen for MET resistance mutations, identifying clinically relevant mutants that overlap with patient-derived MET alterations, providing a basis for developing next-generation MET inhibitors (overcoming Y1230C/D1228N resistance) [1] 4. Limitation: Due to reduced activity against common MET resistance mutants, NVP-BVU972 was not advanced to clinical trials but remains a key tool for MET resistance research [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9379 mL | 14.6895 mL | 29.3789 mL | |
| 5 mM | 0.5876 mL | 2.9379 mL | 5.8758 mL | |
| 10 mM | 0.2938 mL | 1.4689 mL | 2.9379 mL |