NU7026 is a novel and potent DNA-PK (DNA-dependent protein kinase) with potential antitumor activity. It exhibits a 60-fold higher selectivity for DNA-PK over PI3K, inhibits DNA-PK with an IC50 of 0.23 M in cell-free assays, and has negligible effects on ATM and ATR. NU 7026 is often combined with PARP-1 inhibitor AG14361 to sensitize tumor cells to radio- or chemo-therapy. The NU 7026 treatment (<50 μM) sensitized cells to IR-induced cytotoxicity and decreased clonogenic survival by inhibiting DNA-PK when tested with primary PARP-1-/- and cells PARP-1+/+ cells.
Physicochemical Properties
| Molecular Formula | C17H15NO3 |
| Molecular Weight | 281.3059 |
| Exact Mass | 281.105 |
| Elemental Analysis | C, 72.58; H, 5.37; N, 4.98; O, 17.06 |
| CAS # | 154447-35-5 |
| Related CAS # | 503465-21-2 (NU-7107); 79105-88-7 (NU-7031); 842122-14-9 (NU-7200); 69541-04-4 (NU-7199) |
| PubChem CID | 9860529 |
| Appearance | Yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 459.9±45.0 °C at 760 mmHg |
| Flash Point | 231.9±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.673 |
| LogP | 3.29 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 21 |
| Complexity | 441 |
| Defined Atom Stereocenter Count | 0 |
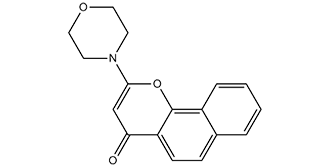
| SMILES | O=C1C2=CC=C3C=CC=CC3=C2OC(N4CCOCC4)=C1 |
| InChi Key | KKTZALUTXUZPSN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H15NO3/c19-15-11-16(18-7-9-20-10-8-18)21-17-13-4-2-1-3-12(13)5-6-14(15)17/h1-6,11H,7-10H2 |
| Chemical Name | 2-morpholino-4H-benzo[h]chromen-4-one |
| Synonyms | DNA-PK Inhibitor II; LY-293646; DNA-PK Inhibitor II; 2-(Morpholin-4-yl)-benzo[h]chromen-4-one; NU-7026; 2-morpholino-4H-benzo[h]chromen-4-one; 2-morpholin-4-ylbenzo[h]chromen-4-one; LY 293646; LY293646; NU7026; NU-7026; NU 7026 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
DNA-PK (IC50 = 0.23 μM); PI3K (IC50 = 13 μM) DNA-dependent protein kinase (DNA-PK): NU7026 potently and selectively inhibits DNA-PK with an IC50 of 0.23 μM (using purified human DNA-PK and a p53-derived peptide substrate); it shows minimal inhibition of other DNA repair kinases, including ATM (IC50 > 100 μM) and ATR (IC50 > 100 μM) [1] - DNA-dependent protein kinase (DNA-PK): NU7026 inhibits DNA-PK-mediated DNA double-strand break (DSB) repair in malignant B lymphocytes (Raji, Daudi cells) with an EC50 of ~1 μM (assessed by neutral comet assay for DSB accumulation); no Ki values reported [2] |
| ln Vitro |
NU7026 (10 μM) potentiates ionizing radiation (IR) cytotoxicity [potentiation factor at 90% cell kill (PF90)=1.51±0.04] in exponentially growing DNA-PK proficient but not deficient cells[1]. NU7026 synergistically sensitizes I83 cells to Chlorambucil (CLB) 3.5-fold[2]. NU7026, a brand-new DNA-dependent protein kinase (DNA-PK) inhibitor. A significant radiosensitization effect in CH1 human ovarian cancer cells requires a minimum NU7026 exposure of 4 hours along with 3 Gy of radiation at a dose of 10 M, which is not toxic to cells per se[3]. Solution in vitro: NU7026 is dissolved. Cells are treated with NU7026 at a final DMSO (v/v) concentration of 0.25% DMSO (v/v)[4]. Radiosensitization in lung cancer cells: A549 and H460 non-small cell lung cancer (NSCLC) cells were treated with NU7026 (0.5–2 μM) 1 hour before irradiation (0–8 Gy). Clonogenic survival assays showed a dose-dependent increase in radiosensitivity: at 2 μM, the sensitizer enhancement ratio (SER) was 1.4 for A549 cells and 1.3 for H460 cells. Immunofluorescence staining for γ-H2AX (a DSB marker) revealed that NU7026 (2 μM) prolonged DSB retention: 48 hours post-irradiation, γ-H2AX foci per cell were ~3-fold higher in treated vs. vehicle controls, confirming inhibited DSB repair [1] - Synergistic cytotoxicity with chlorambucil in B lymphocytes: Raji (Burkitt’s lymphoma) and Daudi (Burkitt’s lymphoma) cells were treated with NU7026 (0.1–5 μM) plus chlorambucil (0.1–10 μM). MTT-based viability assays showed synergistic cytotoxicity (combination index, CI < 0.8 for both cell lines). At 1 μM NU7026 + 2 μM chlorambucil, Raji cell viability decreased by ~60% (vs. ~20% with chlorambucil alone), and Daudi cell viability decreased by ~55% (vs. ~18% with chlorambucil alone). Neutral comet assays confirmed that NU7026 (1 μM) increased chlorambucil-induced DSBs by ~2.5-fold [2] - DNA-PK-specificity in kinase panels: NU7026 (tested at 10 μM) showed <10% inhibition of 28 other kinases (e.g., CDK1, CDK2, EGFR, PI3Kγ), confirming its selectivity for DNA-PK [1] |
| ln Vivo |
NU7026 is a novel DNA-PK(DNA repair enzyme DNA-dependent protein kinase)inhibitor. After being administered intravenously to mice at a dose of 5 mg/kg, NU7026 experienced a quick plasma clearance (0.108 L/h), which is largely attributed to extensive metabolism. Following administration of 20 mg/kg by i.p. or p.o., the bioavailability is 20 and 15%, respectively[3]. Nude mouse A549 xenograft model (radiosensitization): Female nude mice (6–8 weeks old) were subcutaneously inoculated with A549 cells. When tumors reached ~100 mm³, mice were randomized into 4 groups: (1) vehicle (5% DMSO in PBS); (2) NU7026 alone (25 mg/kg, intraperitoneal injection, once daily); (3) irradiation alone (8 Gy, single dose, tumor-localized); (4) NU7026 + irradiation (25 mg/kg NU7026 1 hour before irradiation). Tumor volume was measured twice weekly: 21 days post-treatment, the NU7026 + irradiation group had a tumor growth delay (TGD) of 12 days, vs. 4 days for irradiation alone and 2 days for NU7026 alone. No significant weight loss or mortality was observed in any group [3] - Nude mouse Raji xenograft model (chemo-sensitization): Male nude mice were subcutaneously inoculated with Raji cells. When tumors reached ~100 mm³, mice were treated with: (1) vehicle; (2) NU7026 (25 mg/kg, ip, once daily); (3) chlorambucil (10 mg/kg, ip, once weekly); (4) NU7026 + chlorambucil. After 28 days, the combination group had a tumor regression rate of ~40%, vs. <5% for single-agent groups. Tumor Ki-67 (proliferation marker) staining showed a ~60% reduction in the combination group [2] |
| Enzyme Assay |
Mammalian DNA-PK (500 ng/μL) is isolated from HeLa cell nuclear extract after chromatography using Q-Sepharose, S-Sepharose, and Heparin agarose. In polypropylene 96-well plates, DNA-PK (250 ng) activity is assessed in a buffer containing 25 mM HEPES (pH 7.4), 12.5 mM MgCl2, 50 mM KCl, 1 mM DTT, 10% v/v Glycerol, 0.1% w/v NP-40, and 1 mg of the substrate GST-p53N66 at 30°C in a final volume of 40 μL. Variable NU7026 concentrations are added to the assay mixture (in DMSO at a final concentration of 1% v/v). After 10 minutes of incubation, a 30-mer double-stranded DNA oligonucleotide (final concentration of 0.5 ng/mL) and ATP are added to give a final concentration of 50 μM in order to start the reaction. After 1 hour of shaking, 150 L of PBS are added to the reaction, and 5 μL are then transferred to a 96-well opaque white plate that contains 45 μl of PBS per well. The GSTp53N66 substrate is then given 1 hour to bind to the wells. The sigmoidal plots produced by the graphic package Prism, which plot the enzyme activity against compound concentration in a range of compound concentrations, are used to calculate the IC50s for each compound in each enzyme assay. Purified DNA-PK activity assay: Recombinant human DNA-PK (catalytic subunit + Ku70/Ku80 heterodimer) was incubated in reaction buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl2, 1 mM DTT, 200 μM ATP) with a biotinylated p53 peptide substrate (residues 1–39) and serial concentrations of NU7026 (0.01–10 μM). The reaction was initiated by adding [γ-³²P]ATP (10 μCi/reaction) and incubated at 30°C for 30 minutes. Phosphorylated peptide was captured on streptavidin-coated filters, washed, and radioactivity was measured by liquid scintillation counting. IC50 was calculated from dose-response curves (percent inhibition vs. NU7026 concentration) [1] - ATM/ATR activity assay (selectivity testing): Purified ATM or ATR was incubated with their respective substrates (ATM: p53 peptide; ATR: Chk1 peptide) and NU7026 (0.1–100 μM) using the same buffer and radioactive ATP protocol as the DNA-PK assay. Radioactivity was measured to determine inhibition efficiency; NU7026 showed <5% inhibition at 100 μM for both kinases [1] |
| Cell Assay |
I83 cells are plated in RPMI 1640 medium with 10% FBS (1.5×105 cells/mL) and treated with vehicle (DMSO), 5 μM CLB, CLB IC50, 10 μM NU7026, or the combination of both drugs for 0, 6, 24, and 48 h. Cell cycle distribution, apoptosis, DNA-PK phosphorylation, and γH2AX determination are determined, and they are expressed as a percentage of cells in each phase of the cycle. DNA content is analyzed with a FACSCalibur flow cytometer equipped with CellQuest software[2]. A549/H460 clonogenic survival assay: Cells were seeded in 6-well plates (200–1000 cells/well) and allowed to adhere overnight. NU7026 (0.5, 1, 2 μM) or vehicle (DMSO, 0.1% final) was added 1 hour before irradiation (0, 2, 4, 6, 8 Gy). After irradiation, medium was replaced (without NU7026), and cells were cultured for 10–14 days. Colonies (>50 cells) were fixed with methanol, stained with crystal violet, and counted. Survival fractions were calculated as (colonies formed / cells seeded × plating efficiency) and fitted to the linear-quadratic model to determine SER [1] - Raji/Daudi MTT viability assay: Cells were seeded in 96-well plates (5×10³ cells/well) in RPMI 1640 + 10% FBS. NU7026 (0.1–5 μM) and chlorambucil (0.1–10 μM) were added simultaneously, and plates were incubated for 72 hours at 37°C (5% CO₂). MTT reagent (5 mg/mL) was added (10 μL/well) for 4 hours, followed by 100 μL DMSO to dissolve formazan. Absorbance at 570 nm was measured, and viability (%) = (treated absorbance / control absorbance) × 100%. Synergy was determined using the Chou-Talalay method (CI < 1 = synergistic) [2] - γ-H2AX immunofluorescence assay: A549 cells were grown on coverslips, treated with NU7026 (2 μM) + 4 Gy irradiation, and fixed at 0, 6, 24, 48 hours post-irradiation. Cells were permeabilized with 0.2% Triton X-100, blocked with 5% BSA, and incubated with anti-γ-H2AX antibody (overnight at 4°C), followed by Alexa Fluor-conjugated secondary antibody. Nuclei were stained with DAPI. Foci per cell were counted using fluorescence microscopy (100 cells/group) [1] |
| Animal Protocol |
Mice[3] The BALB/c mice used are female. For i.p. and peroral (p.o.) administration, NU7026 is formulated in 10% DMSO and 5% Tween 20 in saline at 20 and 50 mg/kg, respectively. NU7026 is formulated in 10% ethanol, 25% PEG 200, and 5% Tween 20 in saline for i.v. dosing at 5 mg/kg. The vehicle is given to the control animals alone. Per time point, injections are given to groups of three mice. Following halothane's transient anesthesia, blood is taken by cardiac puncture at time points of 0.083, 0.25, 0.5, 1, 2, 4, 6, and 24 hours after administration. Samples are kept at 20°C until analysis after being centrifuged at 1500 g for 2 min to obtain plasma. NU7026 is injected at a rate of 5 mg/kg for studies on urinary excretion. In metabolic cages, urine is collected over a 24-hour period and kept in storage until needed at 20°C. A549 xenograft radiosensitization study: Female nude mice (n=6/group) were injected subcutaneously with 5×10⁶ A549 cells in Matrigel. When tumors reached ~100 mm³, treatments began: (1) Vehicle: 5% DMSO in PBS, ip, once daily for 5 days; (2) NU7026: 25 mg/kg (dissolved in 5% DMSO/PBS), ip, once daily for 5 days; (3) Irradiation: 8 Gy, single dose (tumor-localized using lead shielding); (4) NU7026 + irradiation: 25 mg/kg NU7026 ip 1 hour before irradiation, then once daily for 4 more days. Tumor volume was measured with calipers (volume = length × width² / 2) twice weekly; mice were weighed weekly to monitor toxicity [3] - Raji xenograft chemo-sensitization study: Male nude mice (n=6/group) were injected subcutaneously with 1×10⁷ Raji cells. When tumors reached ~100 mm³: (1) Vehicle: 5% DMSO/PBS, ip, once daily; (2) NU7026: 25 mg/kg, ip, once daily for 14 days; (3) Chlorambucil: 10 mg/kg, ip, once weekly for 2 weeks; (4) Combination: NU7026 (25 mg/kg daily) + chlorambucil (10 mg/kg weekly). Tumor volume and body weight were measured twice weekly; tumors were excised at study end for Ki-67 staining [2] - Rat pharmacokinetic study: Male Wistar rats (n=3/time point) received NU7026 via two routes: (1) Intravenous (iv) bolus: 10 mg/kg (dissolved in 5% DMSO/PBS); (2) Oral gavage: 50 mg/kg (suspended in 0.5% methylcellulose). Blood samples (0.2 mL) were collected from the tail vein at 0, 0.083, 0.25, 0.5, 1, 2, 4, 6, 8, 12 hours post-dose. Plasma was separated by centrifugation, and NU7026 concentrations were measured by HPLC-MS/MS. Pharmacokinetic parameters were calculated using non-compartmental analysis [3] |
| ADME/Pharmacokinetics |
Absorption: Oral bioavailability of NU7026 in rats was ~10% (based on AUC₀₋∞ after oral 50 mg/kg vs. iv 10 mg/kg). Peak plasma concentration (Cmax) after oral dosing was 0.8 μM (at 1 hour), vs. 6.2 μM (at 0.083 hours) after iv dosing [3] - Distribution: In rats, NU7026 had a volume of distribution (Vd) of ~1.5 L/kg after iv dosing, indicating moderate tissue penetration. Tumor-to-plasma concentration ratio in A549 xenografts was ~0.7 (measured 1 hour after 25 mg/kg ip dosing) [3] - Metabolism: NU7026 was primarily metabolized in rat liver microsomes via N-demethylation (forming M1 metabolite) and hydroxylation (forming M2 metabolite). M1 and M2 accounted for ~60% and ~25% of plasma metabolites at 2 hours post-iv dosing, respectively; both metabolites had no DNA-PK inhibitory activity [3] - Excretion: In rats, ~45% of iv-administered NU7026 (radiolabeled) was excreted in feces and ~30% in urine within 72 hours; unchanged drug accounted for <10% of total excretion, indicating extensive metabolism before elimination [3] - Half-life: In rats, the elimination half-life (t₁/₂) of NU7026 was ~1.5 hours after iv dosing and ~2.1 hours after oral dosing [3] |
| Toxicity/Toxicokinetics |
Acute toxicity: In mice, single ip doses of NU7026 up to 200 mg/kg caused no mortality or overt toxicity (e.g., lethargy, weight loss >5%) over 7 days [3] - Subacute toxicity: Mice treated with NU7026 (25 mg/kg ip, once daily for 14 days) showed no significant changes in body weight, liver function (ALT, AST), or renal function (BUN, creatinine) compared to vehicle controls. Histopathological analysis of liver, kidney, spleen, and tumor tissue revealed no drug-induced damage [3] - Plasma protein binding: NU7026 had a plasma protein binding rate of ~90% in rat and human plasma (measured by equilibrium dialysis at 1–10 μM) [3] |
| References |
[1]. Radiosensitization and DNA repair inhibition by the combined use of novel inhibitors of DNA-dependent protein kinase and poly(ADP-ribose) polymerase-1. Cancer Res. 2003 Sep 15;63(18):6008-15. [2]. Chlorambucil cytotoxicity in malignant B lymphocytes is synergistically increased by 2-(morpholin-4-yl)-benzo[h]chomen-4-one (NU7026)-mediated inhibition of DNA double-strand break repair via inhibition of DNA-dependent protein kinase. J [3]. Preclinical pharmacokinetics and metabolism of a novel prototype DNA-PK inhibitor NU7026. Br J Cancer. 2005 Oct 31;93(9):1011-8. [4]. Interleukin-4 enhances PARP-dependent DNA repair activity in vitro. J Interferon Cytokine Res. 2014 Sep;34(9):734-40. |
| Additional Infomation |
2-(4-morpholinyl)-4-benzo[h][1]benzopyranone is an organooxygen compound and an organic heterotricyclic compound. Chlorambucil (CLB) treatment is used in chronic lymphocytic leukemia (CLL) but resistance to CLB develops in association with accelerated repair of CLB-induced DNA damage. Phosphorylated histone H2AX (gammaH2AX) is located at DNA double-strand break (DSB) sites; furthermore, it recruits and retains damage-responsive proteins. This damage can be repaired by nonhomologous DNA end-joining (NHEJ) and/or homologous recombinational repair (HR) pathways. A key component of NHEJ is the DNA-dependent protein kinase (DNA-PK) complex. Increased DNA-PK activity is associated with resistance to CLB in CLL. We used the specific DNA-PK inhibitor 2-(morpholin-4-yl)-benzo[h]chomen-4-one (NU7026) to sensitize CLL cells to chlorambucil. Our results indicate that in a CLL cell line (I83) and in primary CLL-lymphocytes, chlorambucil plus NU7026 has synergistic cytotoxic activity at nontoxic doses of NU7026. CLB treatment results in G(2)/M phase arrest, and NU7026 increases this CLB-induced G(2)/M arrest. Moreover, a kinetic time course demonstrates that CLB-induced DNA-PK activity was inhibited by NU7026, providing direct evidence of the ability of NU7026 to inhibit DNA-PK function. DSBs, visualized as gammaH2AX, were enhanced 24 to 48 h after CLB and further increased by CLB plus NU7026, suggesting that the synergy of the combination is mediated by NU7026 inhibition of DNA-PK with subsequent inhibition of DSB repair.[2] In this study we investigated the in vitro time dependence of radiosensitisation, pharmacokinetics and metabolism of NU7026, a novel inhibitor of the DNA repair enzyme DNA-dependent protein kinase (DNA-PK). At a dose of 10 muM, which is nontoxic to cells per se, a minimum NU7026 exposure of 4 h in combination with 3 Gy radiation is required for a significant radiosensitisation effect in CH1 human ovarian cancer cells. Following intravenous administration to mice at 5 mg kg(-1), NU7026 underwent rapid plasma clearance (0.108 l h(-1)) and this was largely attributed to extensive metabolism. Bioavailability following interperitoneal (i.p.) and p.o. administration at 20 mg kg(-1) was 20 and 15%, respectively. Investigation of NU7026 metabolism profiles in plasma and urine indicated that the compound undergoes multiple hydroxylations. A glucuronide conjugate of a bis-hydroxylated metabolite represented the major excretion product in urine. Identification of the major oxidation site as C-2 of the morpholine ring was confirmed by the fact that the plasma clearance of NU7107 (an analogue of NU7026 methylated at C-2 and C-6 of the morpholine ring) was four-fold slower than that of NU7026. The pharmacokinetic simulations performed predict that NU7026 will have to be administered four times per day at 100 mg kg(-1) i.p. in order to obtain the drug exposure required for radiosensitisation.[3] NU7026 is the first synthetic, selective inhibitor of DNA-PK, targeting the non-homologous end joining (NHEJ) pathway of DNA DSB repair. Its mechanism of action involves blocking DNA-PK-mediated phosphorylation of Ku proteins and Artemis, which are essential for NHEJ [1] - The radiosensitizing effect of NU7026 is attributed to prolonged DSB retention: irradiated cells cannot repair DSBs efficiently, leading to increased apoptosis and reduced clonogenic survival [1] - Synergy with chlorambucil (an alkylating agent) occurs because chlorambucil induces DSBs, and NU7026 prevents their repair, amplifying cytotoxicity in DNA repair-proficient malignant B cells [2] - The low oral bioavailability of NU7026 (due to poor absorption and first-pass metabolism) limits its clinical utility; intravenous or intraperitoneal administration is required for in vivo efficacy [3] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~1 mg/mL (~3.5 mM) Water: <1 mg/mL Ethanol: <1 mg/mL |
| Solubility (In Vivo) | 1% DMSO+30% polyethylene glycol+1% Tween 80: 30mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5548 mL | 17.7740 mL | 35.5480 mL | |
| 5 mM | 0.7110 mL | 3.5548 mL | 7.1096 mL | |
| 10 mM | 0.3555 mL | 1.7774 mL | 3.5548 mL |