Physicochemical Properties
| Molecular Formula | C18H36N2O |
| Molecular Weight | 296.49 |
| Exact Mass | 368.236 |
| Elemental Analysis | C, 72.92; H, 12.24; N, 9.45; O, 5.40 |
| CAS # | 150661-91-9 |
| PubChem CID | 373395 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 4.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 21 |
| Complexity | 263 |
| Defined Atom Stereocenter Count | 0 |
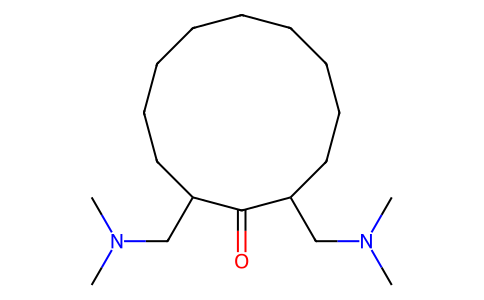
| SMILES | O=C1C(CN(C)C)CCCCCCCCCC1CN(C)C |
| InChi Key | HRQRILLGGNNYSD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H36N2O/c1-19(2)14-16-12-10-8-6-5-7-9-11-13-17(18(16)21)15-20(3)4/h16-17H,5-15H2,1-4H3 |
| Chemical Name | 2,12-bis[(dimethylamino)methyl]cyclododecan-1-one |
| Synonyms | NC 1153; NC-1153; NC1153; 150661-91-9; 2,12-bis[(dimethylamino)methyl]cyclododecanone; 2,12-bis[(dimethylamino)methyl]cyclododecan-1-one; 2,12-Bis[(dimethylamino)methyl]-cyclododecanone; 2,12-Bis((dimethylamino)methyl)cyclododecan-1-one; NSC649641; starbld0001536; NC1153 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | JAK3 |
| ln Vitro | NC1153 blocked IL-2-induced activation of JAK3 and its downstream substrates STAT5a/b more effectively than activation of the closely related prolactin-induced JAK2 or TNF-alpha-driven NF-kappaB. In addition, NC1153 failed to inhibit several other enzymes, including growth factor receptor tyrosine kinases, Src family members, and serine/threonine protein kinases. Although NC1153 inhibited proliferation of normal human T cells challenged with IL-2, IL-4, or IL-7, it did not block T cells void of JAK3 [1]. |
| ln Vivo | In vivo, a 14-day oral therapy with NC1153 significantly extended survival of MHC/non-MHC mismatched rat kidney allografts, whereas a 90-day therapy induced transplantation tolerance (>200 days). Although NC1153 acted synergistically with cyclosporin A (CsA) to prolong allograft survival, it was not nephrotoxic, myelotoxic, or lipotoxic and did not increase CsA-induced nephrotoxicity. In contrast to CsA, NC1153 was not metabolized by cytochrome P450 3A4. Thus, NC1153 prolongs allograft survival without several toxic effects associated with current immunosuppressive drugs[1]. |
| Enzyme Assay |
Tyrosine kinase assay[1] JAK3 autokinase assays were performed by immunopurifying human JAK3 from T cells that were washed three times with lysis buffer followed by a single wash with kinase buffer containing 25 mM HEPES, pH 7.3, 0.1% Triton X-100, 100 mM NaCl, 10 mM MgCl2, 3 mM MnCl2, and 200 μM sodium orthovanadate. Isotope-free tyrosine kinase reactions were initiated by the addition of 100 μM unlabeled ATP and allowed to incubate at 37°C for 15 min in the presence of NC1153. The reactions were quenched by washing the protein A-Sepharose beads with lysis buffer and eluting bound material by boiling in SDS-sample buffer for 4 min and resolved by 7.5% SDS-PAGE. Anti-phosphotyrosine immunoblotting was performed as described above. Quantitations of tyrosine-phosphorylated JAK3 and total JAK3 reblots were assessed using an Expression 633 scanner, and densitometry of bands was standardized against background. Normalized phosphorylation was calculated by dividing the absorbance ratio of phosphorylated-JAK3/total-JAK3 from vehicle or NC1153-treated samples. Analysis of NC1153 effects on other growth factor receptors, Src family members, and protein kinases A and C were performed according to the Upstate Biotechnology Cell Signaling Solutions and Kinase Profiler. All NC1153 inhibitory kinase reactions were initiated in the presence of 100 μM ATP and normalized to vehicle. Values are presented as the percentage of mean (n = 2) of control (without drug) and SD, which is reported as the range/√2. Measurement of P450 activity[1] Effect of NC1153 on different P450 isoforms (3A4, 2D6, 2C19, 1A2, 2C8, and 2C9) were tested using the previously described method. Positive controls with selective substrates were used to monitor P450 3A4 (ketoconazole), P450 2D6 (quinidine), P450 2C19 (tranylcypromine), P450 1A2 (puratyline), P450 2C8 (quercetin), and P450 2C9 (sulfaphenazole) conversion to substrate. Pooled human microsomes were assayed with known P450-selective substrates and competition by escalating concentrations (−8 to −4 log M concentration) of NC1153. Data are plotted as percent inhibition of P450 isoform activity with a competitive substrate without or with NC1153 × 100. |
| Cell Assay |
Flow cytometry[1] T lymphocytes were purified via a negative T cell isolation kit and subsequently activated with PHA for 72 h and then treated without or with 50 μM NC1153 for 16 h; cells were then stained with BD Pharmingen reagents, including PE-labeled mouse anti-human IL2Rα, PE-conjugated mouse anti-human IL2Rβ, and PE-labeled mouse anti-human γc Abs. Cell staining and FACScan were performed as previously described. Proliferation assaysy[1] Quiescent human T cells or Jurkat cells (5.0 × 104/well) were plated in flat-bottom, 96-well microtiter plates in 200 μl of quiescent medium containing RPMI 1640 and 1% FCS in the absence or presence of 1 nM IL-2, IL-4, or IL-7 or PRL. Next, cells were treated for 16 h with NC1153 and then pulsed for 4 h with [3H]thymidine (0.5 μCi/200 μl) and harvested onto fiberglass filters and analyzed by liquid scintillation counting as previously described. |
| Animal Protocol |
Rat kidney and spleen transplants[1] ACI (WF; RT1a) and Lewis (RT1l) rats (160–200 g) obtained from Harlan Sprague Dawley were cared for according to the guidelines of the University of Texas Animal Welfare Committee. Rats were housed in light- and temperature-controlled quarters and given chow and water ad libitum. Orthotopic kidney transplantation was performed using a standard microsurgical technique of end-to-side anastomoses to recipient aorta and vena cava. The cold ischemia times were <30 min. Graft survival time was defined as the last survival day of bilaterally nephrectomized recipients. Recipients remained untreated or were treated with NC1153 alone by daily i.v. injections (2.5–20 mg/kg) for 7 days or by daily oral gavage for 7 days (20–160 mg/kg) or 14 days (40–240 mg/kg); combined therapy comprised 7-day oral gavage with NC1153 (20–160 mg/kg) and/or 3-day oral gavage with CsA (2.5–20 mg/kg). Some recipients were treated with 160 mg/kg NC1153 for 14 days and thereafter three times a week for up to 90 days with 160 or 240 mg/kg NC1153. The results, presented as mean survival time (MST) ± SD, were assessed for statistical significance by Gehan’s survival test. In addition, the interaction between NC1153 and CsA was evaluated by the median effect analysis. Combination index (CI) value of <1 suggests synergistic, >1 antagonistic, or =1 additive interactions (43). Spleen transplantation was performed by a previously described method. Irradiated (750 rad) Lewis rat spleen allografts were transplanted to ACI recipients that remained untreated or were treated with 160 mg/kg NC1153 for 7 days. Toxicity study[1] After a 7-day conditioning period on low-salt chow, rats (n = 5 or 6) were randomly assigned to treatment for 28 days with p.o. 160 or 240 mg/kg NC1153 alone, 10 mg/kg CsA alone, 1.6 mg/kg SRL alone, or NC1153-CsA or CsA-SRL combinations. In addition, there was an untreated control group (six rats) fed a low-salt diet. At day 28, the animals were placed in metabolic cages for 24-h urine collections. Blood samples were used for serum hematocrit and hemoglobin, as well as creatinine, total cholesterol, and high-density lipoprotein (HDL)- and low-density lipoprotein (LDL)-cholesterol determinations. Creatinine clearance values were calculated based on urinary (milligrams per milliliter) and plasma creatinine concentrations (milligrams per milliliter). Results are presented as mean values ± SD, and statistical significance was compared; p < 0.05 was considered as significant. The kidney sections, stained with progressive H&E, were evaluated using a semiquantitative five-grade scale for tubular and glomerular changes: 0, none; 1+, <5%; 2+, 6–25%; 3+, 26–50%; and 4+, >50%. A similar scale was used for vascular changes: 0, none; 1+, minimal; 2+, mild; 3+, moderate; and 4+, severe. The right femurs sectioned and stained with H&E were estimated as the percentage of the marrow space occupied by cellular as opposed to adipose tissue elements. The average number of megakaryocytes in four high-power fields was used to estimate platelet formation. |
| Toxicity/Toxicokinetics | NC1153 does not display nephrotoxicity, myelosuppression, and lipotoxicity We have evaluated the potential toxicities produced by NC1153 alone compared with those produced by CsA or SRL alone. In addition, the potential interactive toxicities between the NC1153-CsA vs SRL-CsA combinations also were evaluated. Our previous work in the salt-depleted rats revealed that SRL alone caused myelosuppression and lipotoxicity, whereas the SRL/CsA combination significantly aggravated CsA-induced nephrotoxicity (14). Our present results documented that a 28-day daily oral administration of NC1153 (160 or 240 mg/kg) did not produce renal dysfunction or lipotoxicity (Fig. 6,A, a–f). Both serum creatinine and creatinine clearance confirmed that the addition of SRL to CsA significantly increased CsA-induced nephrotoxicity (Fig. 6,A, a and b). In contradistinction, addition of 160 or 240 mg/kg NC1153 to the same CsA protocol had no impact on CsA-induced nephrotoxicity (Fig. 6,A, a and b).[1] |
| References | [1]. The Mannich base NC1153 promotes long-term allograft survival and spares the recipient from multiple toxicities. J Immunol . 2005 Oct 1;175(7):4236-46. doi: 10.4049/jimmunol.175.7.4236. |
| Additional Infomation | In conclusion, NC1153 selectively disrupts γc-cytokine pathways and JAK3 kinase activity as opposed to a limited pool of other kinases, which should not be considered an exhaustive study. NC1153 prevents allograft rejection and can induce transplantation tolerance. The combination of NC1153 and CsA produces therapeutic synergism to protect kidney allograft survival. Moreover, NC1153 lack toxicities associated with CsA and SRL alone or in combination. Thus, NC1153 may represent a novel class of molecules with potential for clinical immunosuppression without toxicities associated with currently used agents.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3728 mL | 16.8640 mL | 33.7280 mL | |
| 5 mM | 0.6746 mL | 3.3728 mL | 6.7456 mL | |
| 10 mM | 0.3373 mL | 1.6864 mL | 3.3728 mL |