Physicochemical Properties
| Molecular Formula | C18H19NO4 |
| Molecular Weight | 313.3478 |
| Exact Mass | 313.131 |
| Elemental Analysis | C, 69.00; H, 6.11; N, 4.47; O, 20.42 |
| CAS # | 66648-43-9 |
| Related CAS # | 66648-43-9 (E-configuration); 65646-26-6 (E-configuration); 80510-09-4 (Z-configuration) |
| PubChem CID | 5280537 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 554.2±60.0 °C at 760 mmHg |
| Melting Point | 144.5 - 145 °C |
| Flash Point | 289.0±32.9 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.566 |
| LogP | 3.33 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 23 |
| Complexity | 391 |
| Defined Atom Stereocenter Count | 0 |
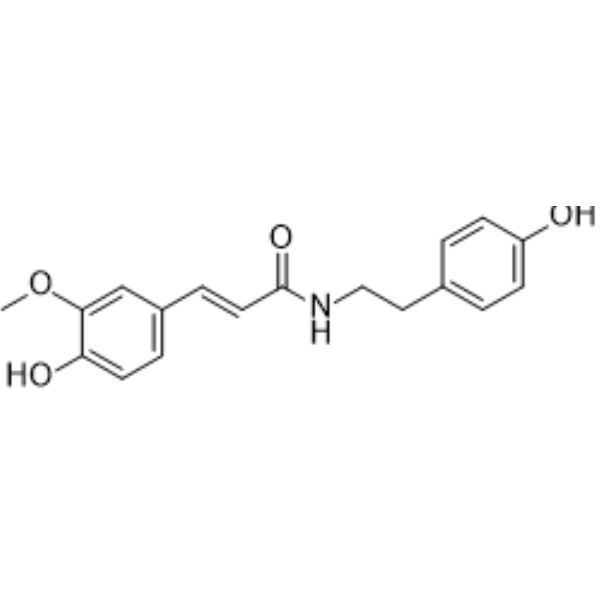
| SMILES | COC1=C(C=CC(=C1)/C=C/C(=O)NCCC2=CC=C(C=C2)O)O |
| InChi Key | NPNNKDMSXVRADT-WEVVVXLNSA-N |
| InChi Code | InChI=1S/C18H19NO4/c1-23-17-12-14(4-8-16(17)21)5-9-18(22)19-11-10-13-2-6-15(20)7-3-13/h2-9,12,20-21H,10-11H2,1H3,(H,19,22)/b9-5+ |
| Chemical Name | (E)-3-(4-hydroxy-3-methoxyphenyl)-N-[2-(4-hydroxyphenyl)ethyl]prop-2-enamide |
| Synonyms | Feruloyltyramine; N-Feruloyltyramine; trans-N-Feruloyltyramine; 65646-26-6; CHEBI:17818; DTXSID30904143; 2-Propenamide, 3-(4-hydroxy-3-methoxyphenyl)-N-(2-(4-hydroxyphenyl)ethyl)-; ...; 66648-43-9; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural alkaloid and antioxidant |
| ln Vitro |
N-trans-ferulamide (N-trans-ferulamide; 10-500 µM; 3 hours before H2O2) protects cells against H2O2-induced damage [1]. N-trans-ferulamine (25-100 µM; 3 hours before H2O2 treatment) significantly lowers Bax and activator caspase-3 levels in H2O2-treated SK-N-SH cells at 100 µM [1] . N-trans-ferulamide can greatly improve the H2O2-mediated increase in ROS levels [1] N-trans-ferulamide (10-500 µM) does not influence the viability of SK-N-SH cells [1]. N-trans-ferulamine (64-320 µM; 24 hours) exerts a proliferation inhibitory impact on HepG2 cells, IC50 cell viability determination [1]
1. Protection against H₂O₂-induced cytotoxicity in human neuroblastoma SK-N-SH cells: SK-N-SH cells were pretreated with N-trans-Feruloyltyramine (10 μM, 20 μM, 40 μM) for 24 h, followed by exposure to 200 μM H₂O₂ for 6 h. N-trans-Feruloyltyramine increased cell viability in a dose-dependent manner (MTT assay): cell viability was (52.3±3.1)% in the H₂O₂-only group, vs. (68.5±2.8)%, (79.2±3.5)%, and (85.7±2.6)% in the 10 μM, 20 μM, and 40 μM pretreatment groups, respectively (p<0.05 vs. H₂O₂ group). It reduced intracellular reactive oxygen species (ROS) accumulation (DCFH-DA probe, flow cytometry): 40 μM N-trans-Feruloyltyramine decreased ROS levels by (42.1±3.2)% (p<0.01). It also modulated oxidative stress markers: malondialdehyde (MDA, lipid peroxidation indicator) decreased from (5.8±0.4) nmol/mg protein (H₂O₂ group) to (3.1±0.3) nmol/mg protein (40 μM group, p<0.01); superoxide dismutase (SOD) activity increased from (85.2±4.3) U/mg protein to (128.6±5.1) U/mg protein, and glutathione peroxidase (GSH-Px) activity increased from (62.3±3.8) U/mg protein to (95.7±4.5) U/mg protein (40 μM, p<0.01). Western blot showed upregulated anti-apoptotic Bcl-2 (relative expression: 0.42±0.05 vs. 0.89±0.07, H₂O₂ vs. 40 μM group, p<0.01) and downregulated pro-apoptotic Bax (1.35±0.08 vs. 0.72±0.06, p<0.01) and cleaved caspase-3 (1.28±0.09 vs. 0.51±0.05, p<0.01) [1] 2. Antioxidant, cytotoxic, and H₂O₂-induced damage protective effects in HepG2 (human hepatocellular carcinoma) and L02 (normal human liver) cells: (1) Antioxidant activity: N-trans-Feruloyltyramine scavenged DPPH and ABTS radicals dose-dependently, with IC₅₀ values of (25.3±1.2) μM (DPPH) and (18.7±0.9) μM (ABTS) (lower than vitamin C: DPPH IC₅₀ 15.2±0.8 μM, ABTS IC₅₀ 12.5±0.7 μM, p<0.05) [2] (2) Cytotoxicity to HepG2: HepG2 cells treated with N-trans-Feruloyltyramine (20–100 μM) for 48 h showed dose-dependent viability reduction (MTT assay), with IC₅₀ (48 h) = (78.5±2.3) μM; no cytotoxicity to L02 cells (viability >85% vs. control, p>0.05) [2] (3) Protection against H₂O₂-induced L02 damage: L02 cells pretreated with N-trans-Feruloyltyramine (20 μM, 40 μM) for 24 h, then exposed to 400 μM H₂O₂ for 8 h. Cell viability increased from (45.6±3.2)% (H₂O₂ group) to (67.8±2.9)% (20 μM) and (81.2±3.4)% (40 μM, p<0.01). MDA decreased from (6.2±0.5) nmol/mg protein to (3.5±0.3) nmol/mg protein (40 μM, p<0.01); intracellular GSH increased from (12.3±1.1) μmol/g protein to (21.5±1.4) μmol/g protein (40 μM, p<0.01). Western blot showed upregulated nuclear Nrf2 (0.35±0.04 vs. 0.82±0.06, H₂O₂ vs. 40 μM group, p<0.01) and HO-1 (0.41±0.05 vs. 0.95±0.07, p<0.01) [2] |
| Enzyme Assay |
1. SOD activity assay (for SK-N-SH/L02 cells):
Harvested cells were homogenized in ice-cold saline, centrifuged (3000 rpm, 10 min, 4°C) to collect supernatant. SOD activity was measured via xanthine oxidase method: the reaction system included supernatant, xanthine, and xanthine oxidase, incubated at 37°C for 20 min. Absorbance was measured at 550 nm. One SOD unit = enzyme amount inhibiting nitrotetrazolium blue reduction by 50%, expressed as U/mg protein [1][2] 2. GSH-Px activity assay (for SK-N-SH cells): Cell supernatant (prepared as above) was mixed with GSH and H₂O₂, incubated at 37°C for 30 min. Remaining GSH reacted with DTNB to form a yellow product; absorbance was measured at 412 nm. GSH-Px activity was calculated based on GSH reduction, expressed as U/mg protein [1] 3. Catalase (CAT) activity assay (for L02 cells): Cell supernatant was mixed with H₂O₂; H₂O₂ decomposition was monitored by absorbance decrease at 240 nm (every 30 s for 3 min). Activity was calculated via H₂O₂ molar extinction coefficient, expressed as U/mg protein [2] |
| Cell Assay |
cell viability determination [1] Cell Types: SK-N-SH Cell Tested Concentrations: 10, 25, 50, 100, 150, 250, 500 µM Incubation Duration: 3 hrs (hours) before H2O2 Experimental Results: Protected cells resisted toxicity induced by H2O2 (150 µM), as determined by a significant increase in percent cell viability. Western Blot Analysis[1] Cell Types: SK-N-SH Cell Tested Concentrations: 25, 50, 100 μM Incubation Duration: 3 hrs (hours) before H2O2 Experimental Results: Bax expression induced by H2O2 was eliminated. Dramatically reduces activated caspase-3 levels. 1. SK-N-SH cell assay (literature [1]): (1) Cell culture: SK-N-SH cells were cultured in DMEM + 10% FBS + 1% penicillin-streptomycin (37°C, 5% CO₂), used in logarithmic growth phase. (2) Treatment protocol: Cells seeded in 96-well plates (5×10³ cells/well) overnight, then pretreated with N-trans-Feruloyltyramine (10/20/40 μM, DMSO <0.1%) for 24 h, followed by 200 μM H₂O₂ for 6 h. Control = 0.1% DMSO; H₂O₂ group = H₂O₂ alone. (3) Cell viability (MTT): 20 μL MTT (5 mg/mL) added, incubated 4 h at 37°C. DMSO dissolved formazan; absorbance measured at 570 nm. Viability = (treatment absorbance / control absorbance) × 100% (4) ROS detection: Cells seeded in 6-well plates (2×10⁵ cells/well) treated as above, then incubated with 10 μM DCFH-DA (30 min, 37°C, dark). Washed with PBS, trypsinized, and analyzed by flow cytometry (Ex 488 nm, Em 525 nm). (5) Western blot (apoptotic proteins): Cells lysed with RIPA + protease inhibitors; protein quantified by BCA. 30 μg protein separated by 12% SDS-PAGE, transferred to PVDF membranes. Blocked with 5% non-fat milk (1 h, RT), incubated with primary antibodies (Bcl-2, Bax, cleaved caspase-3, GAPDH) overnight (4°C). HRP-conjugated secondary antibodies (1 h, RT); bands visualized by ECL, quantified via ImageJ [1] 2. HepG2/L02 cell assay (literature [2]): (1) Cell culture: HepG2/L02 cells cultured in RPMI 1640 + 10% FBS + 1% penicillin-streptomycin (37°C, 5% CO₂). (2) HepG2 cytotoxicity: Cells seeded in 96-well plates (4×10³ cells/well) overnight, treated with N-trans-Feruloyltyramine (20–100 μM) for 48 h. MTT assay as above; IC₅₀ calculated. (3) L02 protection assay: Cells seeded in 96/6-well plates, pretreated with N-trans-Feruloyltyramine (20/40 μM) for 24 h, then 400 μM H₂O₂ for 8 h. Cell viability (MTT), MDA (thiobarbituric acid method, 532 nm), GSH (DTNB method, 412 nm) measured. Western blot for Nrf2 (nuclear protein) and HO-1 (total protein) as in [1] [2] |
| Toxicity/Toxicokinetics |
1. Normal cell toxicity:
(1) SK-N-SH cells: N-trans-Feruloyltyramine (10–40 μM) alone for 24 h showed no cytotoxicity (viability >90% vs. control, p>0.05) [1] (2) L02 cells: N-trans-Feruloyltyramine (up to 100 μM) for 48 h showed no cytotoxicity (viability >85% vs. control, p>0.05) [2] 2. Cancer cell cytotoxicity: N-trans-Feruloyltyramine exhibited dose-dependent cytotoxicity to HepG2 cells (48 h IC₅₀ = 78.5±2.3 μM); viability was (42.3±3.1)% at 100 μM [2] |
| References |
[1]. N-trans-feruloyltyramine Protects Human Neuroblastoma SK-N-SH Cell Line Against H2O2-Induced Cytotoxicity. Natural Product Communications, 2022, 17 (8). [2]. Effects of N-trans-feruloyltyramine isolated from laba garlic on antioxidant, cytotoxic activities and H2O2-induced oxidative damage in HepG2 and L02 cells. Food Chem Toxicol. 2019 Aug:130:130-141. |
| Additional Infomation |
1. Source of N-trans-Feruloyltyramine:
The compound in literature [2] was isolated from laba garlic (traditional fermented garlic, China). Isolation steps: ethanol extraction → macroporous resin purification → HPLC separation. Purity (>98%) confirmed by HPLC and NMR [2] 2. Mechanism of action: (1) In SK-N-SH cells: Protects against H₂O₂ damage by reducing ROS, inhibiting lipid peroxidation (lower MDA), enhancing antioxidant enzymes (SOD, GSH-Px), and regulating apoptotic proteins (up Bcl-2, down Bax/cleaved caspase-3) [1] (2) In L02 cells: Activates Nrf2/HO-1 signaling—promotes Nrf2 nuclear translocation, upregulates HO-1, thereby alleviating oxidative stress [2] 3. Antioxidant selectivity: N-trans-Feruloyltyramine had slightly lower DPPH/ABTS scavenging activity than vitamin C but better selectivity (low toxicity to normal L02 cells, cytotoxic to HepG2 cancer cells) [2] N-feruloyltyramine is a member of tyramines. It has a role as a metabolite. Moupinamide has been reported in Aristolochia kankauensis, Peperomia leptostachya, and other organisms with data available. See also: Tobacco Leaf (part of); Cannabis sativa subsp. indica top (part of); Ipomoea aquatica leaf (part of). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~319.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.98 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1913 mL | 15.9566 mL | 31.9132 mL | |
| 5 mM | 0.6383 mL | 3.1913 mL | 6.3826 mL | |
| 10 mM | 0.3191 mL | 1.5957 mL | 3.1913 mL |