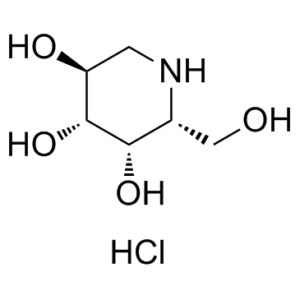
Migalastat hydrochloride (formerly GR-181413A; AT1001; GR181413A; AT-1001; Galafold), the hydrochloride salt of migalastate, is an inhibitor of α-galactosidase A (α-Gal A) and a medication approved by FDA in August 2018 to treat adults with Fabry disease. It is a pharmacological chaperone that potently and selectively binds, stabilizes, and increases cellular levels of α-Gal A with an IC50 of 0.04 μM for human α-Gal A. Oral administration of migalastat HCl reduces tissue GL-3 in Fabry transgenic mice, and in urine and kidneys of some FD patients. Fabry disease (FD) results from mutations in the gene (GLA) that encodes the lysosomal enzyme α-galactosidase A (α-Gal A), and involves pathological accumulation of globotriaosylceramide (GL-3) and globotriaosylsphingosine (lyso-Gb3). Oral administration of migalastat HCl to transgenic mice reduced elevated lyso-Gb3 levels up to 64%, 59%, and 81% in kidney, heart, and skin, respectively, generally equal to or greater than observed for GL-3. Furthermore, baseline plasma lyso-Gb3 levels were markedly elevated in six male FD patients enrolled in Phase 2 studies. Oral administration of migalastat HCl (150 mg QOD) reduced urine GL-3 and plasma lyso-Gb3 in three subjects (range: 15% to 46% within 48 weeks of treatment). In contrast, three showed no reductions in either substrate. These results suggest that measurement of tissue and/or plasma lyso-Gb3 is feasible and may be warranted in future studies of migalastat HCl or other new potential therapies for FD.
Physicochemical Properties
| Molecular Formula | C6H13NO4.HCL |
| Molecular Weight | 199.63266 |
| Exact Mass | 199.061 |
| CAS # | 75172-81-5 |
| Related CAS # | Migalastat;108147-54-2 |
| PubChem CID | 11644097 |
| Appearance | White to light brown solid powder |
| Boiling Point | 382.7ºC at 760 mmHg |
| Melting Point | 260ºC |
| Flash Point | 185.2ºC |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 132 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | C1[C@@H]([C@H]([C@H]([C@H](N1)CO)O)O)O.Cl |
| InChi Key | ZJIHMALTJRDNQI-OLALXQGDSA-N |
| InChi Code | InChI=1S/C6H13NO4.ClH/c8-2-3-5(10)6(11)4(9)1-7-3;/h3-11H,1-2H2;1H/t3-,4+,5+,6-;/m1./s1 |
| Chemical Name | (2R,3S,4R,5S)-2-(hydroxymethyl)piperidine-3,4,5-triol hydrochloride. |
| Synonyms | 1-Deoxygalactonojirimycin hydrochloride; AT1001 HCl; AT 1001; AT-1001; GR181413A; GR 181413A; GR-181413A; 1,5-Dideoxy-1,5-imino-D-galactitol; 1-Deoxygalactonojirimycin; 1-Deoxygalactostatin; Amigal; Migalastat; trade name: Galafold |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Regarding human lysosomal a-Gal A, migasalstat hydrochloride (GR181413A) exhibits an IC50 and Ki value of 0.04 μM [1]. |
| ln Vitro | Regarding human lysosomal a-Gal A, migasalstat hydrochloride (GR181413A) exhibits an IC50 and Ki value of 0.04 μM [1]. |
| ln Vivo |
Insufficient α-galactosidase A activity is the cause of Fabry disease, a latent X-linked illness [2]. In splenic points expressing human mutant α-Gal A (TgM), migalastat (passage, 3 mg/kg daily for 4 weeks) increases cardiac, renal, and cardiac α-Gal A activity at dose and time of illumination [2]. Upon stopping Migalastat for two weeks, half-lives of all major concerns were less than one day [2]. Migalastat lowers FXR, cardiac, and skin solution-GB3 levels by 64%, 59%, and 81%, respectively, when taken lateral (100 mg/kg daily for 28 days) [3]. In a transgenic mouse model of Fabry disease (hR301Q α-Gal A Tg/KO mice), oral administration of Migalastat HCl (100 mg/kg ad libitum in drinking water) for 28 days significantly reduced tissue levels of the disease biomarker globotriaosylsphingosine (lyso-Gb₃). With daily administration, lyso-Gb₃ reductions (mean ± standard error) were: -27±7% in kidney, -40±6% in heart, and -67±6% in skin. With a less-frequent “4 days on/3 days off” regimen, greater reductions were achieved: -59±5% in kidney, -64±3% in heart, and -81±1% in skin. Reductions in tissue levels of the primary substrate globotriaosylceramide (GL-3) were also observed and generally followed a similar pattern, although kidney GL-3 reduction was less pronounced than kidney lyso-Gb₃ reduction. In a subset of male Fabry disease patients from Phase 2 clinical studies, oral administration of Migalastat HCl (150 mg every other day) for up to 48 weeks reduced plasma lyso-Gb₃ levels in 3 out of 6 patients, with reductions ranging from -15% to -46%. These three patients also showed reductions in urine GL-3. The other three patients showed no reduction in plasma lyso-Gb₃ or urine GL-3. [3] |
| Cell Assay |
Cell Viability Assay [4] Cell Types: EHK Cells Mutated α-Gal A Tested Concentrations: 10 μM Incubation Duration: 9 Days Experimental Results: Gb3 accumulation and lysosomal volume reduction. |
| Animal Protocol |
Animal/Disease Models: Male non-transgenic (non-transgenic) Tg)C57BL/6 mice; transgenic mice expressing human mutant R301Q α-Gal A (TgM), α-Gal A knockout mice (KO), on null background Mice expressing human R301Q α-Gal A (TgM/KO)[2] Doses: 3 mg/kg Route of Administration: po (oral gavage); one time/day for 4 weeks Experimental Results: Triacylceramide (Gb3) in mouse kidneys storage is Dramatically diminished. Transgenic mice expressing a human mutant α-Gal A (R301Q) on a GLA knock-out background (hR301Q α-Gal A Tg/KO mice) were used. Migalastat HCl was administered orally ad libitum via drinking water. Dosing solutions were prepared fresh weekly, with the concentration adjusted based on the average daily water consumption of the mice (~5 mL/day per mouse) to achieve the target dose of 100 mg/kg (free-base equivalent). Two dosing regimens were tested: 1) Daily administration for 28 consecutive days. 2) Intermittent administration (“4 on/3 off”): drug in drinking water for four consecutive days followed by three consecutive days of plain water, repeated for a total of 28 days. At the end of the treatment period, mice were euthanized. Blood was collected from the inferior vena cava into lithium heparin tubes, and plasma was separated by centrifugation. Tissues (heart, kidney, brain, skin) were rapidly excised, rinsed in cold phosphate-buffered saline, blotted dry, and stored for analysis. Tissue homogenates were prepared by homogenizing tissue in deionized water using a homogenizer with lysing matrix. [3] |
| ADME/Pharmacokinetics |
Migalastat HCl is administered orally. The study describes oral administration to mice via drinking water and to human patients as an oral capsule (150 mg every other day). [3] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the use of migalastat during breastfeeding. Because no information is available on the use of migalastat during breastfeeding caution should be used, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. In the referenced Phase 2 clinical studies involving male Fabry disease patients, oral administration of Migalastat HCl (150 mg every other day for up to 48 weeks) was generally well-tolerated. [3] |
| References |
[1]. In vitro inhibition and intracellular enhancement of lysosomal alpha-galactosidase A activity in Fabry lymphoblasts by 1-deoxygalactonojirimycin and its derivatives. Eur J Biochem. 2000 Jul;267(13):4179-86. [2]. Preclinical efficacy and safety of 1-deoxygalactonojirimycin in mice for Fabry disease. J Pharmacol Exp Ther. 2009 Mar;328(3):723-31. [3]. Migalastat HCl reduces globotriaosylsphingosine (lyso-Gb3) in Fabry transgenic mice and in the plasma of Fabry patients. PLoS One. 2013;8(3):e57631. [4]. Glucosylceramide synthase inhibition with lucerastat lowers globotriaosylceramide and lysosome staining in cultured fibroblasts from Fabry patients with different mutation types. Hum Mol Genet. 2018 Oct. 27(19):3392-3403. |
| Additional Infomation |
See also: Migalastat (has active moiety). Drug Indication Galafold is indicated for long-term treatment of adults and adolescents aged 16 years and older with a confirmed diagnosis of Fabry disease (α-galactosidase A deficiency) and who have an amenable mutation. Treatment of Fabry disease Migalastat HCl (also known as 1-deoxygalactonojirimycin HCl, AT1001, GR181413A) is an investigational, orally available pharmacological chaperone for the treatment of Fabry disease, a lysosomal storage disorder caused by deficiencies in α-galactosidase A (α-Gal A). Its mechanism of action involves selectively binding to and stabilizing specific mutant forms of α-Gal A in the endoplasmic reticulum, facilitating their proper folding and trafficking to the lysosome, thereby increasing functional enzyme levels and promoting clearance of accumulated substrates (GL-3 and lyso-Gb₃). This study developed a liquid chromatography-tandem mass spectrometry (LC-MS/MS) method for quantifying lyso-Gb₃ in mouse tissues and human plasma and demonstrated for the first time that Migalastat HCl can reduce lyso-Gb₃ levels in a Fabry disease mouse model and in the plasma of some Fabry patients. The reduction of lyso-Gb₃, a sensitive disease biomarker, generally paralleled or exceeded the reduction of the classical substrate GL-3 in mouse tissues, suggesting lyso-Gb₃ may be a useful biomarker for monitoring treatment response. [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (500.93 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.0093 mL | 25.0463 mL | 50.0927 mL | |
| 5 mM | 1.0019 mL | 5.0093 mL | 10.0185 mL | |
| 10 mM | 0.5009 mL | 2.5046 mL | 5.0093 mL |