Physicochemical Properties
| Molecular Formula | C13H27CLN2O3 |
| Molecular Weight | 294.818083047867 |
| Exact Mass | 294.171 |
| Elemental Analysis | C, 52.96; H, 9.23; Cl, 12.02; N, 9.50; O, 16.28 |
| CAS # | 6491-83-4 |
| Related CAS # | 16689-14-8 (HBr) |
| PubChem CID | 15598024 |
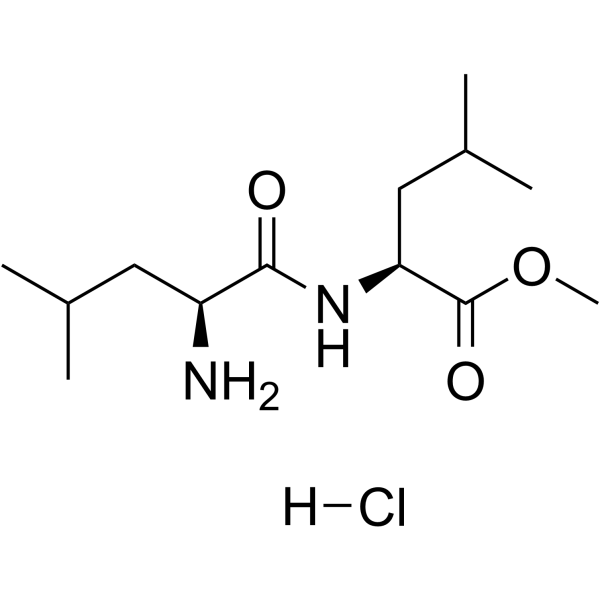
| Sequence | H-Leu-Leu-OMe.HCl; L-leucyl-L-leucine methyl ester hydrochloride |
| SequenceShortening | LL |
| Appearance | Typically exists as white to off-white solids at room temperature |
| Melting Point | 171-181 °C |
| LogP | 2.956 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 19 |
| Complexity | 277 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | Cl.O=C([C@H](CC(C)C)N)N[C@H](C(=O)OC)CC(C)C |
| InChi Key | RVXQFTNCULOWRV-ACMTZBLWSA-N |
| InChi Code | InChI=1S/C13H26N2O3.ClH/c1-8(2)6-10(14)12(16)15-11(7-9(3)4)13(17)18-5;/h8-11H,6-7,14H2,1-5H3,(H,15,16);1H/t10-,11-;/m0./s1 |
| Chemical Name | methyl (2S)-2-[[(2S)-2-amino-4-methylpentanoyl]amino]-4-methylpentanoate;hydrochloride |
| Synonyms | 6491-83-4; H-LEU-LEU-OME HCL; Leu-Leu-ome HCl; H-Leu-leu-OMe hydrochloride; Leu-Leu-ome hydrochloride; Methyl leucylleucinate HCl; L-Leucyl-L-Leucine methyl ester (hydrochloride); 76R0TP2LNW; 76R0TP2LNW; Methyl leucylleucinate hydrochloride; UNII-76R0TP2LNW; L-Leucine, L-leucyl-, methyl ester, hydrochloride (1:1); ...; 6491-83-4; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | LRRK2-mediated Rab10 and Rab12 phosphorylation |
| ln Vitro | In MEFs and A549 cells, LRRK2-mediated Rab10 and Rab12 phosphorylation is enhanced by L-leucyl-L-leucine methyl ester (1 mM; 0.5-2 h)[3]. CD4-lymphocytes convert L-leucyl-L-leucine methyl ester (10-250 μM; 15 min) into an insoluble CCI3COOH product[2]. |
| Enzyme Assay | Mutations that enhance LRRK2 protein kinase activity cause inherited Parkinson's disease. LRRK2 phosphorylates a group of Rab GTPase proteins, including Rab10 and Rab12, within the effector-binding switch-II motif. Previous work has indicated that the PARK16 locus, which harbors the gene encoding for Rab29, is involved in Parkinson's, and that Rab29 operates in a common pathway with LRRK2. Co-expression of Rab29 and LRRK2 stimulates LRRK2 activity by recruiting LRRK2 to the surface of the trans Golgi network. Here, we report that knock-out of Rab29 does not influence endogenous LRRK2 activity, based on the assessment of Rab10 and Rab12 phosphorylation, in wild-type LRRK2, LRRK2[R1441C] or VPS35[D620N] knock-in mouse tissues and primary cell lines, including brain extracts and embryonic fibroblasts. We find that in brain extracts, Rab12 phosphorylation is more robustly impacted by LRRK2 inhibitors and pathogenic mutations than Rab10 phosphorylation. Transgenic overexpression of Rab29 in a mouse model was also insufficient to stimulate basal LRRK2 activity. We observed that stimulation of Rab10 and Rab12 phosphorylation induced by agents that stress the endolysosomal system (nigericin, monensin, chloroquine and LLOMe) is suppressed by LRRK2 inhibitors but not blocked in Rab29 deficient cells. From the agents tested, nigericin induced the greatest increase in Rab10 and Rab12 phosphorylation (5 to 9-fold). Our findings indicate that basal, pathogenic, as well as nigericin and monensin stimulated LRRK2 pathway activity is not controlled by Rab29. Further work is required to establish how LRRK2 activity is regulated, and whether other Rab proteins can control LRRK2 by targeting it to diverse membranes[3]. |
| Cell Assay | L-Leucyl-L-leucine methyl ester (Leu-Leu-OMe), a dipeptide condensation product of L-leucine methyl ester generated by human monocytes (M phi) or polymorphonuclear leukocytes, eliminates all natural killer cell (NK) function from mixed lymphocyte populations. In the present studies, the specificity of the action of Leu-Leu-OMe was examined. It was found that a variety of tissue culture cells and tumor lines of nonlymphoid origin were completely resistant to any demonstrable Leu-Leu-OMe-mediated toxicity. Furthermore, the erythroleukemia line K562, the T cell line Molt-4, the B cell lines HS-Sultan and Daudi, and EBV-transformed B cell lines were unaffected by concentrations of this compound that completely eliminated NK cells. Similarly, the vast majority of OKT4+ lymphocytes manifested no significant toxicity after Leu-Leu-OMe exposure. Furthermore, they retained the capacity to proliferate normally in response to allogeneic cells as well as the ability to provide help for the generation of immunoglobulin-secreting cells (ISC). However, Leu-Leu-OMe caused partial depletion of OKT8+ cells from mixed populations of lymphocytes. After such exposure, the remaining OKT8+ cells were still capable of proliferating in mixed lymphocyte cultures, but the suppressive effect of these cells on ISC generation was abolished. Furthermore, both precursors and activated effectors of cytotoxic T lymphocyte (CTL) and activated NK-like activity generated in mixed lymphocyte cultures were eliminated by exposure to low concentrations of Leu-Leu-OMe. Indeed, both OKT4+ and OKT8+ CTL were eliminated by Leu-Leu-OMe. In addition, both peripheral blood M phi and U937 cells, a human cell line with many M phi-like characteristics, were sensitive to Leu-Leu-OMe-mediated toxicity, although only at two- to fivefold higher concentrations than those completely eliminating NK cells. These findings indicate that Leu-Leu-OMe has selective toxicity for NK cells, CTL, and M phi without adverse effects on a variety of other lymphoid or nonlymphoid cell types[1]. |
| References |
[1]. The immunosuppressive activity of L-leucyl-L-leucine methyl ester: selective ablation of cytotoxic lymphocytes and monocytes. J Immunol. 1986 Feb 1;136(3):1038-48. [2]. Mechanism of L-leucyl-L-leucine methyl ester-mediated killing of cytotoxic lymphocytes: dependence on a lysosomal thiol protease, dipeptidyl peptidase I, that is enriched in these cells. Proc Natl Acad Sci U S A. 1990 Jan;87(1):83-7. [3]. Endogenous Rab29 does not impact basal or stimulated LRRK2 pathway activity. Biochem J. 2020 Nov 27;477(22):4397-4423. |
| Additional Infomation | Exposure of murine or human lymphocytes to L-leucyl-L-leucine methyl ester (Leu-Leu-OMe) results in selective killing of cytotoxic lymphocytes, whereas helper T cells and B cells remain functionally intact. Cytolytic lymphocytes incubated in the presence of toxic concentrations of Leu-Leu-OMe were found to contain membranolytic metabolites of the structure (Leu-Leu)n-OMe, where n greater than or equal to 3. The sensitivity of cytotoxic lymphocytes to Leu-Leu-OMe was found to be dependent upon production of these metabolites by a lysosomal thiol protease, dipeptidyl peptidase I, which is present at far higher levels in cytotoxic lymphocytes than in cells without cytolytic potential or not of bone marrow origin. Thus, this granule enzyme is required for the unique effects of Leu-Leu-OMe and may provide a target for the development of other immunotherapeutic agents designed to delete cytotoxic lymphocyte responses.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~847.98 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.06 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.06 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (7.06 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3919 mL | 16.9595 mL | 33.9190 mL | |
| 5 mM | 0.6784 mL | 3.3919 mL | 6.7838 mL | |
| 10 mM | 0.3392 mL | 1.6960 mL | 3.3919 mL |