Physicochemical Properties
| Molecular Formula | C13H20O3 |
| Molecular Weight | 224.2961 |
| Exact Mass | 224.141 |
| CAS # | 1211-29-6 |
| PubChem CID | 5281929 |
| Appearance | Colorless liquid |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 302.9±15.0 °C at 760 mmHg |
| Melting Point | 25 °C |
| Flash Point | 128.6±20.4 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.469 |
| LogP | 2.12 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 16 |
| Complexity | 281 |
| Defined Atom Stereocenter Count | 2 |
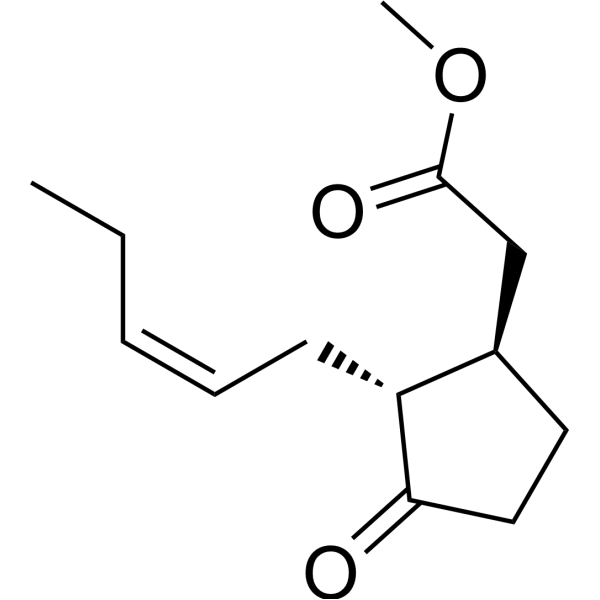
| SMILES | CC/C=C\C[C@@H]1[C@H](CCC1=O)CC(=O)OC |
| InChi Key | GEWDNTWNSAZUDX-WQMVXFAESA-N |
| InChi Code | InChI=1S/C13H20O3/c1-3-4-5-6-11-10(7-8-12(11)14)9-13(15)16-2/h4-5,10-11H,3,6-9H2,1-2H3/b5-4-/t10-,11-/m1/s1 |
| Chemical Name | methyl 2-[(1R,2R)-3-oxo-2-[(Z)-pent-2-enyl]cyclopentyl]acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Toxicity/Toxicokinetics |
Interactions /Investigators/ studied the effects of methyl jasmonate in combination with sucrose on defense-related gene expression, stilbene and anthocyanin production in grapevine cell suspensions. The methyl jasmonate/sucrose treatment was effective in stimulating phenylalanine ammonia lyase, chalcone synthase, stilbene synthase, UDP-glucose: flavonoid-O-glucosyltransferase, proteinase inhibitor and chitinase gene expression, and triggered accumulation of both piceids and anthocyanins in cells, and trans-resveratrol and piceids in the extracellular medium... Capsicum annuum /(C. annuum)/ suspension cell cultures were used to evaluate the effect of cyclodextrins and methyl jasmonate as elicitors of defense responses. The induced defense responses included the accumulation of sesquiterpenes and phytosterols and the activation of pathogenesis-related proteins, leading to reinforcement and modification of the cell wall architecture during elicitation and protection cells against biotic stress. The results showed that the addition of both cyclodextrins and methyl jasmonate induced the biosynthesis of two sesquiterpenes, aromadendrene and solavetivone. This response was clearly synergistic since the increase in the levels of these compounds was much greater in the presence of both elicitors than when they were used separately. The biosynthesis of phytosterols was also induced in the combined treatment, as the result of an additive effect. Likewise, the exogenous application of methyl jasmonate induced the accumulation of pathogenesis-related proteins. The analysis of the extracellular proteome showed the presence of amino acid sequences homologous to PR1 and 4, NtPRp27-like proteins and class I chitinases, peroxidases and the hydrolytic enzymes LEXYL1 and 2, arabinosidases, pectinases, nectarin IV and leucin-rich repeat protein, which suggests that methyl jasmonate plays a role in mediating defense-related gene product expression in C. annuum. Apart from these methyl jamonate-induced proteins, other PR proteins were found in both the control and elicited cell cultures of C. annuum. These included class IV chitinases, beta-1,3-glucanases, thaumatin-like proteins and peroxidases, suggesting that their expression is mainly constitutive since they are involved in growth, development and defense processes. Boron is an essential plant micronutrient, but it is phytotoxic if present in excessive amounts in soil for certain plants such as Artemisia annua L. /(A. annua)/ that contains artemisinin (an important antimalarial drug) in its areal parts. Artemisinin is a sesquiterpene lactone with an endoperoxide bridge... the present research was conducted to determine whether the exogenous application of methyl jasmonate (MeJA) could combat the ill effects of excessive /Boron stress/ (B) present in the soil. According to the results obtained, the B toxicity induced oxidative stress and reduced the stem height as well as fresh and dry masses of the plant remarkably. The excessive amounts of soil B also lowered the net photosynthetic rate, stomatal conductance, internal CO2 concentration and total chlorophyll content in the leaves. In contrast, the foliar application of MeJA enhanced the growth and photosynthetic efficiency both in the stressed and non-stressed plants. The excessive B levels also increased the activities of antioxidant enzymes, such as catalase, peroxidase and superoxide dismutase... the MeJA application to the stressed plants reduced the amount of lipid peroxidation and stimulated the synthesis of antioxidant enzymes, enhancing the content and yield of artemisinin as well. Thus, it was concluded that MeJA might be utilized in mitigating the B toxicity and improving the content and yield of artemisinin in A. annua plant. |
| References |
[1]. The Anti-Proliferative and Anti-Invasive Effect of Leaf Extracts of Blueberry Plants Treated with Methyl Jasmonate on Human Gastric Cancer In Vitro Is Related to Their Antioxidant Properties. Antioxidants (Basel). 2020;9(1):45. Published 2020 Jan 4. |
| Additional Infomation |
(-)-methyl jasmonate is a jasmonate ester that is the methyl ester of jasmonic acid. It has a role as a member of jasmonates, a plant metabolite and a plant hormone. It is a jasmonate ester, a methyl ester and a member of Jasmonate derivatives. Methyl jasmonate has been reported in Solanum tuberosum, Tripterygium wilfordii, and other organisms with data available. Mechanism of Action Using the tomato pathotype of Alternaria alternata (Aa) and its AAL-toxin/tomato interaction as a model system, /the authors/ demonstrate a possible role for /jasmonic acid/ JA in susceptibility of plants against pathogens, which utilize host-specific toxins as virulence effectors. Disease development and in planta growth of the tomato pathotype of Aa were decreased in the def1 mutant, defective in biosynthesis of JA, compared with the wild-type (WT) cultivar. Exogenous methyl jasmonate (MeJA) application restored pathogen disease symptoms to the def1 mutant and led to increased disease in the WT. On the other hand, necrotic cell death was similarly induced by AAL-toxin both on def1 and WT, and MeJA application to the tomatoes did not affect the degree of cell death by the toxin. These results indicate that the JA-dependent signaling pathway is not involved in host basal defense responses against the tomato pathotype of Aa, but rather might affect pathogen acceptability via a toxin-independent manner. Data further suggest that JA has a promotional effect on susceptibility of tomato to toxigenic and necrotrophic pathogens, such that pathogens might utilize the JA signaling pathway for successful infection. ...WRKY plant-specific transcription factors, as one of the flagellin-inducible genes in /its non-host/ A. thaliana. Expression of WRKY41 is induced by inoculation with the incompatible pathogen P. syringae pv. tomato DC3000 (Pto) possessing AvrRpt2 and the non-host pathogens... Arabidopsis overexpressing WRKY41 showed enhanced resistance to the Pto wild-type but increased susceptibility to Erwinia carotovora EC1. WRKY41-overexpressing Arabidopsis constitutively expresses the PR5 gene, but suppresses the methyl jasmonate-induced PDF1.2 gene expression. These results demonstrate that WRKY41 may be a key regulator in the cross talk of salicylic acid and jasmonic acid pathways. Induction of cell death is an important component of plant defense against pathogens. There have been many reports on the role of phytohormones in pathogen-induced cell death, but jasmonic acid (JA) has not been implicated as a regulator of the response. Here, /investigators/ report the function of NbHB1, Nicotiana benthamiana homeobox1, in pathogen-induced cell death in connection with JA signaling. Involvement of NbHB1 in cell death was analyzed by gain- and loss-of-function studies using Agrobacterium-mediated transient overexpression and virus-induced gene silencing, respectively. Expression of NbHB1 following pathogen inoculations and various treatments was monitored by reverse transcription polymerase chain reaction. Transcript levels of NbHB1 were upregulated by infection with virulent and avirulent bacterial pathogens. Ectopic expression of NbHB1 accelerated cell death following treatment with darkness, methyl jasmonate, or pathogen inoculation. Conversely, when NbHB1 was silenced, pathogen-induced cell death was delayed. NbHB1-induced cell death was also delayed by silencing of NbCOI1, indicating a requirement for JA-mediated signaling. Overexpression of the domain-deleted proteins of NbHB1 revealed that the homeodomain, leucine zipper, and part of the variable N-terminal region were necessary for NbHB1 functionality. These results strongly suggest the role of NbHB1 in pathogen-induced plant cell death via the JA-mediated signaling pathway. In this study, /the authors/ employed high throughput Illumina sequencing to identify miRNAs from Taxus chinensis (T. chinensis) cells to investigate the effect of the taxoid elicitor methyl jasmonate (MJ) on miRNA expression. In a dataset of approximately 6.6 million sequences, a total of 58 miRNAs, belonging to 25 families were identified. A majority of them are conserved between angiosperms and gymnosperms. However, two miRNAs (miR1310 and miR1314) appear gymnosperm-specific, with miR1314 likely to exist as a cluster. MJ treatment significantly affected the expression of specific miRNAs; 14 miRNAs from 7 different families (miR156, miR168, miR169, miR172, miR396, miR480 and mir1310) were down regulated whereas 3 miRNAs from 2 families (miR164 and miR390) were up regulated. For more Mechanism of Action (Complete) data for Methyl Jasmonate (13 total), please visit the HSDB record page. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.4583 mL | 22.2916 mL | 44.5831 mL | |
| 5 mM | 0.8917 mL | 4.4583 mL | 8.9166 mL | |
| 10 mM | 0.4458 mL | 2.2292 mL | 4.4583 mL |