Physicochemical Properties
| Molecular Formula | C14H4N2NA2O8 |
| Molecular Weight | 374.1697 |
| Exact Mass | 373.976 |
| CAS # | 122628-50-6 |
| Related CAS # | Pyrroloquinoline quinone;72909-34-3 |
| PubChem CID | 3078772 |
| Appearance | Pink to red solid powder |
| Boiling Point | 1018.6ºC at 760mmHg |
| Flash Point | 569.8ºC |
| Vapour Pressure | 0mmHg at 25°C |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 26 |
| Complexity | 636 |
| Defined Atom Stereocenter Count | 0 |
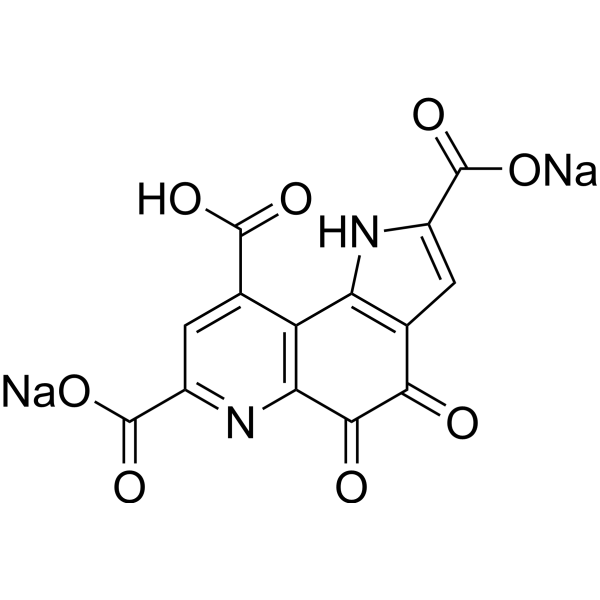
| SMILES | [Na+].[Na+].O=C1C(C2=C(C(C(=O)[O-])=C([H])C(C(=O)[O-])=N2)C2=C1C([H])=C(C(=O)O[H])N2[H])=O |
| InChi Key | UFVBOGYDCJNLPM-UHFFFAOYSA-L |
| InChi Code | InChI=1S/C14H6N2O8.2Na/c17-10-4-2-6(14(23)24)15-8(4)7-3(12(19)20)1-5(13(21)22)16-9(7)11(10)18;;/h1-2,15H,(H,19,20)(H,21,22)(H,23,24);;/q;2*+1/p-2 |
| Chemical Name | disodium;2-carboxy-4,5-dioxo-1H-pyrrolo[2,3-f]quinoline-7,9-dicarboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Methoxatin (Pyrroloquinolinequinone, PQQ) is identified as the tightly bound, likely covalent, organic cofactor in copper-containing amine oxidases (EC 1.4.3.6). [1] Methoxatin (Pyrroloquinolinequinone, PQQ) is suggested to be a component, possibly a free agent, involved in the NADPH oxidase system that drives superoxide (O₂⁻) production in phagocytes (e.g., neutrophils). Its exact role within the multicomponent oxidase complex is not fully stipulated. [2] |
| ln Vitro |
Pyrroloquinoline quinone disodium salt promotes worsening of immunological response, hair loss, slumped posture and susceptibility to aortic aneurysm in pups born and nuts in maternal rats [2]. Methoxatin (PQQ) can catalyze redox cycling in vitro. In the presence of an excess reductant such as glycinate at physiological pH and under aerobic conditions, PQQ oxidizes NADPH to NADP⁺, itself being reduced to its semiquinone form (PQQH⁻), which then reduces oxygen to superoxide (O₂⁻). This suggests PQQ could function as an NADPH oxidase that donates electrons directly to oxygen. [2] The organic cations diphenyleneiodonium (DPI) and diphenyliodonium (BPI), as well as the aromatic o-diamine 4,5-dimethylphenylenediamine (DIMPDA), were shown to sequester synthetic PQQ and inhibit its redox-cycling activity in a model nitroblue tetrazolium (NBT) assay. The concentration of DIMPDA required to inhibit the redox cycling ability of a 33 nM PQQ solution by 50% was 100 µM. The concentrations of iodonium compounds required for 50% inhibition were 10 µM for BPI and 1.5 µM for DPI. [2] |
| Enzyme Assay |
The study employed resonance Raman spectroscopy to probe the structure of the organic cofactor in bovine plasma amine oxidase (BPAO). The enzyme was derivatized with 2,4-dinitrophenylhydrazine (DNPH) by adding a 10% molar excess of DNPH (dissolved in absolute ethanol) to the enzyme in 0.1 M potassium phosphate buffer (pH 7.2) and incubating at room temperature for 16 hours. Protein concentrations ranged from 0.07 to 0.11 mM. The resonance Raman spectra of the resulting BPAO-DNPH adduct were then measured and compared in detail to the spectra of DNPH derivatives of model compounds including authentic methoxatin (PQQ), pyridoxal, 1,10-phenanthroline-5,6-dione, 1,7-phenanthroline-5,6-dione, and 2-pyridinecarboxaldehyde. The excellent match between the vibrational frequencies of the BPAO-DNPH and PQQ-DNPH spectra, particularly in fingerprint regions (300-600 cm⁻¹, 660-800 cm⁻¹, and 1300-1700 cm⁻¹), provided strong evidence that the cofactor is PQQ or a very similar derivative. [1] A glycinate-nitroblue tetrazolium (NBT) assay was used to measure PQQ levels and assess redox-cycling activity. The assay mixture contained the sample to be analyzed, reduced bovine serum albumin, and NBT in a 2 M glycine solution (pH 10). The total reaction volume was 1.5 ml. The mixture was incubated at 25°C for 1 hour in the dark. In this system, PQQ oxidizes glycine, and the reduced PQQ reduces oxygen to superoxide, which then reduces NBT to a purple diformazan. The amount of formazan formed, measured at 550 nm and quantified against a standard curve, amplifies the signal, allowing detection of picomolar levels of PQQ. This assay was also used to demonstrate inhibition of PQQ redox cycling by DPI, BPI, and DIMPDA. [2] |
| Cell Assay |
The standard ferricytochrome c assay was used to measure superoxide (O₂⁻) production by phorbol myristate acetate (PMA)-stimulated guinea pig neutrophils. Briefly, 900 µl of phosphate-buffered saline with glucose, calcium, and magnesium was added to 100 µl of cell suspension (3 x 10⁷ cells). Superoxide dismutase (SOD, 2 µg/ml) was added to control tubes. Inhibitors (DPI, BPI, DIMPDA) and/or PQQ were added, and the mixture was incubated for 5 min at 37°C. Cytochrome c solution was then added, followed by stimulation with PMA (5 µl of a 1 mg/ml solution in DMSO) for 5 min. Superoxide production was measured as the SOD-sensitive reduction of ferricytochrome c. [2] To investigate the presence of PQQ in cells, guinea pig neutrophils were incubated with tritium-labeled inhibitors ([³H]DIMPDA or [³H]DPI). Cells (1 x 10⁸) suspended in PBS with calcium, magnesium, and glucose were incubated with 1 mM [³H]DIMPDA or 5-10 µM [³H]DPI for 5 min at 37°C, followed by stimulation with PMA for 90 s. Cells were then cooled, washed, and processed for adduct isolation. [2] For the isolation of PQQ-inhibitor adducts from cells, the treated and washed cells were sonicated. The sonicate was ultracentrifuged. The supernatant was passed through an ultrafilter, and the ultrafiltrate was extracted with water-saturated ethyl acetate. The aqueous layer, containing the water-soluble adduct, was saved for HPLC analysis. [2] |
| Animal Protocol |
Guinea pig neutrophils were harvested from the peritoneum. Briefly, 30 ml of 12% sodium caseinate was injected intraperitoneally into 500 g Hartley guinea pigs. The peritoneum was opened, and the elicited neutrophils were harvested, rinsed with cold saline, filtered through nylon gauze, and pelleted by centrifugation. Red blood cells were lysed by briefly suspending the pellet in water, followed by immediate restoration to isotonicity with 9% saline. Neutrophils were washed, resuspended in phosphate-buffered saline (PBS), and adjusted to a concentration of 3 x 10⁷ cells/5 ml. Cell preparations were >90% neutrophils with >90% viability by trypan blue exclusion. [2] |
| ADME/Pharmacokinetics |
The text states that previous work (not detailed in this study) showed that [¹⁴C]-PQQ administered to rats was absorbed by the small intestine and excreted in the urine, with the remainder concentrated in red and white blood cells. [2] |
| Toxicity/Toxicokinetics |
The text mentions that mouse pups born to and nursing from PQQ-deprived dams have a compromised immune response, alopecia, a hunched posture, and susceptibility to aortic aneurysms, indicating consequences of dietary deficiency. No data on acute toxicity, LD₅₀, organ toxicity, or drug interactions for administered PQQ are provided in this study. [2] |
| References |
[1]. Evidence for methoxatin (pyrroloquinolinequinone) as the cofactor in bovine plasma amine oxidase from resonance Raman spectroscopy. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8435-9. [2]. Methoxatin (PQQ) in guinea-pig neutrophils. Free Radic Biol Med. 1994 Oct;17(4):311-20. |
| Additional Infomation |
Methoxatin (PQQ) contains electrophilic carbonyl groups and displays one- and two-electron redox chemistry, which is relevant to the catalytic mechanism of amine oxidases where sequential one-electron steps may be involved in reoxidation by O₂. [1] The identification of methoxatin (PQQ) as a cofactor in mammalian enzymes raises the possibility that it may be a previously unrecognized vitamin. [1] The resonance Raman methodology described demonstrates a powerful approach for structural studies of protein-bound chromophores like methoxatin (PQQ) under native-like conditions without requiring isolation of the cofactor. [1] Methoxatin (PQQ) was isolated from guinea pig neutrophils in this study. [2] PQQ is described as an anionic, water-soluble, redox-cycling orthoquinone. In mammalian systems, it appears to be free and dialyzable, having been detected in cell lysates, biological fluids, and eggs. [2] PQQ is stated to be an essential nutrient for mammals and important for immune function. [2] When administered to animals, PQQ has been found to protect against oxidative damage, toxic injury, and the damage caused by ischemia and reperfusion in a rat model of stroke. [2] Inhibitors DPI, BPI, and DIMPDA blocked O₂⁻ release by PMA-stimulated guinea pig neutrophils. This blockade could be released (reversed) by the addition of exogenous PQQ to the cell suspensions. [2] DPI at 1 µM inhibited neutrophil superoxide production by 74%, BPI at 50 µM inhibited by 64%, and DIMPDA at 200 µM inhibited by 50%. The inhibition by DIMPDA and DPI was reversible by washing the cells, and the degree of reversibility correlated with the nature of the inhibitor-PQQ bond (noncovalent for DPI). [2] A [³H]DPI-PQQ adduct and a [³H]DIMPDA-PQQ adduct were isolated from guinea pig neutrophils and characterized by HPLC co-chromatography with authentic standards. Treatment of the isolated [³H]DPI-PQQ adduct with the transchelating agent Tiron broke the adduct and released free PQQ. [2] Based on the recovered radioactivity from the adduct, it was calculated that PQQ was present in the neutrophils at a concentration of about 30 nmol per 10⁸ cells. [2] |
Solubility Data
| Solubility (In Vitro) | H2O : ~1.25 mg/mL (~3.34 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2 mg/mL (5.35 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C). Solubility in Formulation 2: 5 mg/mL (13.36 mM) in 0.5% CMC-Na/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6726 mL | 13.3629 mL | 26.7258 mL | |
| 5 mM | 0.5345 mL | 2.6726 mL | 5.3452 mL | |
| 10 mM | 0.2673 mL | 1.3363 mL | 2.6726 mL |