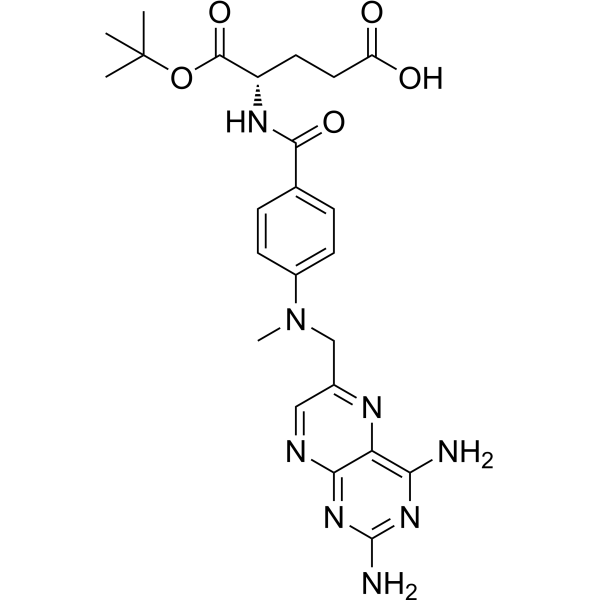
Physicochemical Properties
| Molecular Formula | C24H30N8O5 |
| Molecular Weight | 510.55 |
| Exact Mass | 510.234 |
| CAS # | 79640-70-3 |
| PubChem CID | 12188936 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 3.078 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 37 |
| Complexity | 801 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC(C)(C)OC(=O)[C@H](CCC(=O)O)NC(=O)C1=CC=C(C=C1)N(C)CC2=CN=C3C(=N2)C(=NC(=N3)N)N |
| InChi Key | HTMMDBDGHQUHPW-INIZCTEOSA-N |
| InChi Code | InChI=1S/C24H30N8O5/c1-24(2,3)37-22(36)16(9-10-17(33)34)29-21(35)13-5-7-15(8-6-13)32(4)12-14-11-27-20-18(28-14)19(25)30-23(26)31-20/h5-8,11,16H,9-10,12H2,1-4H3,(H,29,35)(H,33,34)(H4,25,26,27,30,31)/t16-/m0/s1 |
| Chemical Name | (4S)-4-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]-5-[(2-methylpropan-2-yl)oxy]-5-oxopentanoic acid |
| Synonyms | Methotrexate alpha-tert-butyl ester; 79640-70-3; Methotrexate a-tert-Butyl Ester; Methotrexatealpha-tert-ButylEster; Methotrexate ; A-tert-butyl ester; (S)-5-(tert-butoxy)-4-(4-(((2,4-diaminopteridin-6-yl)methyl)(methyl)amino)benzamido)-5-oxopentanoic acid; N-[4-[[(2,4-Diamino-6-pteridinyl)methyl]methylamino]benzoyl]-L-glutamic Acid 1-(1,1-Dimethylethyl) Ester; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | MTX/methotrexate analog; DHFR/dihydrofolate reductase; anticancer |
| ln Vitro | Compared to MTX-PVG, OtBu-MTX-PVG has a smaller HT1080 effect on HT1080 cells. According to the findings, there is less activity in vitro when the MTX α-carboxyl group is protected [1]. |
| ln Vivo | Lys-MTXcleavable- (OtBu) effectively inhibits tumor growth in HT1080 tumor-bearing mice [1]. Protection of the α-carboxyl group of methotrexate can be employed to improve circulation half-life and prevent liver accumulation of comparable MTX-conjugated dendrimers while still keeping anticancer efficacy in vivo [1]. |
| Animal Protocol |
Animal/Disease Models: Female, Balb/c nu/nu, 6 weeks (carrying HT1080 cells) [1] Doses: 30 mg/kg Route of Administration: Once a week for 2 weeks Experimental Results: Demonstrated significant tumor size (63%) Decrease day 12. A generation 5 PEGylated (PEG 1100) polylysine dendrimer, conjugated via a stable amide linker to OtBu protected methotrexate (MTX), was previously shown to have a circulatory half-life of 2 days and to target solid tumors in both rats and mice. Here, we show that deprotection of MTX and substitution of the stable linker with a matrix metalloproteinase (MMP) 2 and 9 cleavable linker (PVGLIG) dramatically increased plasma clearance and promoted deposition in the liver and spleen (50-80% of the dose recovered in the liver 3 days post dose). Similar rapid clearance was also seen using a scrambled peptide suggesting that clearance was not dependent on the cleavable nature of the linker. Surprisingly, dendrimers where OtBu capped MTX was linked to the dendrimer surface via the hexapeptide linker showed equivalent in vitro cytotoxicity against HT1080 cells when compared to the uncapped dendrimer and also retained the long circulating characteristics of the stable constructs. The OtBu capped MTX conjugated dendrimer was subsequently shown to significantly reduce tumor growth in HT1080 tumor bearing mice compared to control. In contrast the equivalent dendrimer comprising uncapped MTX conjugated to the dendrimer via the same hexapeptide linker did not reduce tumor growth, presumably reflecting very rapid clearance of the construct. The results are consistent with the suggestion that protection of the α-carboxyl group of methotrexate may be used to improve the circulatory half-life and reduce the liver accumulation of similar MTX-conjugated dendrimers, while still retaining antitumor activity in vivo.[1] |
| References |
[1]. Capping methotrexate α-carboxyl groups enhances systemic exposure and retains the cytotoxicity of drug conjugated PEGylated polylysine dendrimers. Mol Pharm. 2011;8(2):338-349. [2]. Understanding the mechanisms of action of methotrexate: implications for the treatment of rheumatoid arthritis. Bull NYU Hosp Jt Dis. 2007;65(3):168-73. [3]. Methotrexate in rheumatoid arthritis. Pharmacol Rep. 2006 Jul-Aug;58(4):473-92. [4]. The Effect of L-carnitine on Amethopterin-induced Toxicity in Rat Large Intestine. |
| Additional Infomation |
A variety of disease-modifying antirheumatic drugs (DMARDs) are available to control the clinical activity of rheumatoid arthritis (RA). Methotrexate (MTX), an analogue of folic acid and of aminopterin, is the most commonly used DMARD and is now prescribed worldwide to at least 500,000 patients with RA. The mechanism by which MTX used at a low dose modulates inflammation in RA is still unknown. Monitoring of the therapy in terms of MTX concentration in patients with RA seems not to have a significant influence on the effectiveness of the treatment. Two meta-analyses showed that MTX has one of the best efficacy/toxicity ratios. It should be the first DMARD used in the majority of patients with RA at this time. However, a significant number of patients treated only with MTX fail to achieve optimal disease control, so there are many combinations of DMARD regimes. It is hoped that more aggressive use of conventional DMARDs and biological agents will result in less disability and a higher proportion of patients achieving remission. The therapy of RA is a dynamic process and requires maintaining a delicate balance between benefits and risks. Even with the newer biological agents, MTX continues to serve as a reference point and there is still a role for MTX in the treatment of RA patients.[3] Methotrexate has been widely used for the treatment of rheumatoid arthritis (RA). The mechanisms of action of methotrexate are complex. Developed as a folic acid analogue, methotrexate inhibits purine and pyrimidine synthesis, which accounts for its efficacy in the therapy of cancer as well as for some of its toxicities. Recently, many studies have focused on the adenosine-mediated antiinflammatory effects of methotrexate. Certain aspects of methotrexate toxicities are also attributed to adenosine release. A better understanding of the mechanisms of action and toxicities of methotrexate will direct clinicians in their treatment approach and toxicity monitoring. Toward that objective, the latest developments in the pharmacokinetics, mechanism of action, pharmacogenetics, and toxicity of methotrexate are herein discussed. [2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9587 mL | 9.7934 mL | 19.5867 mL | |
| 5 mM | 0.3917 mL | 1.9587 mL | 3.9173 mL | |
| 10 mM | 0.1959 mL | 0.9793 mL | 1.9587 mL |