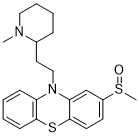
Mesoridazine is a potent phenothiazine dopamine receptor anatagonist that is able to inhibit D2DR and D4DR. It has a high affinity for dopamine D4 receptor. Mesoridazine besylate is an antipsychotic agent.
Physicochemical Properties
| Molecular Formula | C21H26N2OS2 |
| Molecular Weight | 386.57394 |
| Exact Mass | 386.149 |
| CAS # | 5588-33-0 |
| Related CAS # | Mesoridazine benzenesulfonate;32672-69-8 |
| PubChem CID | 4078 |
| Appearance | Oily product |
| Density | 1.3g/cm3 |
| Boiling Point | 570.5ºC at 760mmHg |
| Melting Point | Crystals from ethyl acetate; MP: 115-120 °C /tartrate/ |
| Flash Point | 298.9ºC |
| Index of Refraction | 1.694 |
| LogP | 5.769 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 502 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CN1CCCCC1CCN2C3=CC=CC=C3SC4=C2C=C(C=C4)S(=O)C |
| InChi Key | SLVMESMUVMCQIY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H26N2OS2/c1-22-13-6-5-7-16(22)12-14-23-18-8-3-4-9-20(18)25-21-11-10-17(26(2)24)15-19(21)23/h3-4,8-11,15-16H,5-7,12-14H2,1-2H3 |
| Chemical Name | 10-(2-(1-methylpiperidin-2-yl)ethyl)-2-(methylsulfinyl)-10H-phenothiazine |
| Synonyms | NC-123 NSC 186066 NSC-186066 NSC186066 TPS23 TPS 23 TPS-23 Thioridazine thiomethyl sulfoxide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Mesoridazine inhibits human ether-a-go-go related gene (HERG) currents in a concentration-dependent manner (IC50 = 550 nM, 0 mV). The blocking action increases dramatically along the voltage range where HERG activation occurs, leading to maximal Saturable channel activity when HERG voltage is reached [1]. The total absorption rates of 15.94 ± 4.04% and 39.24 ± 5.11% of mesoridazine (15 mM; 24 hours) in nude mouse and pig skin, respectively, were reported [3]. |
| ln Vivo | Mesoridazine (15 mM) shows strong action and persistent analgesia in preventing cutaneous discomfort when applied topically once or once a day for seven days [3]. Mice in the nude were given a topical application of mesoridazine (15 mM) for six hours. The intradermal concentration of the drug was 0.34 - 0.74 nmol/mg [3]. |
| Animal Protocol |
Animal/Disease Models: 8weeks old female nude mice [3] Doses: 15 mM Route of Administration: Topical administration, once (analgesic test) or one time/day for 7 days (stimulation test) Experimental Results:Shows analgesic effect. A slight increase in transepidermal water loss (TEWL) from 7.8 to 9.9 g/m2/h was observed. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Well absorbed from the gastrointestinal tract. TOTAL URINARY & FECAL EXCRETION BY MONKEYS (10 DAY PERIOD) OF THIORIDAZINE WAS 64-76% & 83-92% FOR MESORIDAZINE (FECAL EXCRETION 2-4 TIMES GREATER). GREATER EXCRETION OF LATTER MAY BE DUE TO LESSER OVERALL TISSUE ADSORPTION OR LESS EXTENSIVE ENTERO-HEPATIC CYCLING. WELL ABSORBED FROM GI TRACT. ONSET & DURATION OF ACTION & METABOLIC FATE... NOT...PRECISELY DETERMINED /HUMAN, ORAL, IM/. IN ANIMAL STUDIES, APPROX 2/3 OF DOSE...EXCRETED IN FECES VIA BILE & 1/3 OF DOSE...EXCRETED IN URINE. /BESYLATE/ PHENOTHIAZINES CROSS PLACENTAL BARRIER & MAY APPEAR IN MILK OF NURSING MOTHERS...PHENOTHIAZINES &...METABOLITES...EXCRETED IN URINE, BILE, & FECES. CERTAIN METABOLITES &...FREE DRUGS...DETECTED IN URINE UP TO 6 MO AFTER THERAPY...DISCONTINUED /HUMAN, ORAL, IM/ PHENOTHIAZINES...ABSORBED WELL FROM GI TRACT & FROM PARENTERAL SITES. GENERALLY...CLEARED FROM PLASMA WITHIN APPROX 3 HR...DISTRIBUTED TO MOST BODY TISSUES...HIGH CONCN OF UNCHANGED DRUG...IN BRAIN...METABOLITES PREDOMINATE IN LUNG, LIVER, KIDNEYS...SPLEEN /HUMAN, ORAL, IM/ Metabolism / Metabolites THIORIDAZINE-2-SULFOXIDE PROBABLY YIELDS THIORIDAZINE-2,5-DISULFOXIDE IN RAT: ZEHNDER, K ET AL, BIOCHEM PHARMAC, 11, 535 (1962). /FROM TABLE/ Thioridazine 2-sulfoxide is a known human metabolite of Thioridazine. Half Life: 24 to 48 hours Biological Half-Life 24 to 48 hours |
| Toxicity/Toxicokinetics |
Toxicity Summary Based upon animal studies, mesoridazine, as with other phenothiazines, acts indirectly on reticular formation, whereby neuronal activity into reticular formation is reduced without affecting its intrinsic ability to activate the cerebral cortex. In addition, the phenothiazines exhibit at least part of their activities through depression of hypothalamic centers. Neurochemically, the phenothiazines are thought to exert their effects by a central adrenergic blocking action. Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because there is no published experience with mesoridazine during breastfeeding, other antipsychotic agents are preferred. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Phenothiazines cause galactorrhea in 26 to 40% of female patients. Hyperprolactinemia appears to be the cause of the galactorrhea. The hyperprolactinemia is caused by the drug's dopamine-blocking action in the tuberoinfundibular pathway. The prolactin level in a mother with established lactation may not affect her ability to breastfeed. Protein Binding 4% Toxicity Data Oral LD50 is 560 ± 62.5 mg/kg and 644 ± 48 mg/kg in mouse and rat, respectively. Interactions CHLORPROMAZINE & OTHER ANTIPSYCHOTIC DRUGS...MAY BLOCK ANTIHYPERTENSIVE EFFECTS OF GUANETHIDINE. /CHLORPROMAZINE/ INTERACTIONS BETWEEN CHLORPROMAZINE & AMPHETAMINE, ALTHOUGH NOT DOCUMENTED, MAY APPLY TO OTHER PHENOTHIAZINE DERIV. /PHENOTHIAZINES/ PRETREATMENT WITH 3 MG/KG OF MESORIDAZINE-HCL ORALLY WAS MORE EFFECTIVE THAN THIORIDAZINE-HCL (6 MG/KG) IN DECR D-AMPHETAMINE SULFATE INDUCED BEHAVIOR IN DOGS. Additive QT interval prolongation may increase the risk of ventricular tachycardia when /probucol is used with phenothiazines/. /Phenothiazines/ For more Interactions (Complete) data for MESORIDAZINE (31 total), please visit the HSDB record page. Non-Human Toxicity Values LD50 MICE INTRAVENOUS 33 MG/KG /BESYLATE/ LD50 MICE SUBCUTANEOUS 611 MG/KG /BESYLATE/ LD50 MICE ORAL 346 MG/KG /BESYLATE/ |
| References |
[1]. Mesoridazine: an open-channel blocker of human ether-a-go-go-related gene K+ channel. J Mol Cell Cardiol. 2004 Jan;36(1):151-60. [2]. Comparison of the effects of thioridazine and mesoridazine on the QT interval in healthy adults after single oral doses. Clin Pharmacol Ther. 2007 Nov;82(5):548-54. [3]. Topically applied mesoridazine exhibits the strongest cutaneous analgesia and minimized skin disruption among tricyclic antidepressants: The skin absorption assessment. Eur J Pharm Biopharm. 2016 Aug;105:59-68. |
| Additional Infomation |
Mesoridazine is a phenothiazine substituted at position 2 (para to the S atom) by a methylsulfinyl group, and on the nitrogen by a 2-(1-methylpiperidin-2-yl)ethyl group. It has a role as a dopaminergic antagonist and a first generation antipsychotic. It is a member of phenothiazines, a sulfoxide and a tertiary amino compound. A phenothiazine antipsychotic with effects similar to chlorpromazine. Mesoridazine is a Phenothiazine. Mesoridazine has been reported in Phomopsis phaseoli with data available. Mesoridazine is only found in individuals that have used or taken this drug. It is a phenothiazine antipsychotic with effects similar to chlorpromazine. [PubChem]Based upon animal studies, mesoridazine, as with other phenothiazines, acts indirectly on reticular formation, whereby neuronal activity into reticular formation is reduced without affecting its intrinsic ability to activate the cerebral cortex. In addition, the phenothiazines exhibit at least part of their activities through depression of hypothalamic centers. Neurochemically, the phenothiazines are thought to exert their effects by a central adrenergic blocking action. A phenothiazine antipsychotic with effects similar to CHLORPROMAZINE. Drug Indication Used in the treatment of schizophrenia, organic brain disorders, alcoholism and psychoneuroses. Mechanism of Action Based upon animal studies, mesoridazine, as with other phenothiazines, acts indirectly on reticular formation, whereby neuronal activity into reticular formation is reduced without affecting its intrinsic ability to activate the cerebral cortex. In addition, the phenothiazines exhibit at least part of their activities through depression of hypothalamic centers. Neurochemically, the phenothiazines are thought to exert their effects by a central adrenergic blocking action. ...MECHANISM OF ACTION OF ANTIPSYCHOTIC DRUGS WITH RESPECT TO THERAPEUTIC EFFICACY & SIDE EFFECTS MAY RELATE TO INHIBITION OF DOPAMINE ACTIVATION OF ADENYLATE CYCLASE. /PHENOTHIAZINES/ Therapeutic Uses Antipsychotic Agents, Phenothiazine; Dopamine Antagonists THE ANTIPSYCHOTIC DRUGS HAVE A HIGH THERAPEUTIC INDEX & ARE REMARKABLY SAFE AGENTS. FURTHERMORE, MOST PHENOTHIAZINES HAVE RELATIVELY FLAT DOSE-RESPONSE CURVE AND CAN BE USED OVER WIDE RANGE OF DOSAGES. ...SIDE EFFECTS ARE OFTEN EXTENSIONS OF MANY PHARMACOLOGICAL ACTIONS OF DRUGS... /PHENOTHIAZINES/ ...INDICATED FOR MGMNT OF SCHIZOPHRENIA, ORG BRAIN DISORDERS, SYMPTOMS OF ALC WITHDRAWAL, & PSYCHONEUROSES. CLINICAL STUDIES TO DATE INDICATE THAT MESORIDAZINE BESYLATE HAS LOW INCIDENCE OF ADVERSE REACTIONS COMPARED WITH OTHER PHENOTHIAZINES. /BESYLATE/ 8 WK THERAPY WITH THIORIDAZINE & MESORIDAZINE BESYLATE. THIORIDAZINE TREATED PT WERE SUPERIOR TO LATTER TREATED PT. IMPLICATIONS FOR CHEMOTHERAPY OF SCHIZOPHRENICS CONSIDERED. For more Therapeutic Uses (Complete) data for MESORIDAZINE (6 total), please visit the HSDB record page. Drug Warnings NEUROLEPTIC AGENTS ... SHOULD BE USED WITH EXTREME CAUTION, IF @ ALL, IN UNTREATED EPILEPTIC PT & IN PT UNDERGOING WITHDRAWAL FROM CENTRAL DEPRESSANT DRUGS SUCH AS ALCOHOL, BARBITURATES, OR BENZODIAZEPINES. MOST ANTIPSYCHOTIC DRUGS ... CAN BE USED SAFELY IN EPILEPTICS IF MODERATE DOSES ARE ATTAINED GRADUALLY AND IF CONCOMITANT ANTICONVULSANT DRUG THERAPY IS MAINTAINED. /PHENOTHIAZINES/ PHENOTHIAZINES INHIBIT EJACULATION WITHOUT INTERFERING WITH ERECTION. /PHENOTHIAZINES/ WT GAIN & INCR IN APPETITE OCCUR WITH ALL PHENOTHIAZINES... PERIPHERAL EDEMA OCCURS IN 1-3% OF PT & MAY BE OF ENDOCRINE ORIGIN. /PHENOTHIAZINES/ The antipsychotic drugs are not addicting ... However, some degree of physical dependence may occur, with malaise and difficulty in sleeping developing several days after abrupt discontinuation. Tolerance usually develops to the sedative effects ... over a period of days or weeks. Tolerance ... is demonstrable in behavioral and biochemical experiments in animals ... /Antipsychotic drugs/ For more Drug Warnings (Complete) data for MESORIDAZINE (20 total), please visit the HSDB record page. Pharmacodynamics Mesoridazine, the besylate salt of a metabolite of thioridazine, is a phenothiazine tranquilizer. Pharmacological studies in laboratory animals have established that mesoridazine has a spectrum of pharmacodynamic actions typical of a major tranquilizer. In common with other tranquilizers it inhibits spontaneous motor activity in mice, prolongs thiopental and hexobarbital sleeping time in mice and produces spindles and block of arousal reaction in the EEG of rabbits. It is effective in blocking spinal reflexes in the cut and antagonizes d-amphetamine excitation and toxicity in grouped mice. It shows a moderate adrenergic blocking activity in vitro and in vivo and antagonizes 5-hydroxytryptamine in vivo. Intravenously administered, it lowers the blood pressure of anesthetized dogs. It has a weak antiacetylcholine effect in vitro. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5869 mL | 12.9343 mL | 25.8685 mL | |
| 5 mM | 0.5174 mL | 2.5869 mL | 5.1737 mL | |
| 10 mM | 0.2587 mL | 1.2934 mL | 2.5869 mL |