Physicochemical Properties
| Molecular Formula | C30H35NO3 |
| Molecular Weight | 457.6038 |
| Exact Mass | 457.261 |
| CAS # | 99254-95-2 |
| PubChem CID | 127269 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 595.6±50.0 °C at 760 mmHg |
| Flash Point | 156.4±27.3 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.590 |
| LogP | 7.36 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 34 |
| Complexity | 634 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])[H])C1C([H])=C([H])C(C#CC2C([H])=C([H])C([H])=C([H])C=2[H])=C(C=1[H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])N(C([H])([H])[H])C([H])([H])C([H])([H])C1C([H])=C([H])C(=C(C=1[H])OC([H])([H])[H])OC([H])([H])[H] |
| InChi Key | HQDXNTRZJRNPLD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C30H35NO3/c1-23(31(2)20-19-25-13-18-29(33-4)30(21-25)34-5)11-14-27-22-28(32-3)17-16-26(27)15-12-24-9-7-6-8-10-24/h6-10,13,16-18,21-23H,11,14,19-20H2,1-5H3 |
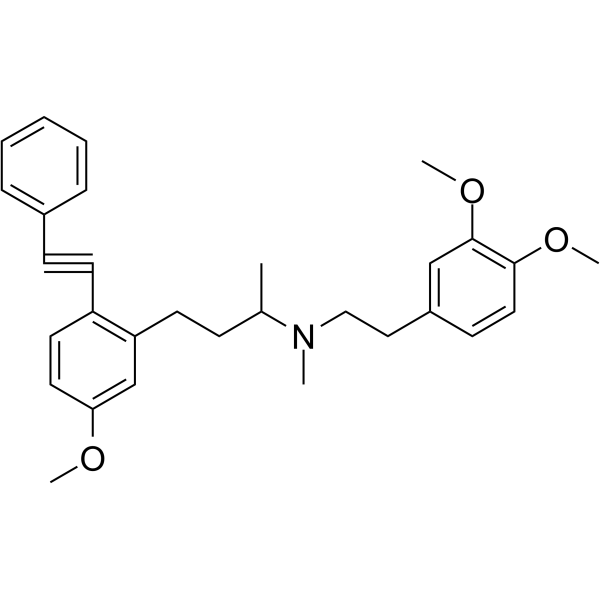
| Chemical Name | N-[2-(3,4-dimethoxyphenyl)ethyl]-4-[5-methoxy-2-(2-phenylethynyl)phenyl]-N-methylbutan-2-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | McN5691 (1 and 10 μM) induces concentration-dependent relaxation of aortic rings contracted by 30 mM KCl (EC50=190 μM) and inhibits contraction and calcium uptake induced by 60 mM KCl. In isolated rabbit thoracic aortic rings, McN5691 (McN-5691) has no effect at 10 μM or less on basal tension or calcium uptake (45Ca). Specific diltiazem binding to benzodiazepine receptors on voltage-sensitive calcium channels in skeletal muscle microsomal membranes is completely and highly-affinity inhibited (Kd=39.5 nM) by McN5691. McN5691 inhibits specific dihydropyridine receptor binding, in contrast to diltiazem. However, the effect is biphasic, consisting of high affinity components (Kd = 4.7 nM) and low affinity components (Kd = 919.8 nM). In the rabbit thoracic aorta, McN5691 causes concentration-dependent relaxation of the contractile ring at 1 μM NE (EC50=159 μM), and it inhibits norepinephrine (NE)-induced contraction (10 μM) and calcium uptake (1 and 10 μM) [1]. |
| ln Vivo | In beagle dogs, the excretion and metabolism of the antihypertensive medication McN5691 (RWJ-26240), an analogue of 2-ethynylphenylamine, were investigated. Urine excreted 2.8% and feces expelled 96.8% of the radioactive dose within 7 days of oral 14C-McN5691 treatment, respectively. Urine contained 2.8% and feces contained 96.8% of the radioactive dosage within 7 days of the oral treatment of 14C-McN5691. Within 48 hours, more than 87% of the dosage is eliminated by feces. In dogs, McN5691 is substantially metabolized. Unchanged McN5691 was found to account for 36% of the sample in 4 hour plasma and 0.1% and 19% of the dose in 0-24 hour urine and 0-48 hour fecal extracts, respectively [2]. Vascular resistance tended to be higher in spontaneously hypertensive rats (SHR) compared to Wistar-Kyoto (WKY) in the McN5691 (McN-5691) investigation, but the difference was only statistically significant in the cerebellum and midbrain [3]. |
| References |
[1]. Flaim SF, et al. Structurally novel antihypertensive compound, McN-5691, is a calcium channel blocker in vascular smooth muscle. J Pharmacol Exp Ther. 1991 Jan;256(1):279-88. [2]. Wu WN, et al. Excretion and metabolism of the antihypertensive agent, RWJ-26240 (McN-5691) in dogs. Drug Metab Dispos. 1998 Feb;26(2):115-25. [3]. Flaim SF, et al. Effects of the novel calcium channel blocker, McN-5691, on cardiocirculatory dynamics and cardiac output distribution in conscious spontaneously hypertensive rat. J Cardiovasc Pharmacol. 1988 Apr;11(4):489-500 |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1853 mL | 10.9266 mL | 21.8531 mL | |
| 5 mM | 0.4371 mL | 2.1853 mL | 4.3706 mL | |
| 10 mM | 0.2185 mL | 1.0927 mL | 2.1853 mL |