Physicochemical Properties
| Molecular Formula | C24H21N5O2S |
| Molecular Weight | 443.520843267441 |
| Exact Mass | 443.141 |
| CAS # | 306959-01-3 |
| PubChem CID | 1142445 |
| Appearance | Off-white to yellow solid powder |
| LogP | 5.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 32 |
| Complexity | 776 |
| Defined Atom Stereocenter Count | 0 |
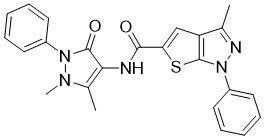
| SMILES | S1C(C(NC2C(N(C3C=CC=CC=3)N(C)C=2C)=O)=O)=CC2C(C)=NN(C3C=CC=CC=3)C1=2 |
| InChi Key | WIEJADKUYLLKPX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H21N5O2S/c1-15-19-14-20(32-24(19)28(26-15)17-10-6-4-7-11-17)22(30)25-21-16(2)27(3)29(23(21)31)18-12-8-5-9-13-18/h4-14H,1-3H3,(H,25,30) |
| Chemical Name | N-(1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)-3-methyl-1-phenyl-1H-thieno[2,3-c]pyrazole-5-carboxamide |
| Synonyms | MYLS-22 MYLS22 MYLS 22 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | By blocking OPA1, MYLS22 inhibits vascular tumorization and associated vascular angiogenesis, hence reducing tumor development and metastasis and successfully returning vascular tumor morphology to normal [2]. Acute myeloid leukocyte (AML) cell clonal development is inhibited by MYLS22 (10-30 μM; 7-10 days) with an IC50 of 12.5 μM [1]. In AML cells, MYLS22 (10-30 μM; 7-10 days) can effectively prevent mitochondrial fusion, lowering ROS levels and blocking the cell cycle during the G0/G1 transition phase [1]. |
| ln Vivo | MYLS22 (30 mg/kg; intraperitoneal injection; once daily for 7 days) significantly reduced the total tumor burden in mice [1]. MYLS22 (10 mg/kg; i.p.; once daily for six days) suppresses tumor growth in subcutaneous B16F10 melanoma mice by targeting OPA1 [3]. |
| References |
[1]. Mitochondrial fusion is a therapeutic vulnerability of acute myeloid leukemia. Leukemia. 2023 Apr;37(4):765-775. [2]. OPA1 and Angiogenesis: Beyond the Fusion Function. Cell Metab. 2020 May 5;31(5):886-887. [3]. Developmental and Tumor Angiogenesis Requires the Mitochondria-Shaping Protein Opa1. Cell Metab. 2020 May 5;31(5):987-1003.e8. [4]. L-OPA1 deficiency aggravates necroptosis of alveolar epithelial cells through impairing mitochondrial function during acute lung injury in mice. J Cell Physiol. 2022 Jul;237(7):3030-3043. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~56.37 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (2.82 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (2.82 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2547 mL | 11.2734 mL | 22.5469 mL | |
| 5 mM | 0.4509 mL | 2.2547 mL | 4.5094 mL | |
| 10 mM | 0.2255 mL | 1.1273 mL | 2.2547 mL |