MK-3328 is a novel and potent candidate PET ligand for the clinical assessment of β-amyloid plaque load. As a β-Amyloid PET ligand, MK-3328 exhibits high binding potency with an IC50 of 10.5 nM. MK-3328 has a favorable potency versus human β-amyloid plaque and has been radiolabeled for further evaluation in in vitro binding and in vivo PET imaging experiments. Studies led to the identification of 17b (MK-3328) as a candidate PET ligand for the clinical assessment of β-amyloid plaque load. MK-3328 is currently being tested for the treatment of Alzheimer's Disease.
Physicochemical Properties
| Molecular Formula | C14H9FN4O |
| Molecular Weight | 268.24586558342 |
| Exact Mass | 268.076 |
| CAS # | 1201323-97-8 |
| PubChem CID | 44555286 |
| Appearance | Off-white to light brown solid powder |
| LogP | 2.915 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 20 |
| Complexity | 371 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | WYQHKOHCTMNFAU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H9FN4O/c1-19-5-4-8-6-9(7-16-12(8)19)13-17-10-2-3-11(15)18-14(10)20-13/h2-7H,1H3 |
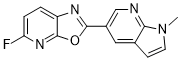
| Chemical Name | 5-fluoro-2-(1-methyl-1H-pyrrolo[2,3-b]pyridin-5-yl)oxazolo[5,4-b]pyridine |
| Synonyms | MK3328; MK 3328; MK-3328 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | MK-3328 demonstrates amyloid binding potency balanced with low levels of non-specific binding [1]. |
| ln Vitro |
MK-3328 demonstrates amyloid binding potency balanced with low levels of non-specific binding [1]. In a competition binding assay using human Alzheimer's disease (AD) brain homogenates, unlabeled MK-3328 demonstrated an IC₅₀ of 10.5 nM against the binding of the reference radioligand [³H₃]DMAB (8) to β-amyloid plaques. [1] Saturation binding studies using its tritium-labeled analog ([³H₃]MK-3328) in AD cortex homogenates from three different donors yielded a dissociation constant (Kd) of 9.6 ± 0.3 nM. The maximum binding capacity (Bmax) was 1100 ± 290 nM, resulting in a Bmax/Kd ratio of 117 ± 34. The nondisplaceable binding (NDB) at the Kd in the presence of 2 µM unlabeled compound was estimated at 35%. [1] Autoradiographic imaging of human AD brain slices with 5 nM [³H₃]MK-3328 showed a punctate binding pattern characteristic of amyloid plaques. This specific binding was completely blocked by co-incubation with 1 µM unlabeled MK-3328, confirming target engagement. The specific-to-nonspecific binding ratio for [³H₃]MK-3328 appeared qualitatively better than that of a close analog ([³H₃]14b). [1] In assays evaluating P-glycoprotein (P-gp) susceptibility and passive permeability, MK-3328 was found not to be a P-gp substrate (transport ratio < 3) and exhibited high passive permeability (Papp > 20 × 10⁻⁶ cm/s). [1] |
| ln Vivo |
In mouse models using positron emission tomography (PET) imaging, [18F]MK-3328 demonstrates good washout and high brain uptake in vivo [1]. In PET imaging studies in healthy, anesthetized rhesus monkeys, the fluorine-18 labeled tracer [¹⁸F]MK-3328 demonstrated favorable brain kinetics. It achieved high peak brain uptake, approximately 2.7 standardized uptake value (SUV) units in the cortex and 3.0 SUV in the cerebellum. The tracer washed out rapidly from the brain, with less than half of the peak uptake remaining in both cortex and cerebellum at 90 minutes post-injection. The time-activity curves for cortex and cerebellum closely correlated. In the 45-90 minute summed PET image coregistered with MRI, cortical regions showed a lack of obvious hotspots corresponding to white matter retention. [1] |
| Animal Protocol |
The in vivo evaluation involved PET imaging in healthy, anesthetized rhesus monkeys. The fluorine-18 labeled tracer ([¹⁸F]MK-3328) was administered intravenously. Dynamic PET scans were performed to generate time-activity curves for brain regions including cortex and cerebellum. Summed PET images from 45-90 minutes post-injection were coregistered with MRI for anatomical reference. The exact formulation, dose, and anesthesia details for the tracer administration are not provided in the manuscript. [1] |
| References |
[1]. Synthesis and Evaluation of 5-Fluoro-2-aryloxazolo[5,4-b]pyridines as β-Amyloid PET Ligands and Identification of MK-3328. ACS Med Chem Lett. 2011 Apr 18;2(7):498-502. [2]. [18F]Fluoroazabenzoxazoles as potential amyloid plaque PET tracers: synthesis and in vivo evaluation in rhesus monkey. Nucl Med Biol. 2011 Nov;38(8):1193-203. |
| Additional Infomation |
MK-3328 is a 5-fluoro-2-aryloxazolo[5,4-b]pyridine derivative identified from a series synthesized as potential β-amyloid PET ligands. [1] It is described as a candidate PET ligand for the clinical assessment of β-amyloid plaque load in Alzheimer's disease. [1] The manuscript mentions that human clinical trials to evaluate [¹⁸F]MK-3328 as an ¹⁸F-based β-amyloid PET ligand were underway at the time of publication. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.7279 mL | 18.6393 mL | 37.2787 mL | |
| 5 mM | 0.7456 mL | 3.7279 mL | 7.4557 mL | |
| 10 mM | 0.3728 mL | 1.8639 mL | 3.7279 mL |