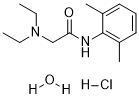
Lidocaine hydrochloride monohydrate (also known as Alphacaine; Lignocaine) is a potent and selective inverse peripheral histamine H1-receptor agonist with an IC50 of >32 μM. Lidocaine is a local anesthetic and cardiac depressant used as an antiarrhythmia agent. Originally developed as a local anesthetic, it has properties as a class IB antiarrhythmic, a long-acting membrane stabilizing agent used against ventricular arrhythmia. Its actions are more intense and its effects more prolonged than those of procaine but its duration of action is shorter than that of bupivacine or prilocaine.
Physicochemical Properties
| Molecular Formula | C14H25CLN2O2 |
| Molecular Weight | 288.816 |
| Exact Mass | 288.16 |
| CAS # | 6108-05-0 |
| Related CAS # | Lidocaine;137-58-6 |
| PubChem CID | 16219577 |
| Appearance | White to off-white solid powder |
| Boiling Point | 350.8ºC at 760 mmHg |
| Melting Point | 68.5ºC |
| Flash Point | 166ºC |
| LogP | 3.394 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 19 |
| Complexity | 228 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(C([H])([H])N(C([H])([H])C([H])([H])[H])C([H])([H])C([H])([H])[H])N([H])C1C(C([H])([H])[H])=C([H])C([H])=C([H])C=1C([H])([H])[H] |
| InChi Key | YECIFGHRMFEPJK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C14H22N2O.ClH/c1-5-16(6-2)10-13(17)15-14-11(3)8-7-9-12(14)4;/h7-9H, 5-6,10H2,1-4H3,(H,15,17);1H; |
| Chemical Name | 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide;hydrate;hydrochloride; |
| Synonyms | Lidocaine Hydrochloride; LIDOCAINE HCL; Lidothesin; Lignocaine hydrochloride; Xyloneural; Xylocitin; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Cell growth is dramatically reduced by lignocaine (10 nM; 48 hours) [2]. Cell viability is inhibited by lidocaine (1-10 nM; 24-72 hours); the maximum inhibitory effect is obtained at a dose of 10 nM and a treatment duration of 48 hours [2]. The rate of apoptosis is markedly increased by lidocaine (10 nM; 48 hours) [2]. Lidocaine (10 nM; 48 hours) dramatically increases the expression of p21 and downregulates Cyclin D1 [2]. |
| ln Vivo | In rats, lidocaine (lidocaine) produces a completely reversible block of the tail nerve. Compared to thermal nociceptive blockade, mechanical nociceptive blockade, which is generated by lidocaine, has a slower onset and a quicker recovery [3]. |
| Cell Assay |
Cell proliferation experiment [2] Cell Types: human gastric cancer cell line MKN45 Tested Concentrations: 10 nM Incubation Duration: 48 hrs (hours) Experimental Results: Cell proliferation diminished Dramatically. Cell viability assay [2] Cell Types: human gastric cancer cell line MKN45 Tested Concentrations: 1, 5 and 10 nM Incubation Duration: 24, 48, 72 hrs (hours) Experimental Results: Inhibition of MKN45 cell viability. Cell apoptosis analysis [2] Cell Types: Human gastric cancer cell line MKN45 Tested Concentrations: 10 nM Incubation Duration: 48 hrs (hours) Experimental Results: The cell apoptosis rate increased Dramatically. Western Blot Analysis [2] Cell Types: human gastric cancer cell line MKN45 Tested Concentrations: 10 nM Incubation Duration: 48 hrs (hours) Experimental Results: Cyclin D1 expression was Dramatically down-regulated, and p21 expression was Dramatically up-regulated. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Lidocaine concentrations in milk during continuous IV infusion, epidural administration and in high doses as a local anesthetic are low and the lidocaine is poorly absorbed by the infant. Lidocaine is not expected to cause any adverse effects in breastfed infants. No special precautions are required. Lidocaine during labor and delivery with other anesthetics and analgesics has been reported by some to interfere with breastfeeding. However, this assessment is controversial and complex because of the many different combinations of drugs, dosages and patient populations studied as well as the variety of techniques used and deficient design of many of the studies. Overall it appears that with good breastfeeding support epidural lidocaine with or without fentanyl or one of its derivatives has little or no adverse effect on breastfeeding success. Labor pain medication may delay the onset of lactation. ◉ Effects in Breastfed Infants Lidocaine in doses ranging from 60 to 500 mg administered to the mother by intrapleural or epidural routes during delivery had no effect on their 14 infants who were either breastfed or received their mother's breastmilk by bottle. A neurology group reported using 1% lidocaine for peripheral nerve blocks in 14 nursing mothers with migraine. They reported no infant side effects and considered the procedure safe during breastfeeding. ◉ Effects on Lactation and Breastmilk A randomized study compared three groups of women undergoing elective cesarean section who received subcutaneous infusion of 20 mL of lidocaine 1% plus epinephrine 1:100:000 at the incision site. One group received the lidocaine before incision, one group received the lidocaine after the incision, and the third received 10 mL before the incision and 10 mL after. Women in the pre-and post-incision administration group initiated breastfeeding earlier than those in the pre-incision administration (3.4 vs 4.1 hours). There was no difference between the post-incision administration group and the other groups in time to breastfeeding initiation. A national survey of women and their infants from late pregnancy through 12 months postpartum compared the time of lactogenesis II in mothers who did and did not receive pain medication during labor. Categories of medication were spinal or epidural only, spinal or epidural plus another medication, and other pain medication only. Women who received medications from any of the categories had about twice the risk of having delayed lactogenesis II (>72 hours) compared to women who received no labor pain medication. An Egyptian study compared lidocaine 2% (n = 75) to lidocaine 2% plus epinephrine 1:200,000 (n = 70) as a wound infiltration following cesarean section. Patients who received epinephrine in combination with lidocaine began breastfeeding at 89 minutes following surgery compared to 132 minutes for those receiving lidocaine alone. The difference was statistically significant. |
| References |
[1]. Setting up for the block: the mechanism underlying lidocaine's use-dependent inhibition of sodium channels. J Physiol. 2007 Jul 1;582(Pt 1):11. [2]. Lidocaine inhibits growth, migration and invasion of gastric carcinoma cells by up-regulation of miR-145. BMC Cancer. 2019 Mar 15;19(1):233. [3]. Evaluation of the antinociceptive effects of lidocaine and bupivacaine on the tail nerves of healthy rats. Basic Clin Pharmacol Toxicol. 2013 Jul;113(1):31-6. |
| Additional Infomation |
Lidocaine Hydrochloride is the hydrochloride salt form of lidocaine, an aminoethylamide and a prototypical member of the amide class anesthetics. Lidocaine interacts with voltage-gated Na+ channels in the nerve cell membrane and blocks the transient increase in permeability of excitable membranes to Na+. This prevents the generation and conduction of nerve impulses and produces a reversible loss of sensation. Lidocaine hydrochloride also exhibits class IB antiarrhythmic effects. The agent decreases the flow of sodium ions into myocardial tissue, especially on the Purkinje network, during phase 0 of the action potential, thereby decreasing depolarization, automaticity and excitability. A local anesthetic and cardiac depressant used as an antiarrhythmia agent. Its actions are more intense and its effects more prolonged than those of PROCAINE but its duration of action is shorter than that of BUPIVACAINE or PRILOCAINE. See also: Lidocaine Hydrochloride (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4624 mL | 17.3118 mL | 34.6236 mL | |
| 5 mM | 0.6925 mL | 3.4624 mL | 6.9247 mL | |
| 10 mM | 0.3462 mL | 1.7312 mL | 3.4624 mL |