Physicochemical Properties
| Molecular Formula | C20H25N7O6 |
| Molecular Weight | 459.46 |
| Exact Mass | 459.186 |
| CAS # | 31690-09-2 |
| Related CAS # | Levomefolic acid-13C,d3;1356019-94-7;Levomefolate calcium;151533-22-1;Levomefolic acid-13C5;2687960-08-1 |
| PubChem CID | 135398561 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.6±0.1 g/cm3 |
| Index of Refraction | 1.733 |
| LogP | -2.61 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 33 |
| Complexity | 865 |
| Defined Atom Stereocenter Count | 2 |
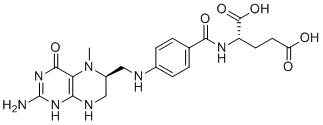
| SMILES | CN1[C@H](CNC2=C1C(=O)NC(=N2)N)CNC3=CC=C(C=C3)C(=O)N[C@@H](CCC(=O)O)C(=O)O |
| InChi Key | ZNOVTXRBGFNYRX-STQMWFEESA-N |
| InChi Code | InChI=1S/C20H25N7O6/c1-27-12(9-23-16-15(27)18(31)26-20(21)25-16)8-22-11-4-2-10(3-5-11)17(30)24-13(19(32)33)6-7-14(28)29/h2-5,12-13,22H,6-9H2,1H3,(H,24,30)(H,28,29)(H,32,33)(H4,21,23,25,26,31)/t12-,13-/m0/s1 |
| Chemical Name | (2S)-2-[[4-[[(6S)-2-amino-5-methyl-4-oxo-3,6,7,8-tetrahydropteridin-6-yl]methylamino]benzoyl]amino]pentanedioic acid |
| Synonyms | LMSR Levomefolinic acidLevomefolinic acid Metafolin BodyfolinNutrifolin Levomefolate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Reagents Glu406, Ile 418, Lys417, and Tyr453 are interacting with levomefolic acid (5-MTHF). Lys 417 and Tyr 453 are physiologically significant in the interaction between S1 and ACE and may function as S1 Potential Inhibitors of: ACE2 interaction with SARS-CoV-2 virus [1]. Levomefolate (5-MTHF) (50 nM, 72 hours) boosts intracellular folate metabolic activity in human lymphoblastoid cell lines (LCL) by 7-fold, which is 2 times greater than folic acid. This makes it an effective folate supplement agent[2]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Absorbed in the proximal small intestines via the active proton-coupled folate transporter (PCFT) that transports both oxidized and reduced folic acids. Passive diffusion also occurs at the proximal and distal portions of the small intestines. A single oral dose of 906nmol of levomefolic acid in healthy females resulted in the mean peak plasma concentration of 39.4nmol/L. Mainly eliminated through renal or fecal excretion. Small proportion of excreted levomefolic acid is in unchanged form as over 99% of tissue folate is in polyglutamate form. Some portions of levomefolic acid is secreted into bile. Circulates in its free form or loosely bound to plasma proteins Metabolism / Metabolites Levomefolic acid is further converted into tetrahydrofolate (THF) via the vitamin B12-dependent enzyme methionine synthase before effective polyglutamylation by folylpolyglutamate synthetase (FPG). Polyglumate forms of folic acids are more effective substrates for associated enzymes in folate-dependent reactions. Biological Half-Life The mean elimination half-life is approximately 3 hours after 5mg of oral L-methylfolate, administered daily for 7 days. |
| Toxicity/Toxicokinetics |
Protein Binding Approximately 56% bound to plasma proteins |
| References |
[1]. Virtual screening and molecular dynamics study of approved drugs as inhibitors of spike protein S1 domain and ACE2 interaction in SARS-CoV-2. J Mol Graph Model. 2020 Dec;101:107716. [2]. Simultaneous quantification of intracellular concentrations of clinically important metabolites of folate-homocysteine cycle by LC-MS/MS. Anal Biochem. 2020 Sep 15;605:113830. |
| Additional Infomation |
5-methyltetrahydrofolic acid is a tetrahydrofolic acid that is 5,6,7,8-tetrahydrofolic acid substituted by a methyl group at position 5. It has a role as a human metabolite. It is functionally related to a 5,6,7,8-tetrahydrofolic acid. It is a conjugate acid of a 5-methyltetrahydrofolate(2-). 5-methyltetrahydrofolic acid is a methylated derivate of tetrahydrofolate. It is generated by methylenetetrahydrofolate reductase from 5,10-methylenetetrahydrofolate and used to recycle homocysteine back to methionine by 5-methyltetrahydrofolate-homocysteine methyltransferases (also called methionine synthases). Levomefolic acid (INN) is the metabolite of folic acid (Vitamin B9) and it is a predominant active form of folate found in foods and in the blood circulation, accounting for 98% of folates in human plasma. It is transported across the membranes including the blood-brain barrier into various tissues where it plays an essential role in the DNA synthesis, cysteine cycle and regulation of homocysteine, where it methylates homocysteine and forms methionine and tetrahydrofolate (THF). Levomefolate is approved as a food additive and is designated a GRAS (generally regarded as safe) compound. It is available commercially as a crystalline form of the calcium salt (Metafolin(R)), which has the stability required for use as a supplement. Supplementation of levomefolic acid is desired over folic acid due to reduced potential for masking vitamin B12 deficiency symptoms. 5-Methyltetrahydrofolic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). Levomefolic acid is a Folate Analog. Levomefolic acid has been reported in Homo sapiens with data available. L-methylfolate is a nutritional supplement containing the biologically active form of the B9 vitamin folate, 5-methyltetrahydrofolate (L-methylfolate), with potential antineoplastic activity. Upon administration, L-methylfolate is able to provide methyl groups allowing an increase in the level of DNA methylation in the promoter regions of certain tumor-promoting genes, thereby reversing the DNA hypomethylation of these genes and inactivating them. This may result in a decrease of both tumor cell proliferation and tumor progression. In addition, administration of L-methylfolate may sensitize tumor cells to the cytotoxic effects of other chemotherapeutic agents. Unlike folic acid, L-methylfolate is able to cross the blood brain barrier and could be beneficial in the treatment of brain tumors. DNA hypomethylation of certain genes leads to chromosome instability and contributes to tumor development. See also: 5-Methyltetrahydrofolic acid (annotation moved to). Drug Indication For the treatment and prevention of folate deficiency and for use as an antidote against folic acid antagonists. Contained in oral contraceptives to reduce the risk of neural tube defects arising from folic acid deficiency for pregnant women who conceived during use or shortly after the discontinuation of the product. Being studied for use as a treatment for cardiovascular diseases and adjunct therapy for patients undergoing antidepressant pharmacotherapy. Mechanism of Action Levomefolic acid plays a critical role in methylating homocysteines into methionine by acting as a methyl donor in a reaction catalyzed by vitamine B12-dependent methionine synthase. Homocysteine must either be further metabolized via transulfuration to become cysteine, taurine, and glutathione via a B6-dependent process, or re-methylated to become methionine again. Methionine formed from remethylation of homocysteine by levomefolic acid forms the downstream metabolite S-adenosylmethionine (SAMe), which is involved in numerous biochemical methyl donation reactions, including reactions forming monoamine neurotransmitters. Studies suggest that high plasma levels of homocysteine is associated with increased incidences of arterial plaque formation. Pharmacodynamics Levomefolic acid is an active metabolite of folic acid and a methyl group donor in one-carbon metabolism reactions. It regulates important cellular functions such as DNA biosynthesis, gene expression regulation, amino acid synthesis and metabolism, and myelin synthesis and repair. As a only form of folate that can cross the blood-brain barrier, it acts as a cofactor in the production of monoamine neurotransmitters such as dopamine, serotonin and norepinephrine. Levomefolic acid is also involved in red blood cell formation. |
Solubility Data
| Solubility (In Vitro) | H2O : ~2.08 mg/mL (~4.53 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1765 mL | 10.8823 mL | 21.7647 mL | |
| 5 mM | 0.4353 mL | 2.1765 mL | 4.3529 mL | |
| 10 mM | 0.2176 mL | 1.0882 mL | 2.1765 mL |