LY2365109 is a novel, potent and selective inhibitor of GlyT1 (glycine transporter 1) with IC50 value of 15.8 nM. Selective inhibitors of the glycine transporter 1 (GlyT1) have been implicated in central nervous system disorders related to hypoglutamatergic function such as schizophrenia. The selective GlyT1 inhibitors ALX5407 (NFPS) and LY2365109 increased cerebrospinal fluid levels of glycine and potentiated NMDA-induced increases in dialysate levels of neurotransmitters in the prefrontal cortex (PFC) and the striatum. LY2365109 appeared slightly more potent than ALX5407 over the dose-range tested on CSF levels of glycine in the rat LY2365109 can act to potentiate NMDA function in the PFC but also mediate sustained inhibition of GlyT1 transporters in caudal areas of the brain.LY2365109 increased seizure thresholds in mice. Importantly, chronic seizures in the mouse model of TLE were robustly suppressed by systemic administration of the GlyT1 inhibitor LY2365109.
Physicochemical Properties
| Molecular Formula | C22H28CLNO5 | |
| Molecular Weight | 421.91 | |
| Exact Mass | 421.165 | |
| Elemental Analysis | C, 62.63; H, 6.69; Cl, 8.40; N, 3.32; O, 18.96 | |
| CAS # | 1779796-27-8 | |
| Related CAS # | 1779796-27-8 (HCl);868265-28-5; | |
| PubChem CID | 56972229 | |
| Appearance | White to off-white solid powder | |
| LogP | 0 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 8 | |
| Heavy Atom Count | 29 | |
| Complexity | 520 | |
| Defined Atom Stereocenter Count | 0 | |
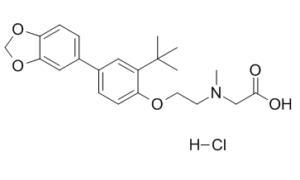
| SMILES | CC(C)(C)C1=C(C=CC(=C1)C2=CC3=C(C=C2)OCO3)OCCN(C)CC(=O)O.Cl |
|
| InChi Key | ZQVOAGQZHDAFRM-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C22H27NO5.ClH/c1-22(2,3)17-11-15(16-6-8-19-20(12-16)28-14-27-19)5-7-18(17)26-10-9-23(4)13-21(24)25;/h5-8,11-12H,9-10,13-14H2,1-4H3,(H,24,25);1H | |
| Chemical Name | 2-[2-[4-(1,3-benzodioxol-5-yl)-2-tert-butylphenoxy]ethyl-methylamino]acetic acid;hydrochloride | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Glycine Transporter 1 (GlyT1) (Ki = 16 nM for human GlyT1) [1] |
| ln Vitro |
In vitro activity: LY2365109 is a novel, potent and selective inhibitor of GlyT1 (glycine transporter 1) with IC50 value of 15.8 nM. Selective inhibitors of the glycine transporter 1 (GlyT1) have been implicated in central nervous system disorders related to hypoglutamatergic function such as schizophrenia. The selective GlyT1 inhibitors ALX5407 (NFPS) and LY2365109 increased cerebrospinal fluid levels of glycine and potentiated NMDA-induced increases in dialysate levels of neurotransmitters in the prefrontal cortex (PFC) and the striatum. Kinase Assay: LY2365109 is a potent and selective GlyT1 inhibitors with IC50 value of 15.8 nM. LY2365109 potently inhibited [³H]glycine uptake in CHO cells expressing human GlyT1, with an IC₅₀ of 17 nM. It exhibited >300-fold selectivity over GlyT2 and negligible activity at other neurotransmitter transporters (e.g., norepinephrine, dopamine, serotonin) or receptors (e.g., NMDA, GABA_A). [1] |
| ln Vivo |
In mice, subcutaneous administration of LY2365109 (3–30 mg/kg) dose-dependently elevated glycine levels in cerebrospinal fluid (CSF) and hippocampal microdialysates, with maximal effects in caudal brain regions (e.g., spinal cord) versus cortex. [1] LY2365109 (10–30 mg/kg, s.c.) significantly reduced seizure severity and mortality in the maximal electroshock (MES) and pentylenetetrazol (PTZ) models of epilepsy. This effect was reversed by glycine receptor antagonists, confirming GlyT1-dependent action. [2] Doses ranging from 0.3 to 30 mg/kg po of LY2365109 hydrochloride cause dose-dependent increases in glycine levels in the CSF(1). Seizures thresholds in mice are raised by LY2365109 hydrochloride[2]. |
| Enzyme Assay |
Competitive binding assays measured affinity for GlyT1 using [³H]LY2365109 in human GlyT1-expressing membranes. Saturation and displacement studies determined Ki values. Functional inhibition was confirmed via [³H]glycine uptake kinetics. [1] |
| Cell Assay |
[³H]Glycine uptake assays were performed in CHO cells stably transfected with human GlyT1. Cells were incubated with LY2365109 and [³H]glycine, then rapidly washed and lysed. Radioactivity was quantified to determine glycine uptake inhibition. [1] |
| Animal Protocol |
For neurochemistry: Mice received subcutaneous (s.c.) injections of LY2365109 (0.3–30 mg/kg) dissolved in saline. CSF and brain microdialysates were collected at 30–120 min post-dose for glycine quantification. [1] For epilepsy models: Mice were pretreated with LY2365109 (10–30 mg/kg, s.c.) 30 min before MES or PTZ challenge. Seizure severity was scored, and mortality recorded over 24h. [2] Animal/Disease Models: Male SD (Sprague-Dawley) rats (250-300 g )[1] Doses: 0.3 mg/kg, 1 mg/kg, 5 mg/kg, 10 mg/kg, 30 mg/kg Route of Administration: Oral administration Experimental Results: Produced dose-dependent elevations in CSF levels of glycine measured 1 h after dosing. |
| References |
[1]. Neurochemical and behavioral profiling of the selective GlyT1 inhibitors ALX5407 and LY2365109 indicate a preferential action in caudal vs. cortical brain areas. Neuropharmacology. 2008 Oct;55(5):743-54. [2]. Glycine transporter 1 is a target for the treatment of epilepsy. Neuropharmacology. 2015 Dec;99:554-65. |
| Additional Infomation |
LY2365109 demonstrates region-specific GlyT1 inhibition, preferentially enhancing glycine neurotransmission in subcortical/spinal areas over cortex. This may underlie its efficacy in seizure models without inducing cortical hyperexcitability. [1] GlyT1 inhibition by LY2365109 suppresses epileptiform activity via disinhibition of glycinergic neurotransmission, particularly in brainstem and spinal circuits, offering a novel mechanism for epilepsy treatment. [2] First-generation GlyT1 inhibitor with demonstrated efficacy in preclinical epilepsy models, though clinical development status is unclear [1][2]. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.93 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.93 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.93 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3702 mL | 11.8509 mL | 23.7017 mL | |
| 5 mM | 0.4740 mL | 2.3702 mL | 4.7403 mL | |
| 10 mM | 0.2370 mL | 1.1851 mL | 2.3702 mL |