Physicochemical Properties
| Molecular Formula | C13H21N5O2 |
| Molecular Weight | 279.34 |
| Exact Mass | 279.17 |
| Elemental Analysis | C, 55.90; H, 7.58; N, 25.07; O, 11.45 |
| CAS # | 177314-99-7 |
| PubChem CID | 15649468 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0.94 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 20 |
| Complexity | 354 |
| Defined Atom Stereocenter Count | 4 |
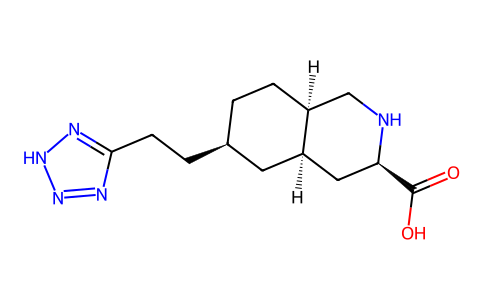
| SMILES | OC([C@H]1C[C@H]2C[C@@H](CCC3N=NNN=3)CC[C@H]2CN1)=O |
| InChi Key | ZXFRFPSZAKNPQQ-LMLFDSFASA-N |
| InChi Code | InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11-/m1/s1 |
| Chemical Name | (3R,4aR,6R,8aR)-6-[2-(2H-tetrazol-5-yl)ethyl]-1,2,3,4,4a,5,6,7,8,8a-decahydroisoquinoline-3-carboxylic acid |
| Synonyms | LY326325; LY326325; LY 326,325; LY-326,325; 177314-99-7; (-)-(3S,4aR,6R,8R)-6-(2-(1(2)H-Tetrazol-5-yl)ethyl)dec ahydroisoquinaline-3-carboxylic acid; (3R,4aR,6R,8aR)-6-[2-(2H-tetrazol-5-yl)ethyl]-1,2,3,4,4a,5,6,7,8,8a-decahydroisoquinoline-3-carboxylic acid; 3-Isoquinolinecarboxylic acid, decahydro-6-(2-(1H-tetrazol-5-yl)ethyl)-, (3alpha,4abeta,6alpha,8abeta)-; 3-Isoquinolinecarboxylic acid, decahydro-6-(2-(1H-tetrazol-5-yl)ethyl)-, (3R,4aR,6R,8aR)-rel-; SCHEMBL13320011; LY 326325 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | AMPA receptor |
| ln Vivo |
The effect of LY326325, a novel AMPA receptor antagonist, on the conditioned avoidance response and catalepsy was investigated in the rat. The conditioned avoidance response is a behavioral methodology which is regarded to predict potential antipsychotic efficacy of experimental drugs. Catalepsy ratings were utilized to assess the putative propensity of LY325326 to induce extrapyramidal side effects. Systemic administration of LY326325, 18 mg/kg subcutaneously, caused a selective suppression of conditioned avoidance response, without effect on escape behavior or intertrial crosses. In addition, no catalepsy was observed. Our present and previous results support an antipsychotic effect of AMPA receptor antagonists with a low liability for extrapyramidal side effects, i.e. pharmacological effects consonant with an atypical antipsychotic profile.[1] Anxiolytic-like effects produced by the novel, water-soluble AMPA/kainate receptor antagonist, LY326325 (3RS,4aRS,6RS,8aRS)-6-[2-(1(2)H-tetrazole-5-yl)e thyl]decahydro-isoquinoline-3-carboxylic acid), were examined in the elevated plus-maze and in a conflict-suppressed drinking situation. Administration of low doses (0.5, 1.2, and 5 mg/kg; i.p., -30 min) of LY326325 to Sprague-Dawley rats did not alter the percentage of entries into the open arms of the plus-maze, whereas only one dose of LY326325 (1 mg/kg) produced a slight, but significant, increase of the time spent in the open arms of the plus maze. In the conflict-suppressed drinking test, similar doses of LY326325 (2.5 and 5 mg/kg; i.p., -30 min) caused a dose-dependent and significant increase of punished drinking behavior without having any significant effects on unpunished drinking. The anxiolytic-like effects of LY326325 in the plus-maze and in the anticonflict tests were observed at doses, which, by themselves, had no influence on various measures of locomotor activity, i.e., horizontal activity, forward locomotion, and corner time. Our data suggest that the putative AMPA/glutamate receptor antagonist, LY326325, produces anxiolytic-like effects similar to those of diazepam in the conflict-suppressed drinking test, but displays considerably weaker anxiety-reducing properties compared to diazepam in the elevated plus-maze.[2] |
| Animal Protocol |
Elevated Plus-Maze Experiments [2] The plus-shaped maze was made of wood and positioned on a height of 50 cm above the floor in a quiet laboratory surrounding. Two opposite arms were open (50 × 10 cm), and the other two were enclosed with walls (50 × 10 × 40 cm). Experiments were carried out in a darkened and quiet room with a constant light of 15 W, located 80 cm above the maze and directed towards the apparatus. The light levels on the open and enclosed arms were equal. Three days before the experiment each rat was handled every day for 5 min. Animals were brought in their home cage into a separate silent room for 60 min before the experiment. Before the start of the pluz-maze behavior recordings each animal was placed into a novel environment, represented by a conventional Skinner box, for 5 min, a procedure that, at least in our hands, increased the exploratory behavior of the animals and produced more consistent data between day-to-day test sessions. The plus-maze experiment was initiated by placing the rat into the center of the plus-maze facing an open arm, after which the number of entries and time spent in each of the two arms were recorded for a period of 5 min by an independent observer with no information of the drug treatment protocol. An “arm entry” was recorded when the rat entered the arm with all four paws into the arm. The maze was carefully cleaned with tap water after each test session and with a weak alcohol washing solution after finishing all the experimental sessions of the day. The open-arm activity was quantified as (a) time spent in the open arms as well as, (b) number of entries into the open arms. The percent time spent in the open arms was expressed as time in the open arms/time spent in the enclosed arms × 100. The percent entries into the open arms was expressed as the number of entries into the open arms/total entries × 100. The AMPA/kainate glutamate receptor antagonist, LY326325, was given intraperitoneally (IP), 30 min prior to the test. The animals tested in the plus-maze were not used in any other experiments. Conflict-Suppressed Drinking Test [2] The drinking training sessions and the conflict-suppressed drinking experiments were conducted in two standard boxes for operant behavior designed for shock-induced suppression of drinking in rats. The training and testing procedures were largely adapted from recently described experimental protocol 27, 29. Briefly, the experiment was carried out during 3 consecutive days. In the morning of the first day the drinking water was removed from the home cage of the animals. During the second and third day the subjects were placed in the test apparatus and allowed a 12 min period of free drinking of 5% (w/v) glucose with no electric shocks delivered. After this 2-day training period, most animals showed a stable baseline of number of licks recorded during the training session. A few animals refused to drink and were removed from the experiments. On the day of the experiments the animals were randomly divided into a control group (receiving solvent), and an experimental group receiving the drug of interest. Thirty minutes after the drug administration the animals were placed into the apparatus and allowed to perform three drinking episodes before the time recording of the experimental session was initiated. To establish unpunished drinking behavior, the number of drinking episodes were recorded during the 4 first minutes of the experimental session, during which no shocks were delivered. After this initial period of unpunished drinking, each following drinking episode, was followed by an electric shock during a period of 8 min, during which the total number of shocks received were recorded. All recordings were carried out between 1200 and 1800 h to avoid to large diurnal variations in the results. In a previous study we have shown that the benzodiazepine receptors agonist, diazepam, when given in a dose of 2.0 mg/kg (IP), produces reliable anticonflict in Sprague–Dawley rats in the currently used experimental paradigm. The AMPA/kainate glutamate receptor antagonist, LY326325was given IP in doses of 2.5 and 5.0 mg/kg, 30 min prior to the start of the behavioral recordings. Locomotor Activity Recordings [2] The locomotor activity of the animals was recorded using a computer-assisted photocell apparatus previously described in detail by Ericson et al. Briefly, the animals were placed individually into Plexiglas boxes (680 × 680 × 450 mm), which were situated in a ventilated and sound-attenuating enclosure and equipped with two rows of eight photocell, sensitive to infrared light, and located 40 and 125 mm, respectively, above the floor. The locomotor activity field was not illuminated during the recordings. The behavioral activity was recorded at every 5 min for a total period of 30 min. “Horizontal activity” indicates overall behavioral activity, “forward locomotion” denotes continuous movement in the same direction (considered useful for the identification of bizarre patterns of movement, and “corner time” described the lack of movement within the corner areas of the locomotor activity box. The AMPA/kainate receptor antagonist, LY326325, was given IP 30 min prior to the the start of the locomotor activity recordings. LY326325 was dissolved in a few drops of 0.2 M NaOH and distilled water with the pH of final volume adjusted to 7.4 using 0.2 m HCl.[2] A shuttle-box (530 3 250 3 225 mm) divided into two compartments by a partition was used (see Wadenberg et al., 1990). Upon presentation of a conditioned stimulus (CS; 80 1004 J. M. Mathé et al. dB white noise) the animals had 10s to move into the adjacent compartment of the shuttle-box. If the rat remained in the same compartment for longer than 10s, the unconditioned stimulus (UCS) was presented, i.e. an intermittent electric shock in the floor grid (4 shocks per 10s, duration 0.5s, approximately 0.2mA), until an escape response was performed, i.e. moving into the other compartment. Avoidance was recorded as a response to the CS within 10 s, escape as response to CS and UCS, i.e. .10s, and intertrial crosses, i.e. movement between compartments between trials. Escape failures were defined as a failure to move into the adjacent chamber within 60s. The animals were trained for three consecutive days and were initially habituated to the shuttle-box for 5 min, and subsequently trained. Each training session consisted of 20 trials randomly distributed over 15min. Experimental trials were preceded by a pre-test to reaffirm the rats’ maintenance of CS responding ($80% avoidance). All pre-tests and experimental trials consisted of 10 trials randomly distributed over 7.5 min. Test sessions were conducted 20, 90 and 240 minutes after systemic administration of drug or vehicle. Animals were subjected to repeated observations using a change-over design (Li, 1964) with a one week inter-trial delay period.[1] Animals were placed on an 60° inclined grid and, excluding the first 30s, the time the rat remained in the same position was measured, for a maximum of 2.5 min. The catalepsy was scored from 0–5 according to the (square root transformation) immobility time (min): 0 5 0–0.08, 1 5 0.09–0.35, 2 5 0.36–0.08, 3 5 0.81–1.42, 4 5 1.42–2.24, 5 $ 2.25. [1] LY326325 (LY293558 monohydrate; [3S(3α,4aα,6â,8aα)]decahydro-6-[2-(1H-tetrazol5-yl)ethyl]3-isoquinolinecarboxylic acid monohydrate 6 and 18mg/kg) was dissolved in deionized water. Vehicle injections refer to deionized water. The treatments were administered subcutaneously (s.c.) at a volume of 2ml/kg.[1] |
| References |
[1]. Antipsychotic-like effect of the AMPA receptor antagonist LY326325 as indicated by suppression of conditioned avoidance response in the rat. J Neural Transm (Vienna). 1999;106(9-10):1003-9. [2]. The putative AMPA receptor antagonist, LY326325, produces anxiolytic-like effects without altering locomotor activity in rats. Pharmacol Biochem Behav. 1998 May;60(1):119-24 |
| Additional Infomation | In conclusion, the working hypothesis that the selective AMPA receptor antagonist, LY326325, may possess anxiolytic properties is supported by our data obtained in the anticonflict test, whereas the results recorded in the elevated plus-maze assay appear to be less supportive for the use of LY326325 as an anxiolytic-like agent. The reason for this discrepancy is not known. One possibility is that various animal models for anxiety measure different forms of anxiety. The advantages and disadvantages of the elevated plus-maze for the discovery of novel anxiolytic agents has recently been thoroughly discussed by Dawson and Tricklebank. For example, a drug-induced change of locomotor activity is considered a confounding factor for the correct identification of anxiolytic and/or anxiogenic effects in the elevated plus-maze. However, at least in the present series of experiments we found no evidence that the negative findings with LY326325 could be explained as due to altered locomotor activity in that there was no increase in the total number of arm entries (data not shown), neither could any significant sedative or stimulatory effects of LY326325 be demonstrated in the locomotor activity test. Another possibility could be that the discovery of anxiolytic-like properties of certain drugs may depend on whether the agents are evaluated in animal models for nonconditioned or conditioned anxiety-like behaviors. Conditioned animal models of anxiety are, in contrast to the elevated-plus maze, considered to be more robust models for the discovery of anxiolytic agents. Thus, it seems that the competitive AMPA receptor antagonist, LY326325, at least in our hands, appears to be less effective for emotional states involving nonconditioned anxiety, whereas its influence on conditioned behaviors could be more convincingly demonstrated. Taken together, our current observations suggest that AMPA/glutamate receptor antagonists may possess therapeutic potential as anxiety-reducing agents in humans, especially because they appear to have the advantage of not causing unwanted psychotomimetic side effects often observed with classical NMDA/glutamate receptor antagonists. [2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5799 mL | 17.8993 mL | 35.7987 mL | |
| 5 mM | 0.7160 mL | 3.5799 mL | 7.1597 mL | |
| 10 mM | 0.3580 mL | 1.7899 mL | 3.5799 mL |