Physicochemical Properties
| Molecular Formula | C11H20NO5P |
| Molecular Weight | 277.26 |
| Exact Mass | 277.108 |
| Elemental Analysis | C, 47.65; H, 7.27; N, 5.05; O, 28.85; P, 11.17 |
| CAS # | 137433-06-8 |
| PubChem CID | 131938 |
| Appearance | Solid powder |
| Density | 1.339g/cm3 |
| Boiling Point | 539.6ºC at 760mmHg |
| Flash Point | 280.1ºC |
| LogP | 0.971 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 18 |
| Complexity | 368 |
| Defined Atom Stereocenter Count | 4 |
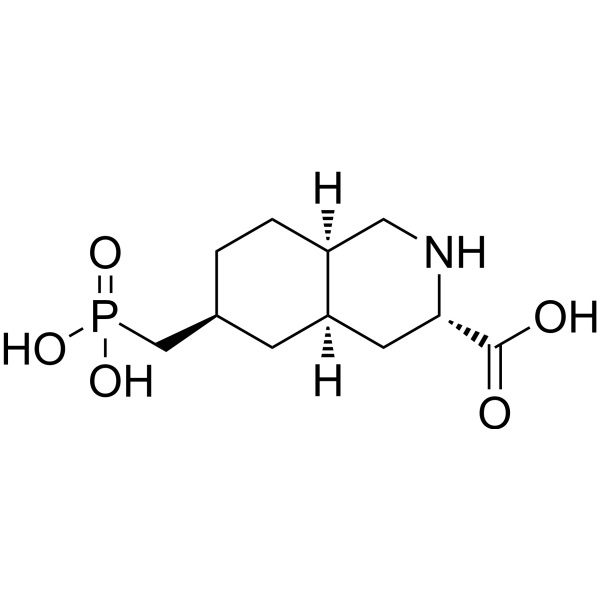
| SMILES | P(CC1CCC2C(CC(C(O)=O)NC2)C1)(O)(O)=O |
| InChi Key | STIRHCNEGQQBOY-QEYWKRMJSA-N |
| InChi Code | InChI=1S/C11H20NO5P/c13-11(14)10-4-9-3-7(6-18(15,16)17)1-2-8(9)5-12-10/h7-10,12H,1-6H2,(H,13,14)(H2,15,16,17)/t7-,8-,9+,10-/m0/s1 |
| Chemical Name | (3S,4aR,6S,8aR)-6-(phosphonomethyl)-1,2,3,4,4a,5,6,7,8,8a-decahydroisoquinoline-3-carboxylic acid |
| Synonyms | LY-235959; LY235959; LY 235959; 137433-06-8; LY-274614; 136109-04-1; LY-235,959; AR2BHC0C6P; LY274614; Decahydro-6-(phosphonomethyl)-3-isoquinolinecarboxylic acid; LY 235959 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | N-methyl-D-aspartate receptor (NMDA) |
| ln Vivo |
Both LY235959 and LY 233053 ( < or = 0.5 and 5 mg/kg, respectively) did not influence the electroconvulsive threshold but potentiated the anticonvulsant action of all antiepileptics studied. The combined treatment of LY 233053 (5 mg/kg) with carbamazepine, diphenylhydantoin, or phenobarbital (providing a 50% protection against maximal electroshock) resulted in the impairment of long-term memory. No adverse effects were observed with combinations of LY235959 with these antiepileptics. The combined treatment of valproate with either LY235959 or LY 233053 was superior to valproate alone, as regards motor impairment, but not the impairment of long-term memory. Neither NMDA-receptor antagonist elevated the total plasma levels of antiepileptic drugs studied.
Conclusions: It may be concluded that NMDA-receptor blockade leads to the enhanced anticonvulsive action of conventional antiepileptics against maximal electroshock-induced seizures. A pharmacokinetic interaction does not seem probable. [1] Positive modulators of glutamate alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors can enhance cognitive function in several species. The present experiments compared the actions of a novel biarylpropylsulfonamide compound, LY404187, with the prototypical benzoylpiperidine, 1-(quinoxalin-6-ylcarbonyl)-piperidine (CX516), on AMPA receptors of prefrontal cortex (PFC) pyramidal neurons. LY404187 (0.03-10 microM) selectively enhanced glutamate-evoked currents through AMPA receptor/channels of acutely isolated pyramidal neurons with considerably greater potency (EC50 = 1.3 +/- 0.3 microM) and efficacy (Emax = 45.3 +/- 8.0-fold increase) than did CX516 (EC50 = 2.8 +/- 0.9 mM; Emax = 4.8 +/- 1.4-fold increase). Both LY404187 and CX516 increased the potency of the glutamate concentration-response profile by 6- and 3-fold, respectively. Rapid perfusion experiments demonstrated that LY404187 produced a marked suppression in the magnitude but no change in the kinetics of receptor desensitization; whereas CX516 produced little change in the degree and a modest deceleration of the desensitization process. In PFC slices, both spontaneous and stimulus-evoked AMPA receptor-mediated excitatory postsynaptic potentials were enhanced by nanomolar concentrations of LY404187. Voltage-sensitive N-methyl-D-aspartate (NMDA) receptor-dependent synaptic responses also were indirectly augmented as a consequence of greater postsynaptic depolarization. Consistent with the in vitro data, LY404187 was 1000-fold more potent than CX516 in enhancing the probability of discharge of PFC neurons in response to stimulation of glutamatergic afferents from hippocampus in vivo. This potentiation by LY404187 was reduced by both selective AMPA (LY300168, 1 mg/kg, i.v.) and NMDA (LY235959, 5 mg/kg, i.v.) receptor antagonists. Collectively, these results demonstrate that LY404187 is an extremely potent and centrally active potentiator of native AMPA receptors and has a unique mechanism of action. The therapeutic implications of AMPA receptor potentiators are discussed. [2] |
| Animal Protocol |
Chimney test [1] The effects of AEDs alone or in combination witheither LY235959 or LY 233053 on motor impairmentwere quantified with the chimney test of Boissier etal. (14). In this test, animals had to climb backwardup the plastic tube (3-cm inner diameter, 25-cmlength). Motor impairment was indicated by the in-ability of the animals to climb backward up the tubewithin 60 s, and the results were shown as a percent-age of animals that failed to perform the test. Estimation of the plasma levels of AEDs [1] The animals were administered either saline plusone of the AEDs and LY235959 or LY 233053 plusone of these drugs. The mice were killed by decapita-tion at times scheduled for the convulsive test, andsamples of blood of -1 ml were collected into Ep-pendorf tubes. Samples of blood were centrifuged at 10,000 r/min for 3 min, and plasma samples of 70 pl weretransferred to Abbott System cartridges. The levelsof AEDs were estimated by immunofluorescence,by using an Abbott Tdx analyzer. Plasma levels were expressed asmeans t SD of at least seven determinations. |
| References |
[1]. Competitive NMDA-receptor antagonists, LY 235959 and LY 233053, enhance the protective efficacy of various antiepileptic drugs against maximal electroshock-induced seizures in mice. Epilepsia. 1996 Jul;37(7):618-24. [2]. Positive modulation of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors in prefrontal cortical pyramidal neurons by a novel allosteric potentiator. J Pharmacol Exp Ther. 2001 Jul; 298(1): 86-102. |
| Additional Infomation |
Our study demonstrated that competitive NMDAantagonists, LY 235959 and LY 233053, exerted ananticonvulsant efficacy, elevating the threshold forelectroconvulsions in mice. Both agents, adminis-tered in subthreshold doses, significantly enhanced the protective activity of valproate, carbamazepine,diphenylhydantoin, and phenobarbital against maxi-mal electroshock-induced seizures. A pharmacoki-netic interaction, in terms of total plasma levels atleast, does not seem probable, because plasma levelsof AEDs remained unchanged in the presence ofboth LY 235959 and LY 233053. Our findings support the results of previous stud-ies pointing out that an antagonism at the NMDAreceptor results in the pronounced enhancement ofthe antielectroshock activity of conventional AEDs,with reduced adverse effects in some cases.It may have a significant clinical utility in the treat-ment of epilepsy. Another approach with the use ofnon-NMDA-receptor antagonists for the combinedtreatment with AEDs may also prove promisingfrom the clinical point of view. It is alsoprobable that epilepsy could be controlled by meansof excitatory amino acid antagonists only. Recentdata by Loscher et al. and Czuczwar et al. provided evidence on the potent interaction ofNMDA and non-NMDA-receptor antagonists in thekindling model of epilepsy and electroconvulsions. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6067 mL | 18.0336 mL | 36.0672 mL | |
| 5 mM | 0.7213 mL | 3.6067 mL | 7.2134 mL | |
| 10 mM | 0.3607 mL | 1.8034 mL | 3.6067 mL |