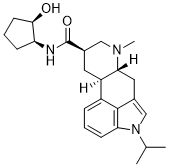
LY 215840 is a novel and highly potent antagonist of the 5-hydroxytryptamine (5-HT)2 receptor with anti-hypertensive and muscle relaxant effects in animal studies.
Physicochemical Properties
| Molecular Formula | C24H33N3O2 |
| Molecular Weight | 395.537726163864 |
| Exact Mass | 395.257 |
| Elemental Analysis | C, 72.88; H, 8.41; N, 10.62; O, 8.09 |
| CAS # | 137328-52-0 |
| PubChem CID | 132049 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.34g/cm3 |
| Boiling Point | 642.1ºC at 760mmHg |
| Flash Point | 342.1ºC |
| Vapour Pressure | 2.3E-17mmHg at 25°C |
| Index of Refraction | 1.689 |
| LogP | 3.99 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 29 |
| Complexity | 625 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | O=C([C@H]1CN(C)[C@]2([H])CC3=CN(C(C)C)C4=CC=CC(=C34)[C@@]2([H])C1)N([H])[C@H]1CCC[C@H]1O |
| InChi Key | IMSDOBUYDTVEHN-ILMFXRJHSA-N |
| InChi Code | InChI=1S/C24H33N3O2/c1-14(2)27-13-15-11-21-18(17-6-4-8-20(27)23(15)17)10-16(12-26(21)3)24(29)25-19-7-5-9-22(19)28/h4,6,8,13-14,16,18-19,21-22,28H,5,7,9-12H2,1-3H3,(H,25,29)/t16-,18-,19+,21-,22-/m1/s1 |
| Chemical Name | Ergoline-8-carboxamide, N-(2-hydroxycyclopentyl)-6-methyl-1-(1-methylethyl)-, (8beta(1S,2R))- |
| Synonyms | LY 215840; LY-215840; LY 215,840; 137328-52-0; LY215,840; D2G5U4P5B3; LY-215,840; SCHEMBL14908006; DTXSID20929769; Ergoline-8-carboxamide, N-(2-hydroxycyclopentyl)-6-methyl-1-(1-methylethyl)-, [8beta(1S,2R)]-; LY215840. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT2 Receptor; 5-HT7 Receptor |
| ln Vitro | The most potent agonist for coronary artery relaxation and one with the highest affinity for the 5-HT7 receptor is 5-carboxamidotryptamine. LY215840 (1 μM) dramatically increases the contractile effects of this agonist [1]. In vitro, 5-HT2 receptors that mediate 5-HT contraction are blocked by LY215840 (3 x 10-10 to 10-8 M) [2]. |
| ln Vivo |
The canine coronary artery possesses a smooth muscle relaxant serotonin (5-HT) receptor distinct from previously characterized 5-HT receptors. On the basis of the ability of LY53857 to block weakly coronary smooth muscle relaxation to 5-HT, we examined several structurally related ergolines in endothelium denuded rings of canine coronary artery precontracted with PGF2 alpha (10 microM). 5-HT (10 nM-100 microM)-induced relaxation was antagonized competitively by the ergoline esters LY53857 (-log KB = 6.5) and sergolexole (-log KB = 6.4) and by the ergoline amide amersergide, (-log KB = 6.7). In contrast to the relatively low affinity of these ergolines, LY215840, another ergoline amide, antagonized 5-HT-induced relaxation in a competitive manner with the highest affinity (-log KB = 8.3). This effect was independent of the 5-HT2 receptor affinity of these ergolines, because LY215840, LY53857, sergolexole and amesergide all possessed similar 5-HT2 receptor affinity. Further, all four ergolines possessed affinity for the human 5-HT7 receptor, and LY215840 had the highest 5-HT7 receptor affinity (Ki = 14.7 nM). Finally, in vascular smooth muscle under basal tone, LY215840 (1 microM) blocked the relaxant response to high concentrations of 5-HT and 5-MeOT without altering their contractile potency. LY215840 (1 microM) did not alter contraction to sumatriptan, an agent that lacks relaxant activity. In contrast, LY215840 (1 microM) markedly potentiated contraction to 5-carboxamidotryptamine, the most potent coronary relaxant agonist and the agonist with the highest 5-HT7 receptor affinity. The ability of LY215840 to block the relaxant 5-HT receptor in canine coronary artery may reflect its 5-HT7 receptor antagonist activity and make it a useful tool to probe the relationship between the 5-HT7 receptor and the coronary vasoactive properties of 5-HT.[1] Certain ergolines are potent and selective 5-hydroxytryptamine (5-HT)2 receptor antagonists. Previous studies with two ergoline esters, LY53857 and sergolexole, documented their potency as 5-HT2 receptor antagonists and their metabolism in rats to a less active metabolite, 1-isopropyl dihydrolysergic acid. LY215840, an ergoline amide, has been identified as a potent 5-HT2 receptor antagonist that is not hydrolyzed to 1-isopropyl dihydrolysergic acid. In the rat jugular vein, LY215840 (3 x 109-10) to 10(-8) M) blocked 5-HT2 receptors mediating contraction to 5-HT in vitro. After i.v. and p.o. administration to rats, LY215840 was a potent 5-HT2 receptor antagonist, documented by its ability to block the pressor response to 5-HT administered i.v. Furthermore, after i.v. and p.o. administration of LY215840, blockade of vascular 5-HT2 receptors persisted in excess of 2 and 6 hr, respectively. LY215840 also blocked vascular 5-HT2 receptors in doses that did not affect alpha-1, beta-1 receptors or angiotensin II pressor responses, documenting the selectivity of LY215840 as an inhibitor of 5-HT2 and not other vascular receptors that modulate vasoconstriction. In addition to inhibiting vascular 5-HT2 receptors, LY215840 also inhibited 5-HT-amplified, ADP-induced aggregation (another 5-HT2 receptor-mediated response) in both rabbit and human platelets. Because of its ability to block both platelet and vascular 5-HT2 receptors, we studied the effectiveness of LY215840 in the rabbit carotid artery model of vascular occlusion. Low i.v. doses of LY215840 markedly prolonged time to vascular occlusion [2]. |
| References |
[1]. LY215840, a high-affinity 5-HT7 receptor ligand, blocks serotonin-induced relaxation in canine coronary artery. J Pharmacol Exp Ther. 1996 Jun;277(3):1560-6. [2]. LY215840, a potent 5-hydroxytryptamine (5-HT)2 receptor antagonist, blocks vascular and platelet 5-HT2 receptors and delays occlusion in a rabbit model of thrombosis. J Pharmacol Exp Ther. 1992 Apr;261(1):202-8. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5282 mL | 12.6409 mL | 25.2819 mL | |
| 5 mM | 0.5056 mL | 2.5282 mL | 5.0564 mL | |
| 10 mM | 0.2528 mL | 1.2641 mL | 2.5282 mL |