LLP-3 is a novel Survivin inhibitor that disrupts Survivin-Ran interaction in cancer cells and may be used for Glioblastoma multiforme (GBM).
Physicochemical Properties
| Molecular Formula | C32H23CLN2O4 |
| Molecular Weight | 534.989027261734 |
| Exact Mass | 534.134 |
| CAS # | 1453835-43-2 |
| PubChem CID | 56935694 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 787.1±60.0 °C at 760 mmHg |
| Flash Point | 429.8±32.9 °C |
| Vapour Pressure | 0.0±2.8 mmHg at 25°C |
| Index of Refraction | 1.704 |
| LogP | 7.62 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 39 |
| Complexity | 929 |
| Defined Atom Stereocenter Count | 0 |
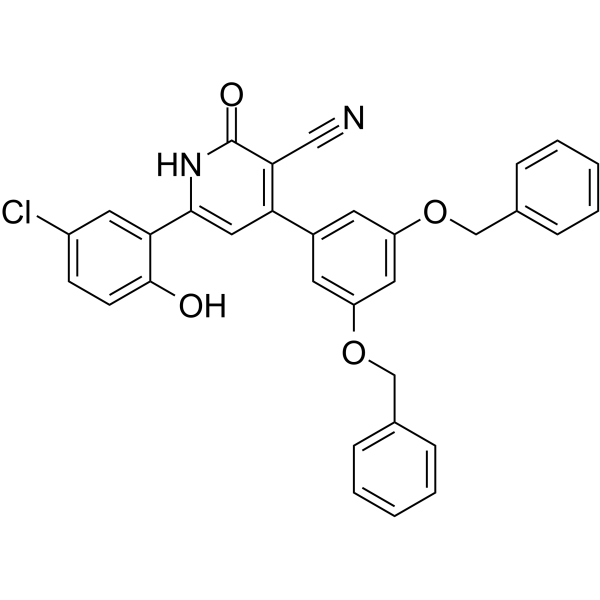
| SMILES | C1(=O)NC(C2=CC(Cl)=CC=C2O)=CC(C2=CC(OCC3=CC=CC=C3)=CC(OCC3=CC=CC=C3)=C2)=C1C#N |
| InChi Key | NZRBRJQYGLEINZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H23ClN2O4/c33-24-11-12-31(36)28(15-24)30-17-27(29(18-34)32(37)35-30)23-13-25(38-19-21-7-3-1-4-8-21)16-26(14-23)39-20-22-9-5-2-6-10-22/h1-17,36H,19-20H2,(H,35,37) |
| Chemical Name | 4-[3,5-bis(phenylmethoxy)phenyl]-6-(5-chloro-2-hydroxyphenyl)-2-oxo-1H-pyridine-3-carbonitrile |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Survivin's binding to Smac/DIABLO, a pro-apoptotic protein, is weakened by LLP-3 (20 μM, 24 hours); however, this effect is not observed in Survivin's interaction with other chromosomal passenger complex (CPC) proteins [1]. In U87 cells, LLP-3 (20 μM, 24 hours) decreases the Survivin-Ran interaction [1]. Tumor cell death results from the arrest of cells in the G0-G1 phase by LLP-3 (20 μM, 24 hours) [1]. In HT1080 cells, LLP-3 (20 μM, 24 hours) causes caspase-dependent apoptosis [1]. By inhibiting p53, LLP-3 (0-100 μM, 72 hours) reduces the survival of tumor cells [1]. |
| ln Vivo | In mice with GBM spheroid tumors, LLP-3 (intraperitoneal injection, 25 mg/kg, for 10 days) can extend survival [1]. |
| Cell Assay |
Cell viability assay [1] Cell Types: U87E6, U87MG Cell Tested Concentrations: 0-100 μM Incubation Duration: 72 h Experimental Results: Inhibit tumor cell survival, IC50 values are 13.6 μM (U87E6) and 38.1 μM (U87MG). Cell cycle analysis [1] Cell Types: U87 Cell Tested Concentrations: 20 μM Incubation Duration: 24 hrs (hours) Experimental Results: The proportion of cells in S phase and G2-M phase diminished (from 9% to 5%, from 25% to 17%), respectively). The G0-G1 cell population increased (from 60% to 74%). Immunofluorescence[1] Cell Types: U87, HT1080 Cell Tested Concentrations: 40 μM Incubation Duration: 24 h Experimental Results: The colocalization of TPX2 and acetylated α-tubulin is weakened. |
| Animal Protocol |
Animal/Disease Models: GBM spheroid tumor model (GBM83 and 1600) [1] Doses: 25 mg/kg Route of Administration: intraperitoneal (ip) injection for 10 days (days 10-14 and 17-21) Experimental Results: Prolonged tumor The survival time of mice did not show any lethality until day 35. |
| References |
[1]. Impairment of glioma stem cell survival and growth by a novel inhibitor for Survivin-Ran protein complex. Clin Cancer Res. 2013 Feb 1;19(3):631-42. |
| Additional Infomation | LLP-3 is a pyridone that is pyridin-2(1H)-one which is substituted at positions 3, 4, and 6 by cyano, 3,5-bis(benzyloxy)phenyl, and 5-chloro-2-hydroxyphenyl groups, respectively. It is an inhibitor for Survivin-Ran protein complex. It has a role as an inhibitor, an antineoplastic agent, a survivin dimerisation modulator and an antimitotic. It is a hydroxynitrile, a pyridone, a member of phenols, a benzyl ether, an aromatic ether and a member of monochlorobenzenes. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8692 mL | 9.3460 mL | 18.6919 mL | |
| 5 mM | 0.3738 mL | 1.8692 mL | 3.7384 mL | |
| 10 mM | 0.1869 mL | 0.9346 mL | 1.8692 mL |