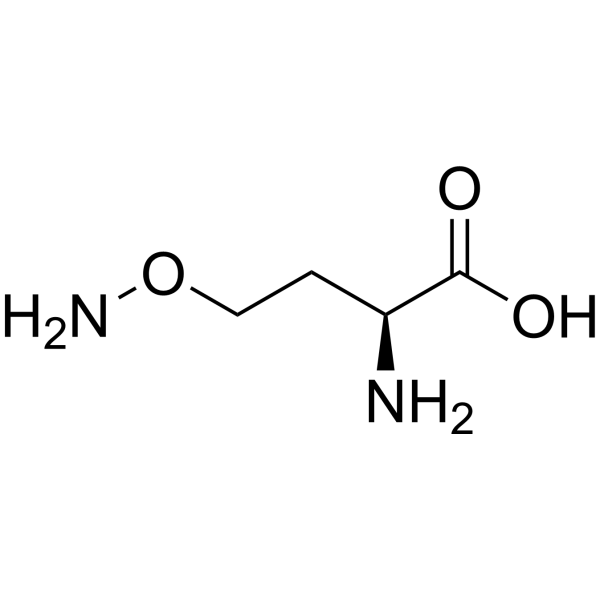
Physicochemical Properties
| Molecular Formula | C4H10N2O3 |
| Molecular Weight | 134.1338 |
| Exact Mass | 134.069 |
| CAS # | 496-93-5 |
| PubChem CID | 441443 |
| Appearance | White to off-white solid powder |
| Density | 1.298g/cm3 |
| Boiling Point | 378.1ºC at 760mmHg |
| Flash Point | 182.5ºC |
| Vapour Pressure | 9.22E-07mmHg at 25°C |
| Index of Refraction | 1.51 |
| LogP | 0.079 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 9 |
| Complexity | 95.8 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | C(CON)[C@@H](C(=O)O)N |
| InChi Key | FQPGMQABJNQLLF-VKHMYHEASA-N |
| InChi Code | InChI=1S/C4H10N2O3/c5-3(4(7)8)1-2-9-6/h3H,1-2,5-6H2,(H,7,8)/t3-/m0/s1 |
| Chemical Name | (2S)-2-amino-4-aminooxybutanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | After being stimulated with phorbol 12-myristate-13-acetate (PMA) or through a mixed lymphocyte reaction, PBMC proliferation was inhibited by L-canaline treatment. The highest effect on cells was produced by PMA, and the IC50 of L-canaline was 0.26 mM. When PBMC are stimulated by a mixed lymphocyte reaction, L-canaline is marginally less toxic (IC50 0.54 mM) [1]. In astrocytes and astrocytoma cells, L-canaline competitively inhibits L-lysine flux (Ki of 4.6 mM) [2]. |
| ln Vivo | Male Wistar rat medulla oblongata aspartate content is decreased by L-canaline, but this has no effect on the non-protein amino acid's induced release into these tissues [2]. When male Sprague-Dawley rats were given an intraadiaphragmatic injection of 100 μg of L-canaline, the amount of ornithine aminotransferase activity in the diaphragm tissue measured an hour later was 90% lower [2]. |
| References |
[1]. The antiproliferative and immunotoxic effects of L-canavanine and L-canaline. Anticancer Drugs. 2002 Mar;13(3):313-20. [2]. Rosenthal GA. L-canaline: a potent antimetabolite and anti-cancer agent from leguminous plants. Life Sci. 1997;60(19):1635-41. [3]. Berger BJ. Antimalarial activities of aminooxy compounds. Antimicrob Agents Chemother. 2000 Sep;44(9):2540-2. |
| Additional Infomation |
L-canaline is a non-proteinogenic L-alpha-amino acid that is L-homoserine in which the hydroxy group at position 4 is substituted by an aminooxy group. It has been isolated from legumes and plays an essential role in lugume chemical defense against insects. It has a role as a plant metabolite, an antineoplastic agent, an antimetabolite and a phytogenic insecticide. It is functionally related to a L-homoserine. It is a tautomer of a L-canaline zwitterion. Canaline has been reported in Vicia faba and Medicago sativa with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.4555 mL | 37.2773 mL | 74.5545 mL | |
| 5 mM | 1.4911 mL | 7.4555 mL | 14.9109 mL | |
| 10 mM | 0.7455 mL | 3.7277 mL | 7.4555 mL |