Levacetylleucine (N-acetyl-L-leucine, NALL) exhibits cellular and animal biological activity indicative of neuroprotective and lysosome-modulating effects. In vitro, at clinically relevant concentrations, NALL triggers nuclear translocation and activation of TFEB, a key regulator of lysosomal biogenesis and autophagy. This leads to the increased expression of TFEB target genes, such as LAMP1—effects not produced by L-leucine or its D-enantiomer. In vivo, studies in *Npc1-/-* mice (a model for Niemann-Pick disease type C) show that NALL treatment reduces glycosphingolipid accumulation (e.g., GM1a, GM2, GA2) in the forebrain and cerebellum, decreases cerebellar neuroinflammation (as indicated by CD68-positive microglia), improves motor coordination, and modestly extends survival when administered pre-symptomatically. Further research in rats and mice reported enhanced postural recovery following labyrinthectomy (with 60 mg/kg IV dosing for 15 days) and reduced cortical neuronal loss after traumatic brain injury (with 100 mg/kg oral daily dosing for 28 days).
Levacetylleucine (N-acetyl-L-leucine) is an orally available and brain-penetrant acetylated derivative of the amino acid leucine, serving as the active form of N-acetyl-leucine (NAL). It demonstrates neuroprotective properties by attenuating neuronal death and neuroinflammation in mouse cortical tissue, while also improving lysosomal and metabolic dysfunction. In animal models, the compound aids in the recovery of postural symptoms following unilateral chemical labyrinthectomy in rats. These characteristics make Levacetylleucine a promising candidate for research into the neurological manifestations of Niemann-Pick disease type C, as well as for potential applications in traumatic brain injury and the prevention of neurodegeneration.
Physicochemical Properties
| Molecular Formula | C8H15NO3 |
| Molecular Weight | 173.2096 |
| Exact Mass | 173.105 |
| Elemental Analysis | C, 55.47; H, 8.73; N, 8.09; O, 27.71 |
| CAS # | 1188-21-2 |
| Related CAS # | 1188-21-2;121-76-6 (monoethanolamine); |
| PubChem CID | 70912 |
| Sequence | Ac-Leu-OH; N-acetyl-L-leucine |
| SequenceShortening | L |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 369.6±25.0 °C at 760 mmHg |
| Melting Point | 187-190 °C(lit.) |
| Flash Point | 177.4±23.2 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.458 |
| LogP | 0.34 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 12 |
| Complexity | 177 |
| Defined Atom Stereocenter Count | 1 |
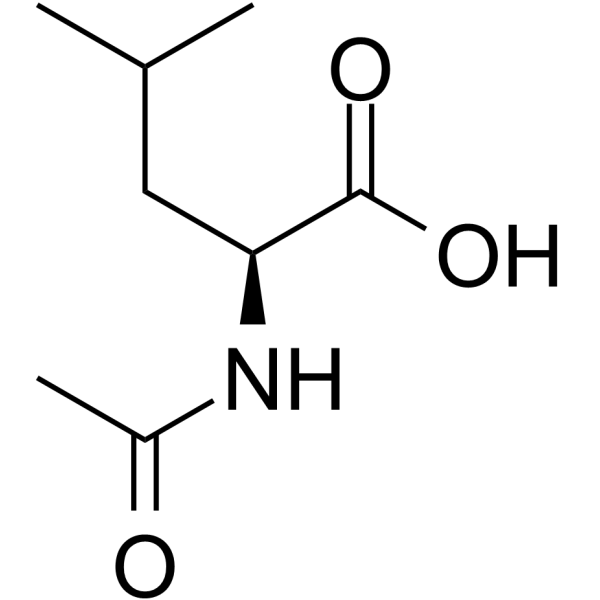
| SMILES | CC(C)C[C@@H](C(=O)O)NC(=O)C |
| InChi Key | WXNXCEHXYPACJF-ZETCQYMHSA-N |
| InChi Code | InChI=1S/C8H15NO3/c1-5(2)4-7(8(11)12)9-6(3)10/h5,7H,4H2,1-3H3,(H,9,10)(H,11,12)/t7-/m0/s1 |
| Chemical Name | (2S)-2-acetamido-4-methylpentanoic acid |
| Synonyms | N-Acetyl-L-leucine; Levacetylleucine; 1188-21-2; N-Acetylleucine; L-Acetylleucine; (S)-2-Acetamido-4-methylpentanoic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Neuroprotective amino acid |
| ln Vitro |
N-acetyl-L-leucine is the N-acetyl derivative of L-leucine. It has a role as a metabolite. It is a N-acetyl-L-amino acid and a L-leucine derivative. It is a conjugate acid of a N-acetyl-L-leucinate. It is an enantiomer of a N-acetyl-D-leucine.
Levacetylleucine (L-acetylleucine) is a modified acetylated derivative of the amino acid [leucine]. The racemic (i.e. DL-) form has been available in France since 1957 for the treatment of vertigo. Observational studies in patients with Niemann-Pick disease type C (NPC) suggested a beneficial symptomatic as well as neuroprotective disease-modifying effect of the DL-form of N-acetylleucine, after which animal studies showed that the L-enantiomer (i.e. levacetylleucine) has potential clinical benefits compared to the racemic mixture. In September 2024, the FDA approved levacetylleucine for the treatment of select patients with NPC. The mechanism of action of levacetylleucine is as a P-Glycoprotein Inhibitor, and Breast Cancer Resistance Protein Inhibitor, and Bile Salt Export Pump Inhibitor, and Organic Anion Transporter 1 Inhibitor, and Organic Anion Transporter 3 Inhibitor. |
| ln Vivo | Levacetylleucine is indicated for the treatment of neurological symptoms of Niemann-Pick disease type C (NPC) in adults and pediatric patients weighing ≥15 kg. LEVACETYLLEUCINE is a small molecule drug with a maximum clinical trial phase of IV (across all indications) that was first approved in 2024 and is indicated for niemann-pick disease type c and has 3 investigational indications. |
| ADME/Pharmacokinetics |
Absorption The Cmaxand AUC0-24h of levacetylleucine following oral administration were 8.3 μg/mL and 33.2 h*μg/mL, respectively. The time to maximum concentration (Tmax) is approximately 1 hour. Volume of Distribution The apparent volume of distribution following oral administration of levacetylleucine is 253 liters. Clearance The apparent oral clearance of levacetylleucine is 139 L/h. Metabolism / Metabolites Levacetylleucine is metabolized into acetate and L-leucine by ubiquitously expressed enzymes. Biological Half-Life The half-life of levacetylleucine is approximately 1 hour. |
| Toxicity/Toxicokinetics |
Critically, levacetylleucine was observed to be well-tolerated in patients throughout the trial. The most common adverse reactions, according to IntraBio, were abdominal pain, upper respiratory tract infections, vomiting, and dysphagia. There were no adverse events (AEs) that led to the premature discontinuation of the trial; though 3 patients each had 1 AE that the investigator assessed to be related to levacetylleucine. Some limitations of the trial were reported by the original investigators. They noted that the 12-week duration of the study limited their ability to provide insight into the effect of levacetylleucine on disease progression and AE incidence. Furthermore, some groups of patients were excluded due to the focus on symptomatic end points. However, these results confirmed the significant efficacy of levacetylleucine for patients with this rare condition, leading to its regulatory approval. According to Factor, clinical research indicates that levacetylleucine could potentially treat other types of neurodevelopmental and neurodegenerative disorders. “We will continue to rapidly develop Aqneursa [levacetylleucine] for these additional indications,” [1]. https://www.pharmacytimes.com/view/levacetylleucine-receives-fda-approval-as-stand-alone-therapy-for-niemann-pick-disease-type-c [2]. Trial of n-acetyl-l-leucine in Niemann-Pick disease type c. N Engl J Med. 2024;390:421-431. doi:10.1056/NEJMoa231 |
| References |
[1]. Trial of N-Acetyl-l-Leucine in Niemann-Pick Disease Type C. N Engl J Med. 2024 Feb 1;390(5):421-431. [2]. N-acetyl-L-leucine: a promising treatment option for traumatic brain injury. Neural Regen Res. 2022 Sep;17(9):1957-1958. [3]. N-acetyl-L-leucine accelerates vestibular compensation after unilateral labyrinthectomy by action in the cerebellum and thalamus. PLoS One. 2015 Mar 24;10(3):e0120891. [4]. N-Acetyl-L-leucine improves functional recovery and attenuates cortical cell death and neuroinflammation after traumatic brain injury in mice. Sci Rep. 2021 Apr 29;11(1):9249. |
| Additional Infomation |
N-acetyl-L-leucine is the N-acetyl derivative of L-leucine. It has a role as a metabolite. It is a N-acetyl-L-amino acid and a L-leucine derivative. It is a conjugate acid of a N-acetyl-L-leucinate. It is an enantiomer of a N-acetyl-D-leucine. N-Acetyl-L-leucine has been reported in Drosophila melanogaster, Brassica napus, and other organisms with data available. See also: Acetylleucine (annotation moved to). Mechanism of Action: Following oral administration, levacetylleucine is taken up via ubiquitously expressed monocarboxylate transporters and distributed to all body tissues. It enters enzyme-controlled pathways that correct metabolic dysfunction and improves adenosine triphosphate (ATP) energy production. The normalization of energy metabolism ameliorates mitochondrial and lysosomal dysfunction and leads to a reduction in the storage of unesterified cholesterol and sphingolipids. Levacetylleucine has also been shown to normalize neuronal membrane potentials in a guinea pig model, thereby improving cellular signaling processes and restoring and protecting neuronal circuits. The specific molecular target of levacetylleucine in the context of Niemann-Pick disease type C (NPC) is unknown. Patients and families in the NPC community have long awaited an effective, FDA-approved treatment, and we are proud to bring hope to those affected by this devastating disease,” Mallory Factor, president and CEO of IntraBio, said in a news release. This approval follows the recent clearance of arimoclomol (Miplyffa; Zevra Therapeutics), an oral therapy for the treatment of patients with NPC. FDA approved arimoclomal in combination with miglustat, an enzyme inhibitor. Though this combination was the first FDA-approved NPC treatment, yesterday’s announcement marks the first approval of a stand-alone therapy for NPC. FDA investigators assessed the primary outcome using the Scale for the Assessment and Rating of Ataxia, referred to as the functional SARA (fSARA), which assesses factors such as gait, stability, and speech. Results from the trial indicated that patients who received levacetylleucine compared with placebo demonstrated a greater improvement in fSARA score. After 12 weeks of receiving levacetylleucine, the mean change from baseline SARA score was -1.97±2.43; for those treated with placebo, the difference was -0.60±2.39 points. Investigators determined a mean treatment difference of -0.4 (95% CI: -0.7, -0.2), with a 2-sided p-value of < .001. These results were supported by consistent results that the original SARA indicated. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~577.33 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (14.43 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (14.43 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (14.43 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.7733 mL | 28.8667 mL | 57.7334 mL | |
| 5 mM | 1.1547 mL | 5.7733 mL | 11.5467 mL | |
| 10 mM | 0.5773 mL | 2.8867 mL | 5.7733 mL |