Physicochemical Properties
| Molecular Formula | C5H10N2O2S |
| Molecular Weight | 162.2101 |
| Exact Mass | 162.046 |
| CAS # | 38520-57-9 |
| PubChem CID | 10176265 |
| Appearance | White to off-white solid powder |
| LogP | 0.896 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 10 |
| Complexity | 149 |
| Defined Atom Stereocenter Count | 1 |
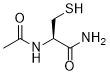
| SMILES | CC(N[C@@H](CS)C(N)=O)=O |
| InChi Key | UJCHIZDEQZMODR-BYPYZUCNSA-N |
| InChi Code | InChI=1S/C5H10N2O2S/c1-3(8)7-4(2-10)5(6)9/h4,10H,2H2,1H3,(H2,6,9)(H,7,8)/t4-/m0/s1 |
| Chemical Name | (2R)-2-acetamido-3-sulfanylpropanamide |
| Synonyms | NACANac amide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | N-acetylcysteine amide exhibited considerable cytotoxicity at 10–20 mM, although it had no discernible influence on the viability of H9c2 cells treated with <1 mM doxorubicin (DOX). N-acetyl cysteine amide (750 μM) decreases ROS levels and lipid peroxidation caused by DOX, and it also restores the GSH/GSSG ratio and antioxidant enzyme activity, including catalase (CAT), glutathione reductase (GR), and glutathione peroxidase (GPx) [1]. Methamphetamine (METH)-induced cell death is prevented in human brain microvascular endothelium (HBMVEC) by N-acetylcysteine amide (1 mM) [3]. |
| ln Vivo | The central nervous system is more bioavailable when N-acetylcysteine amide is present. In rats with traumatic brain injury (TBI), N-acetylcysteine amide (150 mg/kg, i.p.) increases mitochondrial bioenergetics, decreases oxidative stress, preserves mitochondrial trough, and enhances cortical protection and functional outcomes[2]. |
| References |
[1]. N-acetylcysteine amide decreases oxidative stress but not cell death induced by doxorubicin in H9c2 cardiomyocytes. BMC Pharmacol. 2009 Apr 15;9:7. [2]. N-acetylcysteine amide confers neuroprotection, improves bioenergetics and behavioral outcome following TBI. Exp Neurol. 2014 Jul;257:106-13. [3]. N-Acetylcysteine amide protects against methamphetamine-induced oxidative stress and neurotoxicity in immortalized human brain endothelial cells. Brain Res. 2009 Jun 12;1275:87-95. |
| Additional Infomation | N-Acetylcysteine Amide is the amide form of N-acetylcysteine (NAC), a synthetic N-acetyl derivative and prodrug of the endogenous amino acid and antioxidant glutathione (GSH) precursor L-cysteine, with potential antioxidant and anti-inflammatory activities. Upon administration, N-acetylcysteine amide (NACA) increases GSH levels. GSH scavenges reactive oxygen species (ROS), reduces oxidative stress and prevents ROS-mediated cell damage and apoptosis. NACA has enhanced lipophilicity and membrane permeability compared with NAC. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~200 mg/mL (~1232.97 mM) DMSO : ≥ 100 mg/mL (~616.48 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (15.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (15.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (15.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (616.48 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.1648 mL | 30.8242 mL | 61.6485 mL | |
| 5 mM | 1.2330 mL | 6.1648 mL | 12.3297 mL | |
| 10 mM | 0.6165 mL | 3.0824 mL | 6.1648 mL |