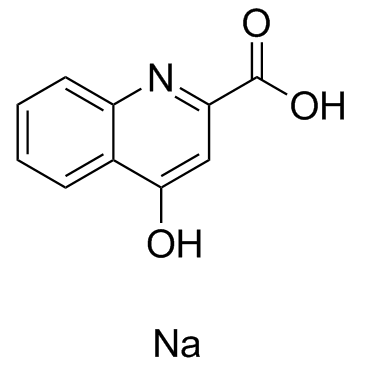
Kynurenic acid sodium is an endogenous tryptophan metabolite with activity against NMDA, glutamate, α7 nicotinic acetylcholine receptors. Also acting as an agonist of GPR35/CXCR8.
Physicochemical Properties
| Molecular Formula | C10H6NNAO3 |
| Molecular Weight | 211.14931344986 |
| Exact Mass | 211.024 |
| CAS # | 2439-02-3 |
| Related CAS # | Kynurenic acid;492-27-3 |
| PubChem CID | 52974250 |
| Appearance | Light yellow to gray solid powder |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 15 |
| Complexity | 314 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Na+].O=C1C=C(C(=O)[O-])NC2C=CC=CC=21 |
| InChi Key | RCAZGXKUQDXSSK-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C10H7NO3.Na/c12-9-5-8(10(13)14)11-7-4-2-1-3-6(7)9;/h1-5H,(H,11,12)(H,13,14);/q;+1/p-1 |
| Chemical Name | sodium;4-oxo-1H-quinoline-2-carboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Kynurenic acid binds to orphan G protein-coupled receptor GPR35, with an EC50 value of 3.3 μM for intracellular Ca²⁺ mobilization in GPR35-transfected CHO cells [1] - Kynurenic acid acts as an antagonist of α7 nicotinic acetylcholine receptors (α7 nAChR), with an IC50 value of 7.6 μM for inhibiting ACh-induced currents in Xenopus oocytes expressing human α7 nAChR [2] |
| ln Vitro |
GPR35 is a receptor for kynurenic acid, an intermediary in the kynurenine process. In the presence of G qi/o chimeric G proteins, kynurenic acid causes calcium mobilization and inositol phosphate synthesis in a manner that is dependent on GPR35. In GPR35-expressing cells, kynurenic acid increases [35S]guanosine 5′-O-(3-thiotriphosphate) binding; treatment with pertussis toxin reverses this effect. Moreover, kynurenic acid causes GPR35 to internalize[1]. The neuroprotective, anticonvulsant, and neuroinhibitory properties of KYNA are seen at millimolar concentrations of the molecule. The observation that KYNA concentrations in the mammalian brain are in the sub-micromolar range, coupled with the low affinity of KYNA at each of the three ionotropic glutamate receptors responsible for these effects (NMDA, alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA), and kainate), suggested that other receptors might be targets of endogenous kynurenic acid. With an IC50 in the low micromolar range, kynurenic acid antagonizes α7nAChRs on cultured hippocampus neurons non-competitively and with a steeper inhibition curve[2]. - In GPR35-transfected CHO cells, kynurenic acid (1 μM - 100 μM) dose-dependently induced intracellular Ca²⁺ mobilization, which was blocked by pertussis toxin, indicating coupling to Gi/o proteins [1] - In human α7 nAChR-expressing Xenopus oocytes, kynurenic acid (1 μM - 100 μM) competitively inhibited acetylcholine (ACh)-evoked inward currents, with no agonist activity observed [2] - In rat cortical synaptosomes, kynurenic acid (10 μM - 100 μM) reduced ACh release induced by K⁺ depolarization, consistent with α7 nAChR antagonism [2] |
| ln Vivo |
Leukocyte activity in the peripheral blood of mice is influenced by kynurenic acid; however, the most significant effect is produced by the lowest concentration (2.5 mg/L) and the maximum concentration (250 mg/L) has the least effect. After 7 and 28 days, an animal's T lymphocyte proliferative response is stimulated (p<0.05) by a dosage of kynurenic acid[3]. - Oral administration of kynurenic acid (50 mg/kg, 100 mg/kg, 200 mg/kg) to BALB/c mice for 7 consecutive days significantly increased the percentage of CD4⁺ T cells and decreased the percentage of CD8⁺ T cells in peripheral blood leukocytes [3] - At doses of 100 mg/kg and 200 mg/kg, oral kynurenic acid enhanced the proliferative response of peripheral blood leukocytes to concanavalin A (Con A) and lipopolysaccharide (LPS) in mice [3] - Oral kynurenic acid (200 mg/kg) increased the production of IL-2 and IFN-γ in mouse peripheral blood leukocytes stimulated with Con A [3] |
| Enzyme Assay |
CHO-GPR35 stable cells are pretreated with or without pertussis toxin (100 ng/mL) for 16 h before harvesting. Cells are resuspended and homogenized in 10 mM Tris-HCl (pH 7.4), 1 mM EDTA followed by centrifugation at 1000 ×g for 10 min at 4 °C to remove nuclei and cellular debris. Membrane fractions are collected by spinning the supernatant at 38,000 ×g for 30 min and resuspended in 20 mM HEPES (pH 7.5) and 5 mM MgCl2. 25 μg of membranes is incubated at room temperature for 1 h in assay buffer (20 mM HEPES, 5 m MMgCl2, 0.1% bovine serum albumin (pH 7.5)) containing 3 μM GDP and 0.1 nM[35S]GTPγS in the absence or presence of kynurenic acid. Reactions are terminated by vacuum filtration through GF/B filters, and the retained radioactivities are quantified on liquid scintillation counter[1]. - For GPR35 binding and functional assay: GPR35-transfected CHO cells were seeded in 96-well plates and loaded with a Ca²⁺-sensitive fluorescent dye. After incubation, kynurenic acid at various concentrations was added, and fluorescence intensity was measured to assess intracellular Ca²⁺ mobilization. Dose-response curves were generated to calculate EC50 [1] - For α7 nAChR antagonism assay: Xenopus oocytes were injected with human α7 nAChR cRNA. Two to three days later, oocytes were voltage-clamped, and ACh was applied to evoke currents. Kynurenic acid at different concentrations was co-applied with ACh, and current inhibition was recorded to determine IC50 [2] |
| Cell Assay |
- For synaptosomal ACh release assay: Rat cerebral cortex was homogenized and fractionated to isolate synaptosomes. Synaptosomes were preincubated with kynurenic acid for 15 min, then depolarized with KCl. Released ACh was quantified using a radiometric assay [2] - For leukocyte subset analysis: Peripheral blood from mice was collected, and leukocytes were isolated by density gradient centrifugation. Cells were stained with fluorochrome-conjugated antibodies against CD4 and CD8, then analyzed by flow cytometry [3] - For leukocyte proliferation assay: Isolated mouse peripheral blood leukocytes were cultured with Con A or LPS in the presence of kynurenic acid-treated serum. Cell proliferation was measured by MTT assay [3] |
| Animal Protocol |
Mouse: The experiment is performed on 160 male BALB/c mice, aged 10-12 weeks, with body weight of 22-26 g. The animals are maintained on a 12-h light/dark cycle at controlled temperature (20 ±1°C) and supplied with rodent chow and water ad libitum throughout the experiment. Mice are divided randomLy into four equal groups: control group (0) not receiving the Kynurenic acid, and three experimental groups administered the Kynurenic acid solution in drinking water at concentrations of 2.5, 25 or 250 mg/L. After 3, 7, 14 and 28 consecutive days of administration of the Kynurenic acid solution, 10 individuals from each group are sacrificed. The animals are anesthetized by inhalation of Aerrane and their blood is collected by heart puncture. Blood collected from five individuals of each group is used for the MTT assay, and from the next five for the flow cytometry[3]. - Animals: BALB/c mice (6-8 weeks old, both sexes) were used and housed under standard laboratory conditions with free access to food and water [3] - Drug administration: Kynurenic acid was dissolved in 0.9% NaCl solution, and administered orally via gavage at doses of 50 mg/kg, 100 mg/kg, or 200 mg/kg once daily for 7 consecutive days. Control group received equal volume of 0.9% NaCl [3] - Sample collection: 24 hours after the last administration, mice were anesthetized, and peripheral blood was collected by cardiac puncture for leukocyte isolation and analysis [3] |
| Toxicity/Toxicokinetics |
- No acute toxicity or adverse effects were observed in mice after oral administration of kynurenic acid at doses up to 200 mg/kg for 7 days [3] |
| References |
[1]. Kynurenic acid as a ligand for orphan G protein-coupled receptor GPR35. J Biol Chem. 2006 Aug 4;281(31):22021-8. [2]. Kynurenic acid as an antagonist of α7 nicotinic acetylcholine receptors in the brain: facts and challenges. Biochem Pharmacol. 2013 Apr 15;85(8):1027-32. [3]. Effect of oral administration of kynurenic acid on the activity of the peripheral blood leukocytes in mice. Cent Eur J Immunol. 2014;39(1):6-13. |
| Additional Infomation |
- Kynurenic acid is an endogenous metabolite of the kynurenine pathway of tryptophan degradation [1][2][3] - GPR35 activation by kynurenic acid may be involved in immune regulation and inflammatory responses [1] - α7 nAChR antagonism by kynurenic acid in the brain may contribute to modulation of cholinergic neurotransmission and cognitive functions [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~50 mg/mL (~236.80 mM) H2O : ~0.1 mg/mL (~0.47 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7360 mL | 23.6798 mL | 47.3597 mL | |
| 5 mM | 0.9472 mL | 4.7360 mL | 9.4719 mL | |
| 10 mM | 0.4736 mL | 2.3680 mL | 4.7360 mL |