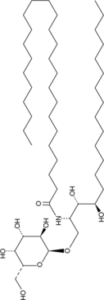
KRN 7000 is a novel, potent and selective synthetic α-
Physicochemical Properties
| Molecular Formula | C50H99NO9 |
| Molecular Weight | 858.32236 |
| Exact Mass | 857.731 |
| Elemental Analysis | C, 69.97; H, 11.63; N, 1.63; O, 16.78 |
| CAS # | 158021-47-7 |
| PubChem CID | 2826713 |
| Appearance | White to off-white solid powder |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 939.8±65.0 °C at 760 mmHg |
| Melting Point | 190 °C |
| Flash Point | 522.2±34.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.512 |
| LogP | 15.96 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 44 |
| Heavy Atom Count | 60 |
| Complexity | 928 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | CCCCCCCCCCCCCCCCCCCCCCCCCC(=O)N[C@@H](CO[C@@H]1[C@@H]([C@H]([C@H]([C@H](O1)CO)O)O)O)[C@@H]([C@@H](CCCCCCCCCCCCCC)O)O |
| InChi Key | VQFKFAKEUMHBLV-BYSUZVQFSA-N |
| InChi Code | InChI=1S/C50H99NO9/c1-3-5-7-9-11-13-15-17-18-19-20-21-22-23-24-25-26-27-29-31-33-35-37-39-45(54)51-42(41-59-50-49(58)48(57)47(56)44(40-52)60-50)46(55)43(53)38-36-34-32-30-28-16-14-12-10-8-6-4-2/h42-44,46-50,52-53,55-58H,3-41H2,1-2H3,(H,51,54)/t42-,43+,44+,46-,47-,48-,49+,50-/m0/s1 |
| Chemical Name | N-((2S,3S,4R)-3,4-dihydroxy-1-(((2S,3R,4S,5R,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)octadecan-2-yl)hexacosanamide |
| Synonyms | KRN-7000; KRN 7000; KRN7000; 158021-47-7; alpha-GalCer; Krn 7000; Krn-7000; alpha-Galactosylceramide; .alpha.-galcer; CCRIS 8968; KRN7000; α-Galactosylceramide. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CD1d; NKT cells; TCR
The target of KRN 7000 is cluster of differentiation 1d (CD1d), a non-classical major histocompatibility complex (MHC) class I-like molecule that presents lipid antigens to invariant natural killer T (iNKT) cells. For mouse CD1d-mediated iNKT cell activation, the half-maximal effective dose (ED₅₀) in B16 melanoma-bearing mice is ~2 μg/kg (intravenous injection) [4] ; for human CD1d, it induces activation of Vα24⁺ iNKT cells with an optimal concentration of 100 ng/mL in vitro [3] . It exhibits high specificity for CD1d, with no binding to classical MHC class I/II molecules or other lipid antigen-presenting molecules [1,3] |
| ln Vitro |
Supernatants from activated Vα24+ NKT cell cultures treated with monocyte-derived dendritic cells (Mo-DC) pulsed with α-galactosylceramide showed antiproliferative action against melanoma cells. The main cause of this impact is the release of IL-12 and, to a lesser extent, IFN-γ. There is also the release of other cytokines, such as IL-4 and IL-10, but their antiproliferative effects are not as strong. Soluble mediators' anti-proliferative actions enable Vα24+ NKT cells, driven by α-galactosylceramide-pulsed Mo-DC, to exhibit anti-tumor activity against human melanoma [3]. 1. Activation of human invariant Vα24⁺ NKT cells: - Peripheral blood mononuclear cells (PBMCs) from healthy donors were enriched for Vα24⁺ iNKT cells. Incubation with KRN 7000 (100 ng/mL) for 7 days induced iNKT cell proliferation (3.5-fold increase vs. vehicle) and activation (CD69⁺ expression up to 85%) [3] - Activated iNKT cells secreted high levels of Th1/Th2 cytokines: interferon-γ (IFN-γ, 1200 ± 150 pg/mL) and interleukin-4 (IL-4, 350 ± 50 pg/mL) (ELISA), with a Th1-dominant profile [3] 2. Antitumor activity against melanoma cells: - Activated Vα24⁺ iNKT cells (stimulated with KRN 7000) exhibited dose-dependent cytotoxicity against human melanoma cell lines (M14, A375, SK-MEL-28) in ⁵¹Cr-release assay. At an effector-to-target (E:T) ratio of 10:1, specific lysis rates were 62% (M14), 58% (A375), and 55% (SK-MEL-28), vs. <10% in vehicle-stimulated iNKT cells [3] - The cytotoxicity was CD1d-dependent: blocking CD1d with neutralizing antibody reduced lysis rate by 70% [3] 3. Immune regulatory effects: - KRN 7000 (100 ng/mL) enhanced the cross-talk between CD1d⁺ antigen-presenting cells (APCs) and iNKT cells, promoting APC maturation (CD80/CD86 upregulation by 2.0–2.5-fold) and subsequent activation of naive T cells (proliferation increased by 3.0-fold) [1] |
| ln Vivo |
Mice treated with α-galactosylceramide do not develop primary tumors on their own when exposed to carcinogens or oncogenes. C-glycoside analogs of α-galactosylceramide that selectively induce IFN-γ synthesis were more efficient than α-galactosylceramide in inhibiting B16 melanoma metastasis, which is consistent with a key role for IFN-γ in NKT cell-mediated tumor responses. Amides function better [1]. α-galactosylceramide is regarded as a biological response modulator and non-specific heterosexual immune stimulator [4]. 1. Antitumor efficacy in B16 melanoma-bearing mice: - C57BL/6 mice (6–8 weeks old) were subcutaneously inoculated with 2×10⁵ B16-F10 melanoma cells. KRN 7000 was administered via intravenous injection on day 3 post-inoculation at doses of 0.1, 1, 10 μg/kg [4] - 0.1 μg/kg: No significant tumor growth inhibition [4] - 1 μg/kg: Inhibited tumor growth by 58% vs. vehicle; extended median survival from 18 days (vehicle) to 26 days [4] - 10 μg/kg: Inhibited tumor growth by 82% vs. vehicle; median survival extended to 35 days; 30% of mice achieved complete tumor regression [4] 2. Immune mechanism in vivo: - KRN 7000 (10 μg/kg) activated hepatic and splenic iNKT cells, increasing IFN-γ secretion in serum (800 ± 100 pg/mL vs. 50 ± 10 pg/mL in vehicle) and enhancing NK cell cytotoxicity (specific lysis of YAC-1 cells increased by 45%) [4] - Depletion of iNKT cells (via anti-Vα14 antibody) abolished the antitumor effect of KRN 7000, confirming iNKT cell dependence [4] |
| Enzyme Assay |
1. Mouse CD1d-iNKT cell binding/activation assay: - CD1d⁺ CHO cells (stable transfectants) were seeded into 96-well plates at 2×10⁴ cells/well and cultured overnight [1] - Serial dilutions of KRN 7000 (0.01–1000 ng/mL) or vehicle were added to the wells and incubated for 4 hours at 37°C with 5% CO₂ to allow CD1d binding and antigen presentation [1] - Hepatic iNKT cells were isolated from C57BL/6 mice, labeled with CFSE, and added to CD1d⁺ CHO cells at a ratio of 1:1 (iNKT cell:CHO cell) [1] - After 72 hours of co-incubation, iNKT cell proliferation (CFSE dilution) and activation (CD69 expression) were analyzed by flow cytometry. The concentration of KRN 7000 inducing 50% iNKT cell proliferation was defined as the functional binding EC₅₀ [1] 2. Human CD1d-Vα24⁺ iNKT cell activation assay: - Human CD1d⁺ dendritic cells (DCs) were generated from PBMC-derived monocytes and seeded into 24-well plates [3] - KRN 7000 (10–1000 ng/mL) was added to DCs and incubated for 6 hours. Enriched human Vα24⁺ iNKT cells were added at a DC:iNKT cell ratio of 1:5 [3] - After 48 hours, culture supernatants were collected to measure IFN-γ and IL-4 concentrations by ELISA, and iNKT cell activation was assessed by CD69 expression via flow cytometry [3] |
| Cell Assay |
α-galactosylceramide (KRN 7000, α-GalCer) has shown potent in vivo anti-tumour activity in mice, including against melanoma and the highly specific effect of inducing proliferation and activation of human Vα24+NKT-cells. We hypothesized that human Vα24+NKT-cells activated by α-GalCer might exhibit anti-tumour activity against human melanoma. To investigate this, Vα24+NKT-cells were generated from the peripheral blood of patients with melanoma after stimulation with α-GalCer pulsed monocyte-derived dendritic cells (Mo-DCs). Vα24+NKT-cells did not exhibit cytolytic activity against the primary autologous or allogeneic melanoma cell lines tested. However, proliferation of the melanoma cell lines was markedly suppressed by co-culture with activated Vα24+NKT-cells (mean ± SD inhibition of proliferation 63.9 ± 1.3%). Culture supernatants of activated Vα24+NKT-cell cultures stimulated with α-GalCer pulsed Mo-DCs exhibited similar antiproliferative activities against melanoma cells, indicating that the majority of the inhibitory effects were due to soluble mediators rather than direct cell-to-cell interactions. This effect was predominantly due to release of IFN-γ, and to a lesser extent IL-12. Other cytokines, including IL-4 and IL-10, were released but these cytokines had less antiproliferative effects. These in vitro results show that Vα24+NKT-cells stimulated by α-GalCer-pulsed Mo-DCs have anti-tumour activities against human melanoma through antiproliferative effects exerted by soluble mediators rather than cytolytic effects as observed against some other tumours. Induction of local cytokine release by activated Vα24+NKT-cells may contribute to clinical anti-tumour effects of α-GalCer[3]. 1. Human iNKT cell enrichment and activation assay: - Peripheral blood was collected from healthy donors, and PBMCs were isolated by Ficoll-Hypaque density gradient centrifugation [3] - Vα24⁺ iNKT cells were enriched using magnetic bead-based positive selection (anti-Vα24 antibody-conjugated beads) [3] - Enriched iNKT cells were seeded into 24-well plates at 1×10⁵ cells/well, stimulated with KRN 7000 (100 ng/mL) plus IL-2 (100 U/mL), and cultured for 7 days [3] - iNKT cell proliferation was measured by ³H-thymidine incorporation, and activation markers (CD69, CD44) were analyzed by flow cytometry [3] 2. Melanoma cell cytotoxicity assay (⁵¹Cr-release assay): - Human melanoma cells (M14, A375) were labeled with ⁵¹Cr (100 μCi/1×10⁶ cells) for 1 hour at 37°C, then washed three times to remove unincorporated ⁵¹Cr [3] - Labeled melanoma cells (1×10⁴ cells/well) were co-cultured with KRN 7000-activated iNKT cells at E:T ratios of 1:1, 5:1, 10:1, 20:1 in 96-well U-bottom plates [3] - After 4 hours of incubation at 37°C, 50 μL of supernatant was collected from each well, and radioactivity was measured using a gamma counter [3] - Specific lysis rate (%) = [(experimental release - spontaneous release)/(maximum release - spontaneous release)] × 100 [3] |
| Animal Protocol |
Agelasphin-9b, (2S,3S,4R)-1-O-(alpha-D-galactopyranosyl)-16-methyl-2- [N-((R)-2- hydroxytetracosanoyl)-amino]- 1,3,4-heptadecanetriol, is a potent antitumor agent isolated from the marine sponge Agelas mauritianus. Various analogues of agelasphin-9b (a lead compound) were synthesized, and the relationship between their structures and biological activities was examined using several assay systems. From the results, KRN7000, (2S,3S,4R)-1-O-(alpha-D- galactopyranosyl)-2-(N-hexacosanoylamino)-1,3,4-octadecanetriol , was selected as a candidate for clinical application, as it has activity against B16-bearing mice. 1. B16-F10 melanoma xenograft model: - Male C57BL/6 mice (6–8 weeks old, 18–22 g) were randomly divided into 4 groups (n=8 per group): vehicle (0.1% BSA in sterile saline), KRN 7000 0.1 μg/kg, 1 μg/kg, 10 μg/kg [4] - B16-F10 melanoma cells (2×10⁵ cells/0.2 mL) were subcutaneously injected into the right flank of each mouse [4] - On day 3 post-tumor inoculation, KRN 7000 was administered via tail vein injection (volume: 100 μL/mouse). Vehicle group received the same volume of 0.1% BSA-saline [4] - Tumor volume was measured every 3 days using calipers (volume = length × width² / 2), and mouse survival was recorded daily [4] - For immune mechanism analysis, a subset of mice (n=3 per group) was euthanized on day 7 post-administration. Liver and spleen were collected to isolate iNKT cells and NK cells, which were analyzed for activation status (CD69, NKG2D) and cytotoxicity [4] |
| Toxicity/Toxicokinetics |
1. In vitro cytotoxicity: KRN 7000 at concentrations up to 1 μg/mL had no inherent cytotoxicity against human PBMCs, CD1d⁺ APCs, or normal skin fibroblasts, with cell viability >95% vs. vehicle (MTT assay) [3] 2. In vivo toxicity: Intravenous administration of KRN 7000 (up to 10 μg/kg) in C57BL/6 mice caused no significant changes in body weight, food intake, or hematological parameters (WBC, RBC, platelets). Histopathological examination of liver, kidney, heart, and lung revealed no inflammation, necrosis, or abnormal proliferation [4] |
| References |
[1]. Going both ways: immune regulation via CD1d-dependent NKT cells. J Clin Invest. 2004 Nov;114(10):1379-88. [2]. Practical Total Synthesis of (2S,3S,4R)-1-O-(α-D-Galactopyranosyl)-N-hexacosanoyl-2-amino-1,3,4-octadecanetriol, the Antitumorial and Immunostimulatory α-Galactosylcer-amide, KRN7000. Biosci Biotechnol Biochem. 1996 Jan;60(2):288-92. [3]. In vitro anti-tumour activity of alpha-galactosylceramide-stimulated human invariant Valpha24+NKT cells against melanoma. Br J Cancer. 2001 Sep 1;85(5):741-6. [4]. Structure-Activity Relationship of .alpha.-Galactosylceramides against B16-Bearing Mice. J. Med. Chem. 1995, 38, 12, 2176-2187. |
| Additional Infomation |
1-O-(alpha-D-galactosyl)-N-hexacosanoylphytosphingosine is a glycophytoceramide having an alpha-D-galactosyl residue at the O-1 position and a hexacosanoyl group attached to the nitrogen. It has a role as an antineoplastic agent, an epitope, an antigen, an immunological adjuvant and an allergen. It is a glycophytoceramide and a N-acyl-beta-D-galactosylphytosphingosine. It is functionally related to an alpha-D-galactose. KRN7000 has been used in trials studying the treatment of Lung Cancer, Chronic Hepatitis C, Hepatitis B, Chronic, Unspecified Adult Solid Tumor, Protocol Specific, and Prevention of GvHD in Patients With Hematological Malignancies Undergoing AHSCT. alpha-GalCer has been reported in Bacteroides fragilis with data available. Alpha Galactosylceramide is a potent alpha galactosylceramide modified from marine-sponge that stimulates the immune system to exhibit antitumor activity. 1. KRN 7000 is a synthetic α-galactosylceramide (α-GalCer) with the chemical structure (2S,3S,4R)-1-O-(α-D-galactopyranosyl)-N-hexacosanoyl-2-amino-1,3,4-octadecanetriol. A practical total synthesis method was reported in literature [2] 2. Mechanism of action: KRN 7000 binds to the lipid-binding groove of CD1d molecules on APCs (dendritic cells, macrophages), forming a CD1d-KRN7000 complex. This complex is recognized by the T cell receptor (TCR) of invariant NKT (iNKT) cells, triggering rapid activation of iNKT cells to secrete IFN-γ (Th1 cytokine) and IL-4 (Th2 cytokine). The activated iNKT cells further activate NK cells, CTLs, and mature APCs, initiating a cascade of antitumor immune responses; simultaneously, it modulates immune balance to avoid excessive inflammation [1,3,4] 3. Therapeutic potential: Based on preclinical data, KRN 7000 has potential utility in tumor immunotherapy, particularly for melanoma and other CD1d⁺ or iNKT cell-responsive tumors. It also shows promise in immune regulation for autoimmune diseases, though relevant data are not provided in the specified literatures [3,4] 4. Research application: As a prototypical CD1d ligand, KRN 7000 is widely used as a tool compound to study the biological functions of the CD1d-iNKT cell pathway, including antitumor immunity, immune regulation, and antigen presentation mechanisms [1,3,4] 5. Structural feature: The α-anomeric configuration of the galactose moiety and the specific acyl chain lengths (N-hexacosanoyl, 26-carbon; sphingosine chain, 18-carbon) are critical for CD1d binding and iNKT cell activation. Modifications to acyl/sphingosine chains alter its biological activity [4] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~0.5 mg/mL (~0.58 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1651 mL | 5.8253 mL | 11.6507 mL | |
| 5 mM | 0.2330 mL | 1.1651 mL | 2.3301 mL | |
| 10 mM | 0.1165 mL | 0.5825 mL | 1.1651 mL |