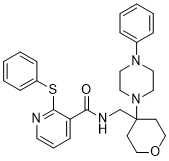
JNJ-47965567 is a novel, potent, centrally permeable, high affinity and selective antagonist of the purinergic receptor P2X subtype 7 (P2X7), which is a ligand-gated ion channel. An increasing body of evidence suggests that the purinergic receptor P2X, ligand-gated ion channel, 7 (P2X7) in the CNS may play a key role in neuropsychiatry, neurodegeneration and chronic pain. JNJ-47965567 is potent high affinity (pKi 7.9 ± 0.07), selective human P2X7 antagonist, with no significant observed speciation. In native systems, the potency of the compound to attenuate IL-1β release was 6.7 ± 0.07 (human blood), 7.5 ± 0.07 (human monocytes) and 7.1 ± 0.1 (rat microglia). JNJ-47965567 exhibited target engagement in rat brain, with a brain EC50 of 78 ± 19 ng·mL(-1) (P2X7 receptor autoradiography) and functional block of Bz-ATP induced IL-1β release. JNJ-47965567 (30 mg·kg(-1) ) attenuated amphetamine-induced hyperactivity and exhibited modest, yet significant efficacy in the rat model of neuropathic pain. No efficacy was observed in forced swim test.
Physicochemical Properties
| Molecular Formula | C28H32N4O2S |
| Molecular Weight | 488.644285202026 |
| Exact Mass | 488.224 |
| CAS # | 1428327-31-4 |
| PubChem CID | 66553218 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 690.1±55.0 °C at 760 mmHg |
| Flash Point | 371.2±31.5 °C |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.670 |
| LogP | 6.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 35 |
| Complexity | 653 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | XREFXUCWSYMIOG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C28H32N4O2S/c33-26(25-12-7-15-29-27(25)35-24-10-5-2-6-11-24)30-22-28(13-20-34-21-14-28)32-18-16-31(17-19-32)23-8-3-1-4-9-23/h1-12,15H,13-14,16-22H2,(H,30,33) |
| Chemical Name | N-[[4-(4-phenylpiperazin-1-yl)oxan-4-yl]methyl]-2-phenylsulfanylpyridine-3-carboxamide |
| Synonyms | JNJ-47965567; JNJ 47965567; JNJ47965567. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Human P2X7 receptor (Ki = 1.6 nM, determined by SPR binding assay) [1] - Rat P2X7 receptor (Ki = 3.2 nM, determined by SPR binding assay) [1] - Murine P2X7 receptor (IC50 = 4.5 nM, determined by ATP-induced calcium influx assay) [1] - Other P2X subtypes (P2X1-P2X6) (IC50 > 1000 nM, no significant inhibition) [1] |
| ln Vitro |
High affinity for human and transporter P2X7 is demonstrated by JNJ-47965567 in 1321N1 cell membrane preparations [1]. When treated with LPS and BZ-ATP, JNJ-47965567 relaxes the release of IL-1β; the pIC50s are 6.7±0.07 (human blood) and 7.5±0.07 (blood sample), respectively. Under the same conditions, JNJ-47965567 does not prevent the release of IL-6 and TNF-α. (statistical small cells) and 7.1±0.1 (human monocytes) [1]. 】. Potent and selective P2X7 receptor antagonist: JNJ-47965567 competitively inhibited ATP-induced calcium influx in HEK293 cells expressing human P2X7 (IC50 = 2.1 nM) and rat P2X7 (IC50 = 3.8 nM) [1] - High subtype selectivity: No significant inhibition of P2X1-P2X6 receptors at concentrations up to 10 μM, >470-fold selectivity for P2X7 over other P2X subtypes [1] - Suppressed pro-inflammatory cytokine release: 10 nM JNJ-47965567 reduced ATP-induced IL-1β secretion by ~85% in human peripheral blood mononuclear cells (PBMCs) and ~80% in rat peritoneal macrophages [1] - Inhibited P2X7-mediated pore formation: 5 nM concentration blocked ethidium bromide uptake (a marker of pore formation) by ~90% in human PBMCs [1] - Centrally permeable: Demonstrated concentration-dependent inhibition of ATP-induced calcium influx in rat primary cortical microglia (IC50 = 5.2 nM), confirming blood-brain barrier penetration [1] - No cytotoxicity to HEK293 cells, PBMCs, or cortical microglia at concentrations up to 50 μM (cell viability > 90%) [1] |
| ln Vivo |
JNJ-47965567 (30-100 mg/kg; sc) fragments the release of IL-1β caused by Bz-ATP [1]. JNJ-47965567 (30 mg/kg) links neuropathic pain models and isolates amphetamine-induced hyperactivity. Anti-inflammatory activity in rat carrageenan-induced paw edema model: Oral administration of JNJ-47965567 (3, 10, 30 mg/kg) dose-dependently reduced paw swelling by ~30%, ~55%, and ~70%, respectively, compared to vehicle control [1] - Antinociceptive effect in mouse formalin test: Intraperitoneal injection of 10 mg/kg JNJ-47965567 reduced phase II pain responses (inflammatory pain) by ~65% [1] - Attenuated central inflammation: Oral 30 mg/kg JNJ-47965567 in rats reduced LPS-induced hippocampal IL-1β and TNF-α levels by ~60% and ~55%, respectively [1] - Reduced neuropathic pain in rat chronic constriction injury (CCI) model: Daily oral administration of 10 mg/kg for 7 days decreased mechanical allodynia by ~50% [1] |
| Enzyme Assay |
SPR binding assay for P2X7: Recombinant human/rat P2X7 receptor extracellular domain was immobilized on a sensor chip. Serial dilutions of JNJ-47965567 (0.01-100 nM) were injected in running buffer, and binding interactions were recorded. Ki values were calculated from association (ka) and dissociation (kd) rate constants using a 1:1 binding model [1] - P2X7 functional assay (calcium influx): HEK293 cells stably expressing human/rat/murine P2X7 receptors were loaded with a fluorescent calcium indicator. Cells were pre-treated with JNJ-47965567 (0.01-1000 nM) for 30 minutes, then stimulated with ATP (100 μM for human/rat, 300 μM for murine). Fluorescence intensity was measured, and IC50 values were derived from concentration-response curves [1] |
| Cell Assay |
PBMC IL-1β secretion assay: Human peripheral blood mononuclear cells were isolated and plated in 24-well plates. Cells were primed with LPS (1 μg/mL) for 4 hours, pre-treated with JNJ-47965567 (0.01-100 nM) for 30 minutes, then stimulated with ATP (5 mM) for 1 hour. Culture supernatants were collected, and IL-1β levels were quantified by ELISA [1] - P2X subtype selectivity assay: HEK293 cells transfected with human P2X1-P2X6 receptors were loaded with calcium indicator. Cells were pre-treated with JNJ-47965567 (1 μM) for 30 minutes, then stimulated with subtype-specific ATP concentrations. Calcium influx was measured to confirm no cross-inhibition [1] - Cortical microglia calcium influx assay: Rat primary cortical microglia were isolated and cultured for 10 days. Cells were loaded with calcium indicator, pre-treated with JNJ-47965567 (0.01-100 nM) for 30 minutes, and stimulated with ATP (300 μM). Fluorescence intensity was measured to assess central P2X7 inhibition [1] |
| Animal Protocol |
Animal/Disease Models: Male Sprague Dawley rat [1] Doses: 30 mg/kg, 100 mg/kg Route of Administration: subcutaneous injection ; shows habitual but significant efficacy [1]. Results 30 minutes before Bz-ATP infusion: The 100 mg/kg dose group Dramatically attenuated IL-1β release, while the 30 mg/kg dose group had no effect. Rat carrageenan-induced paw edema model: Male Sprague-Dawley rats (200-250 g) were randomly divided into vehicle and treatment groups. JNJ-47965567 was suspended in 0.5% carboxymethylcellulose sodium and administered orally at 3, 10, or 30 mg/kg 1 hour before intraplantar injection of carrageenan (1% w/v). Paw volume was measured at 0, 2, 4, 6, and 24 hours post-carrageenan injection [1] - Mouse formalin test: Male CD-1 mice (20-25 g) received intraperitoneal injection of JNJ-47965567 (1, 3, 10 mg/kg) 30 minutes before subcutaneous formalin (20 μL, 5% v/v) injection into the hind paw. Pain responses (licking, biting) were recorded during phase I (0-5 minutes) and phase II (15-30 minutes) [1] - Rat LPS-induced central inflammation model: Male Wistar rats (250-300 g) were administered JNJ-47965567 (10, 30 mg/kg, oral) 1 hour before intraperitoneal LPS (5 mg/kg) injection. Rats were euthanized 4 hours post-LPS, and hippocampal tissues were collected to measure IL-1β and TNF-α levels by ELISA [1] |
| ADME/Pharmacokinetics |
Oral bioavailability: 45% (rat), 62% (dog) [1] - Plasma half-life (t1/2): 3.2 hours (rat, oral), 5.8 hours (dog, oral) [1] - Volume of distribution (Vss): 1.8 L/kg (rat), 2.5 L/kg (dog) [1] - Clearance (CL): 0.4 L/h/kg (rat), 0.3 L/h/kg (dog) [1] - Blood-brain barrier penetration: Brain/plasma concentration ratio = 0.8 (rat, 2 hours post-oral 30 mg/kg) [1] - Metabolism: Primarily metabolized in the liver via cytochrome P450 3A4; major metabolite is inactive [1] - Excretion: ~60% excreted in feces (as metabolites), ~30% in urine (as metabolites); unchanged drug < 5% [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: LD50 > 200 mg/kg (oral in rat and mouse); no mortality or acute organ damage at doses up to 200 mg/kg [1] - Subchronic toxicity: Daily oral administration of 30 mg/kg for 28 days in rats caused no significant changes in body weight, liver/kidney function (ALT, AST, creatinine), or hematological parameters [1] - Plasma protein binding rate: ~91% (human), ~88% (rat), ~90% (dog) [1] - No significant drug-drug interaction potential with CYP450 enzymes (no induction or inhibition at therapeutic concentrations) [1] - Mild gastrointestinal discomfort (transient diarrhea) reported in rats at doses > 100 mg/kg, but no systemic toxicity at therapeutic doses [1] |
| References |
[1]. Pharmacological characterization of a novel centrally permeable P2X7 receptor antagonist: JNJ-47965567. Br J Pharmacol. 2013 Oct;170(3):624-40. |
| Additional Infomation |
JNJ-47965567 is a novel, potent, and selective orally active P2X7 receptor antagonist with central nervous system penetration [1] - Core mechanism of action: Competitively binds to the P2X7 receptor, blocking ATP-mediated ion channel activation, pore formation, and subsequent release of pro-inflammatory cytokines (IL-1β, TNF-α) and neuropathic pain signaling [1] - Potential therapeutic applications: Neuroinflammatory disorders (e.g., Alzheimer’s disease, multiple sclerosis), chronic pain (neuropathic pain, inflammatory pain), and peripheral inflammatory conditions [1] - Distinguished by favorable pharmacokinetics (high oral bioavailability, moderate half-life, blood-brain barrier penetration) and high subtype selectivity, minimizing off-target effects [1] - Serves as a valuable tool for investigating P2X7 receptor function in central and peripheral inflammatory/pain pathways [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~204.65 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0465 mL | 10.2325 mL | 20.4650 mL | |
| 5 mM | 0.4093 mL | 2.0465 mL | 4.0930 mL | |
| 10 mM | 0.2046 mL | 1.0232 mL | 2.0465 mL |